
Photo by Gabriel Dalton on Unsplash
In A Nutshell
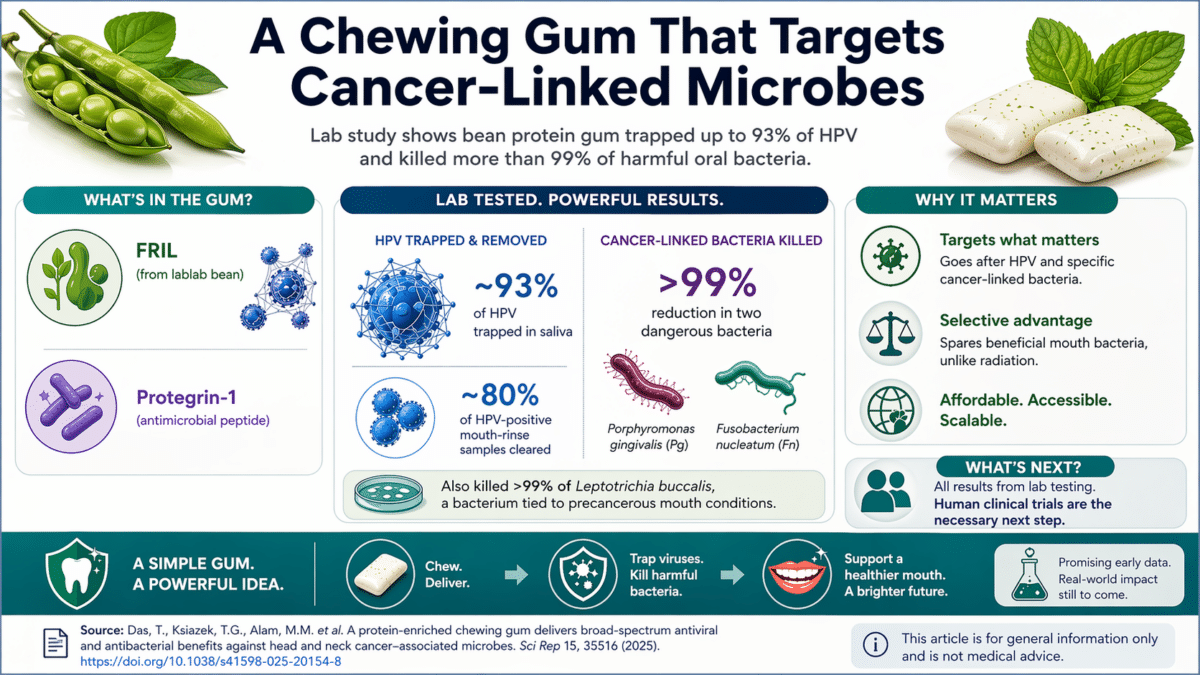
- Researchers tested a chewing gum made with a bean protein and an antimicrobial peptide on saliva and mouth-rinse samples from head and neck cancer patients: and it wiped out more than 99% of two cancer-linked bacteria while trapping up to 93% of HPV.
- All testing was done in a laboratory on patient samples, not in living people. Human clinical trials are the necessary next step before anyone can know whether the gum works as hoped.
- The gum spared the mouth’s healthy, beneficial bacteria, giving it a selectivity advantage over radiation therapy, which can disrupt the entire oral microbial community.
A stick of chewing gum probably isn’t the first thing that comes to mind when thinking about cancer prevention. But a new study suggests it might be worth a second look. Researchers loaded a gum with proteins derived from an edible bean and tested it against the specific viruses and bacteria most strongly tied to head and neck cancer. In lab experiments using real patient samples, the results were notable enough to push this idea toward clinical trials.
All of this work was done in a laboratory, not in living patients. No one chewed the gum and was monitored for health outcomes. Human clinical trials will be needed before anyone can say whether it works in practice. Still, the early data offers an intriguing glimpse of what might be possible.
Head and neck cancer, the kind that strikes the mouth, throat, and tonsils, accounts for an estimated 890,000 new cases worldwide every year, with roughly half of patients dying after treatment. Radiation can disrupt the mouth’s bacterial balance, reducing helpful bacteria and allowing harmful microbes to increase. Scientists have known for years that three specific oral microbes, HPV and two types of bacteria called Porphyromonas gingivalis (Pg) and Fusobacterium nucleatum (Fn), show up at much higher levels in cancer patients and are tied to worse outcomes. The researchers behind this study argue that practical, affordable ways to lower these microbes in the mouth are still needed.
A team from the University of Pennsylvania, UCLA, the Veterans Administration Greater Los Angeles Healthcare System, and the University of Kansas Medical Center set out to address that. Their approach was simple in concept: load a chewing gum with plant-derived proteins that attack cancer-associated microbes right where they live. The results, published in Scientific Reports, suggest these gums could one day serve as a powerful add-on to existing cancer treatments or even as a preventive tool for people at high risk.
How the Bean-Based Chewing Gum Study Worked
This was a lab-based study, meaning the researchers collected real biological samples from cancer patients and healthy volunteers, then tested the gum ingredients on those samples rather than having patients chew the gum directly. Saliva samples came from patients treated at the VA hospital in Los Angeles, while mouth-rinse samples were collected at the University of Kansas Medical Center. In total, the study drew on samples from dozens of head and neck cancer patients and cancer-free participants.
The gum contains two active ingredients. The first is a protein called FRIL, found naturally in the lablab bean, a legume eaten around the world. FRIL works like a molecular trap: it latches onto sugar structures on the surface of viruses, clumping virus particles together into large clusters that can no longer infect cells. The second ingredient is protegrin-1, an antimicrobial peptide first identified in pigs that has already been tested in advanced human clinical trials for mouth sores caused by chemotherapy. Protegrin-1 is especially effective at killing certain types of bacteria thanks to its shape, which is locked in place by chemical bonds that make it unusually stable.
To test the antiviral gum, the team mixed bean gum extract with patient samples and measured how much HPV remained free in the liquid. For the antibacterial tests, they combined the bean gum extract with protegrin-1 and let the mixture sit with patient samples for one hour at body temperature. They then grew bacteria from the treated and untreated samples on specialized plates designed to detect specific microbes and counted the colonies that formed.
Bean Gum Cleared HPV and Killed 99% of Cancer-Linked Oral Bacteria
The HPV results were eye-opening. Researchers found the virus in every single saliva sample from cancer patients and in about 75% of mouth-rinse samples. When treated with the bean gum extract, about 93% of HPV in saliva was trapped and removed. In mouth-rinse samples, about 80% of HPV-positive cases showed the virus being clumped together, with 13 out of 22 positive samples showing complete virus removal. Spinning the samples at high speed without the bean extract did nothing to remove the virus. It was the FRIL protein doing the work.
Cancer patients harbored staggering amounts of the two dangerous bacteria compared to cancer-free participants. Saliva from cancer patients contained roughly 1,000 times more Pg and Fn than samples from healthy controls, and mouth-rinse samples showed about 100 times more. A single dose of the bean gum plus protegrin-1 combination wiped out more than 99% of both bacteria across all sample types, a reduction the researchers described as statistically highly significant.
Just as telling as what the treatment killed is what it spared. Streptococcus bacteria, which include many species that are normal, healthy residents of the mouth, survived the treatment largely intact. Many Streptococcus species produce protective sugar coatings that shield them from protegrin-1’s attack. The bacteria linked to cancer, Pg and Fn, lack this protection, making them vulnerable. That built-in selectivity is a real advantage over radiation therapy, which can disrupt the entire oral bacterial community without discrimination.
The study also screened for other microbes. A bacterium called Leptotrichia buccalis, tied to precancerous mouth conditions, turned up in 97% of cancer patients’ mouth-rinse samples at levels 100 times higher than in healthy participants, and the treatment killed more than 99% of it. A yeast sometimes linked to oral cancer, Candida albicans, appeared at low rates in cancer patients and was absent from all healthy participant samples.
Why a Chewing Gum Could Go Where HPV Vaccines Cannot
One-third of men globally are infected with HPV according to a major review of 65 studies spanning 45,000 males across 35 countries, and one-fifth carry the high-risk HPV-16 strain most commonly linked to cancer. Although HPV vaccines exist in more than 100 countries, the rate of throat cancer has actually increased since vaccination programs began. HPV vaccines are injected into muscle and trigger a body-wide immune response, but they do not produce the type of immune defense that protects the moist surfaces of the mouth and throat. Vaccinated individuals can still harbor and transmit the virus orally. A gum that traps and removes HPV directly in the mouth could, in theory, offer a layer of protection that vaccines currently cannot, though this potential has not yet been tested in real-world conditions.
Safety is also part of the picture. The bean powder carries an FDA designation as “Generally Regarded as Safe,” and the dose in a single gum tablet is more than 6,000 times less than the amount consumed safely in human toxicology testing. The chewing gum formula has already been submitted to the FDA and approved for evaluation in a clinical trial targeting coronavirus infection and transmission. The individual components have established safety profiles, but the effectiveness and safety of this specific formulation for targeting oral cancer microbes still need to be validated in human trials.
Beyond the surface of the mouth, the researchers point out that FRIL can enter human cells and block viruses at a deeper level, and that protegrin-1 has been shown to get inside head and neck cancer cells. Prolonged local delivery through chewing could, in theory, fight microbes both outside and inside cancer cells. Whether this plays out in a patient’s mouth remains to be demonstrated.
It’s also worth noting the bean gum tablets used in these experiments had been stored at room temperature for up to 33 months and still worked. That kind of shelf stability could make the product practical and affordable at a global scale, particularly given that plant-based medicines approved by the FDA cost less than 3% of comparable drugs produced through other methods.

A Different Approach to a Deadly Problem
Radiation kills cancer cells but can also disrupt the mouth’s bacterial balance, sometimes worsening the microbial environment long after treatment ends. A chewing gum that selectively clears the specific virus and bacteria most strongly associated with head and neck cancer, while leaving the mouth’s healthy residents alone, is a meaningfully different kind of tool. It would not replace surgery or radiation. It would go after the microbial actors that help cancer start, spread, and come back.
Getting from a lab dish to a patient’s mouth is a significant leap, and the science is nowhere near the finish line. But few ideas in this space are as low-cost, low-barrier, and broadly deployable as a piece of gum, and the early data gives researchers a legitimate reason to keep going.
Disclaimer: This article is based on a laboratory study using patient-derived saliva and mouth-rinse samples. No human participants chewed the gum or were monitored for health outcomes. The findings have not been replicated in clinical trials, and the gum has not been approved by the FDA to prevent, treat, or cure any disease. Readers should not interpret these results as evidence that the product is safe or effective for human use in a cancer context. Anyone with concerns about head and neck cancer, HPV, or oral health should consult a qualified medical professional.
Paper Notes
Limitations
The authors acknowledge several important limitations. This was a lab-based study, meaning the gum ingredients were tested on patient samples in a laboratory setting rather than having patients actually chew the gum and monitoring changes in their mouths over time. It is not a human clinical study designed to track long-term changes in the mouth’s bacterial environment. Future clinical studies will be needed to provide that data. Additionally, the current study evaluated saliva and mouth-rinse samples but did not examine tumor tissue directly. Future work will need to assess whether FRIL and protegrin-1 can get inside tumor cells and kill HPV or dangerous bacteria within them. The researchers also noted that gum disease was not examined or recorded in cancer patients at the medical clinic, meaning an increase in P. gingivalis due to gum disease could not be ruled out as a contributing factor. The detection method used measures only the virus-trapping effect of FRIL, not its ability to block viruses at a deeper cellular level, meaning the full antiviral strength may be underestimated by the results presented.
Funding and Disclosures
Funding for research in the Daniell lab at the University of Pennsylvania was provided by NIH grant 5-R01-HL 107904-13 awarded to Henry Daniell. Sample collection at the VA Greater Los Angeles Healthcare System was supported by the Surgical Education Research Program. The Thomas lab’s sample collection was supported in part through the University of Kansas Cancer Center, National Cancer Institute Cancer Center Support Grant P30 CA168524. Lead author Henry Daniell is a patentee in plant-based oral and topical drug delivery and was previously funded by Novo Nordisk, Bayer, Shire, and Takeda, though no specific financial conflict related to this manuscript was declared. Bean gum tablets were manufactured by Per Os Biosciences. The chewing gum composition has been submitted to the FDA (IND 154897) and approved for evaluation of coronavirus infection and transmission in a clinical trial (NCT05433181).
Publication Details
Title: Ex vivo HNSCC clinical studies using saliva and antiviral or antibacterial chewing gums reveal reduction in carcinogenic microbes | Authors: Henry Daniell, Geetanjali Wakade, Rahul Singh, Smruti Nair, Saroj K. Basak, Eri S. Srivatsan, Andrés M. Bur, Sufi M. Thomas, and Marilene B. Wang | Affiliations: Department of Basic and Translational Sciences, School of Dental Medicine, University of Pennsylvania, Philadelphia, PA; Department of Surgery, Veterans Administration Greater Los Angeles Healthcare System, Los Angeles; The Molecular Biology Institute, Los Angeles; Department of Head and Neck Surgery, David Geffen School of Medicine, University of California at Los Angeles, Los Angeles, CA; Department of Otolaryngology, University of Kansas Medical Center, Kansas City, KS | Journal: Scientific Reports (2026), 16:7886 | DOI: 10.1038/s41598-026-39062-w







