A conceptual graphic shows how growth factors BMP2 and FGF2 are applied to the injury site to stimulate tissue regeneration, highlighting new research into restoring damaged digits. (Credit: Texas A&M University)
Two Proteins, Applied in the Right Order, Helped Mice Regrow Amputated Finger Bones
In A Nutshell
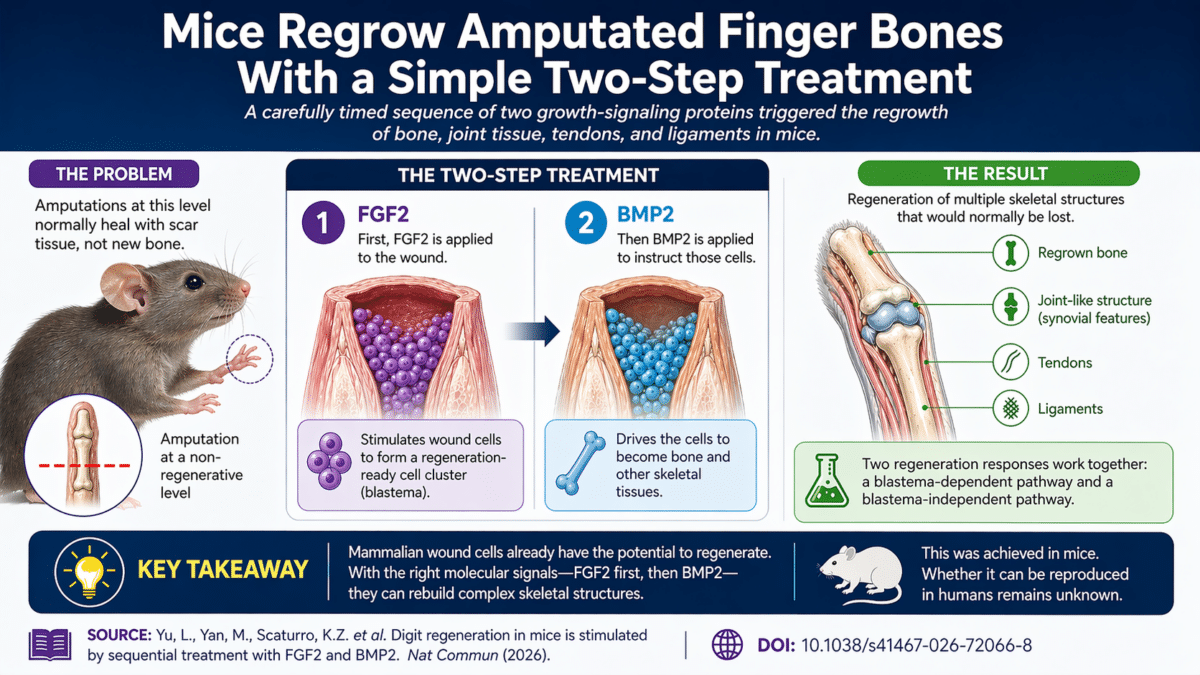
- Researchers triggered bone, joint, tendon, and ligament regrowth in amputated mouse digits using a two-step treatment with proteins FGF2 and BMP2, applied in sequence.
- Mammals were long thought incapable of regrowing lost skeletal structures, healing with scar tissue instead. This study challenges that assumption.
- Two separate regeneration processes occurred simultaneously: one rebuilt bone, the other produced a joint-like structure with features of a natural finger joint.
- Mammalian wound cells appear to already carry regenerative potential. They may simply need the right molecular signal, delivered in the right order, to act on it.
Salamanders can regrow entire limbs and Starfish can regenerate lost arms. But mammals? They’ve (We’ve) long been considered out of luck when it comes to regrowing lost body parts. Now, however, a team of researchers working with mice coaxed the regrowth of amputated finger bones using a carefully timed, sequential treatment with two growth-signaling proteins. The result challenges the longstanding assumption that mammalian wounds are destined to heal with scar tissue instead of new structures.
Published in Nature Communications, the study found that treating amputated mouse digits first with a protein called FGF2 and then with a protein called BMP2 triggered the regrowth of bone, joint tissue, tendons, and ligaments. In some cases, the combined response covered multiple skeletal structures removed by the amputation. The regrowth wasn’t perfect. But the fact that it happened at all, at a wound site that would normally just scar over, marks a real shift in how scientists think about regeneration in mammals.
How Two Proteins Triggered Bone Regrowth in Mice
To grasp why this matters, it helps to know a bit about how regeneration works in animals that are good at it. Salamanders regrow limbs by forming a cluster of unspecialized cells at a wound site. That cluster then reorganizes into the missing body part. Mammals can do a limited version of this at the very tips of their fingers and toes. Children who lose fingertips in accidents sometimes regrow them. But amputation at slightly deeper levels results in scarring, not regrowth.
Led by senior researcher Ken Muneoka and colleagues at Texas A&M University, Tulane University, Arizona State University, Stanford University, and the Ludwig Boltzmann Institute for Traumatology in Vienna, the team tested what would happen if they amputated mouse digits at a level that does not normally regenerate, then applied growth-signaling proteins to the wound. FGF2, delivered first, stimulated wound cells to form a regeneration-ready cluster, kick-starting the process. BMP2, a bone-building signal, was then applied to instruct those cells to become bone and other tissues.
Order mattered. FGF2 first, then BMP2. That one-two sequence drove the regrowth.
Cell-tracking studies revealed that wound cells may have been reprogrammed, diverted from their default scarring path toward rebuilding lost bone and joint. Evidence from the study points to organized rebuilding that appeared to mirror how bones originally form during embryonic development.
Two Separate Regeneration Responses at the Same Wound Site
Perhaps most unexpectedly, the researchers found the treatment triggered two distinct regeneration responses at the same time. One followed the expected pathway: wound cells formed a cluster that then became the missing bone segment. The second was entirely different. It regenerated a joint-like structure with features of a synovial joint, the lubricated, hinge-type joint found between finger bones, along with tendon, ligament, and a small bone similar to a kneecap.
Together, the two responses could account for multiple skeletal components removed by the amputation. The paper notes the regeneration was “imperfect,” but the range of tissue types that grew back from a wound that would otherwise have simply scarred over is notable.
The Cells Were There All Along
One of the big questions in regeneration research has been whether mammals lack the right cells for regeneration, or whether they simply lack the right signals. This study makes a strong case for the second option. As the researchers write, the induced regeneration response “demonstrates the availability of regeneration competent cells at a non-regenerating wound, and that FGF and BMP signaling is sufficient to trigger a regenerative outcome at wounds that heal by fibrosis.”
Put plainly, the raw materials for regeneration are already sitting at the wound. The cells just need to be told what to do.
Scarring is the default mammalian wound-healing response. Cut yourself, break a bone badly, or lose a body part, and the body patches things up with fibrous tissue rather than rebuilding what was lost. Scientists have often described this as an evolutionary tradeoff: fast wound closure at the expense of true regeneration. The sequential FGF2-then-BMP2 approach appears to override that default, steering the wound response from scarring toward regrowth.
What Mouse Finger Bone Regeneration Could Mean
Translating findings from mice to humans is always a leap, and this study is a long way from being used in a clinic. Whether this approach would work in larger animals or humans remains unknown. FGF2 and BMP2 are not exotic molecules; both are well-studied growth factors already used in various medical settings. But no one should expect to regrow a lost finger anytime soon.
Still, the core finding, that mammalian wound cells may retain the ability to regenerate when given the right molecular instructions in the right order, reopens a question many had considered settled. Regenerative failure in mammals now looks less like a hard biological limit and more like a problem of missing signals. Getting those signals right in humans is where the real work begins.
Paper Notes
Limitations
The study demonstrates regeneration of digit skeletal structures in mice, but the authors explicitly note the regeneration achieved was “imperfect.” The paper does not detail the specific ways the regenerated structures differ from the originals, though the use of that qualifier indicates the outcome did not fully replicate the pre-amputation anatomy. The work was performed in neonatal mice, and the extent to which these findings would translate to larger mammals or humans remains unknown. The study focused on digit amputations at a specific non-regenerative level, and whether the same approach would work for more extensive amputations or other types of tissue loss is unclear.
Funding and Disclosures
Research was supported by grants from DARPA, the US Army Research Center, the National Institutes of Health, the John L. and Mary Wright Ebaugh Endowment Fund at Tulane University, and Texas A&M University. The authors declare no competing interests.
Publication Details
Title: Digit regeneration in mice is stimulated by sequential treatment with FGF2 and BMP2 | Authors: Ling Yu, Mingquan Yan, Katherine Zimmel Scaturro, Osama Qureshi, Yu-Lieh Lin, Benjamin B. Bartelle, C. Addison Smith, Daniel Osorio Hurtado, James J. Cai, Lindsay A. Dawson, Regina Brunauer, Larry J. Suva, Manjong Han, Connor P. Dolan, and Ken Muneoka | Journal: Nature Communications (2026) | DOI: 10.1038/s41467-026-72066-8 | Received: May 27, 2025 | Accepted: April 8, 2026 | Published: April 17, 2026 | Corresponding Author: Ken Muneoka | Institutional Affiliations: Texas A&M University (College Station, TX); Stanford University School of Medicine (Stanford, CA); Arizona State University (Tempe, AZ); Ludwig Boltzmann Institute for Traumatology (Vienna, Austria); Tulane University (New Orleans, LA) | Data Availability: RNA sequencing data deposited in GEO under accession number GSE297901.







