An illustration of a neuron, one of the brain’s key cells for processing information. (Vector_Artist/Shutterstock)
A New Tool Can Forge Precise Links Between Brain Cells. It Changed Mouse Behavior.
In A Nutshell
- Researchers engineered two fish proteins into an exclusive electrical connector, called LinCx, that links only two targeted brain cell types without accidentally connecting to other cells.
- When tested in worms, LinCx successfully coupled two specific neurons and predictably changed the animals’ temperature-seeking behavior.
- In mice, LinCx strengthened communication between two targeted cell types in the prefrontal cortex and produced measurable changes in social behavior and stress responses.
- This is early-stage research conducted in animals under lab conditions; LinCx has not been tested in disease models or evaluated for human safety.
For years, neuroscientists have dreamed of a tool precise enough to strengthen the connection between just two specific types of brain cells without accidentally linking to anything else. In specific worm and mouse circuits, a team of researchers has now shown that they can do exactly that, using proteins borrowed from a fish to build an engineered electrical link that works with a level of accuracy not previously possible in mammals.
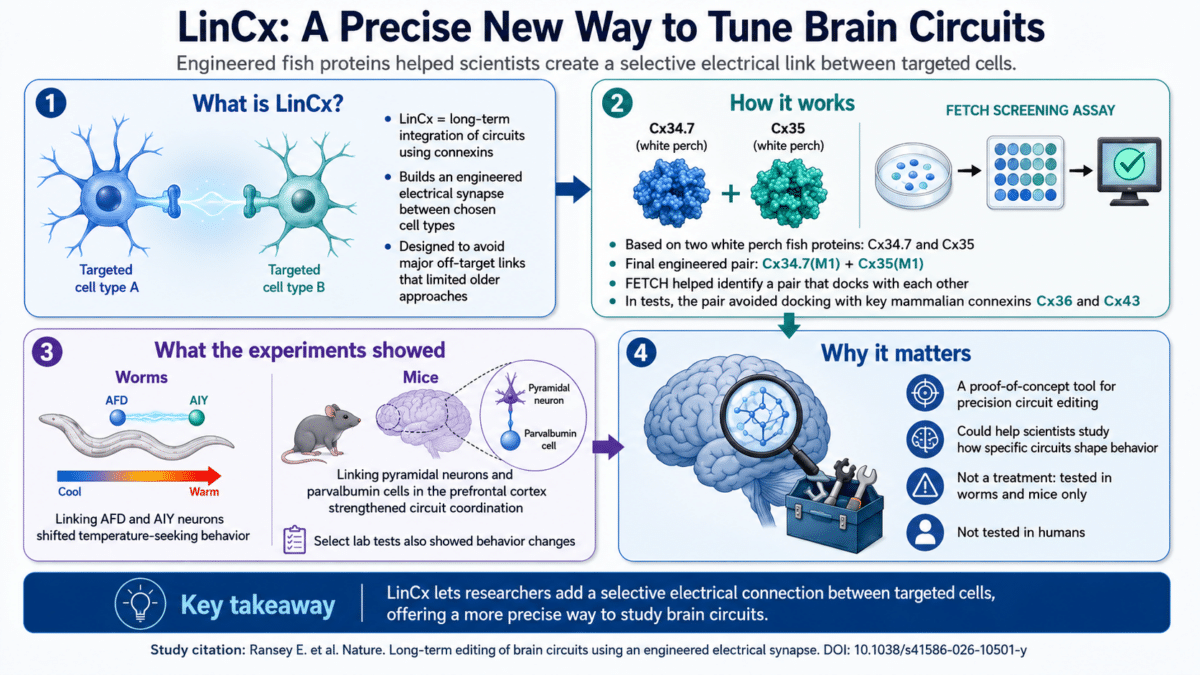
Described in the journal Nature, the tool is called LinCx, short for “long-term integration of circuits using connexins.” It works by forming an exclusive electrical connection between two chosen cell types, strengthening how they communicate and steering behavior in a predictable direction.
Brain cells communicate through several mechanisms. Some use chemical signals. Others use direct electrical links built from proteins called connexins, which act like physical bridges between cells. Earlier attempts to use these bridges as a circuit-editing tool ran into a fundamental problem: the proteins were not selective enough. They would connect with the wrong cells and produce unintended effects. LinCx was built specifically to get around that barrier.
How Scientists Built a More Precise Brain Circuit Tool
To solve the off-target problem, the research team needed proteins that would connect only with each other and not with the connexins already present in mammalian brains. They turned to two connexin proteins from the white perch fish, Morone americana, called connexin 34.7 and connexin 35. These two proteins naturally prefer to link with each other rather than with themselves, a relatively rare property that made them a promising starting point.
From there, the team introduced roughly 70 individual changes across both proteins and tested each variation. To screen the results, they developed an original method called FETCH, which tags each connexin protein with a different color of fluorescent dye, places the two cell populations together, and measures how much color gets exchanged. Color exchange means the proteins connected; no exchange means they did not. This let researchers rapidly screen dozens of variants.
After that screening and additional computational modeling, the team arrived at a final engineered pair: Cx34.7(M1) and Cx35(M1). In the team’s tests, these mutant proteins docked with each other effectively and did not meaningfully connect with two major mammalian brain connexins, Cx36 and Cx43.
LinCx Changed Worm Behavior in Predictable Ways
Before moving to mice, the researchers tested the tool in the roundworm Caenorhabditis elegans, a millimeter-long creature widely used in neuroscience because its nervous system is extremely well mapped. Worms do not naturally produce connexin proteins, making it a clean test environment.
A known quirk in worm behavior helped here: worms trained at a particular temperature while food is present will later seek out that temperature, a preference controlled by the connection between a heat-sensing neuron called AFD and a downstream neuron called AIY. When the team inserted one mutant protein into AFD cells and the other into AIY cells, the two neurons became electrically coupled and the worms began persistently migrating toward warmer temperatures. When both neurons received the same mutant protein instead of the complementary pair, nothing changed, confirming that the effect required both halves working together. Pairing either mutant protein with Cx36 or Cx43 also produced no behavioral shift.
What the Mouse Experiments Revealed About Brain Circuit Editing
To test whether LinCx could work in the far more complex mammalian brain, the team moved to mice. They focused on a well-studied circuit in the prefrontal cortex involving excitatory pyramidal neurons and fast-spiking cells that express a protein called parvalbumin. These two cell types form a loop tied to attention, memory, and other cognitive functions.
Modified viruses delivered the two connexin proteins so that an electrical connection could only form where a pyramidal neuron met a parvalbumin cell. Brain activity recorded from the prefrontal cortex showed that mice carrying the full LinCx system had significantly higher coordination between slower and faster brain wave frequencies compared with control mice, with no change in the raw strength of those waves. This pointed to an effect on circuit-level coordination rather than a broad change in overall brain activity.
Behavior followed. Mice with LinCx in this circuit showed higher social preference and more exploration in a novel open field, without a broad change in movement after habituation. In a separate experiment targeting a long-range circuit between two other brain regions, mice with LinCx showed less stress-related behavioral adaptation during repeated tail-suspension tests, while general movement did not differ between groups.
Fish Proteins, Redesigned From Scratch, Clear a Long-Standing Barrier in Neuroscience
This is proof of concept, not a treatment. The experiments were conducted in worms and mice under controlled laboratory conditions, and the study was not designed to answer the safety questions that would matter for any future human use. LinCx has also not been tested in any disease model, and it only works between cells already in physical contact.
Even so, building a precise, exclusive electrical connection between two defined cell types in a living mammalian brain, one that measurably changes both circuit activity and behavior, moves the field meaningfully forward. Off-target connections were the fundamental barrier that undermined earlier approaches, and a pair of proteins from a fish, redesigned from the ground up, may now have a credible answer to that problem.
Disclaimer: This article is based on a peer-reviewed research study. It is intended for informational purposes only and should not be taken as medical advice. The research was conducted in animals under laboratory conditions and has not been tested in humans.
Paper Notes
Limitations
The authors note several important limitations. LinCx is only suitable for editing circuits composed of cells that make physical contact with each other. Because the connexin channels are chronically expressed, the authors also anticipate that LinCx induces activity-dependent changes in local chemical synapses, and this plasticity may contribute to some of the physiological and behavioral effects observed. One of the mutant proteins, Cx34.7(M1), showed inconsistent behavior under homotypic conditions across different experimental systems, an issue the authors attribute to differences between the Xenopus oocyte model and mammalian cell systems. The study also found preliminary signals that the engineered proteins may interact with Cx31.3, a connexin expressed in the mammalian brain, though the functional significance of this potential interaction remains uncharacterized. The study does not address long-term safety or immunogenicity associated with viral delivery.
Funding and Disclosures
Funding sources include the Ernest E. Just Life Science Institute Postdoctoral Research Fellowship, the Burroughs Wellcome Fund, the Hartwell Postdoctoral Research Fellowship, multiple NIH grants, the Howard Hughes Medical Institute, and institutional grants from Duke University. The FETCH method and LinCx proteins are subjects of two US patent applications filed by Duke University (US20250186620A1 and US20240248078A1). The authors declare no competing interests beyond those patent applications.
Publication Details
Paper Title: Long-term editing of brain circuits using an engineered electrical synapse | Authors: Elizabeth Ransey, Gwenaëlle E. Thomas, Elias M. Wisdom, Agustin Almoril-Porras, Ryan Bowman, Elise Adamson, Kathryn K. Walder-Christensen, Jesse A. White, Dalton N. Hughes, Hannah Schwennesen, Caly Ferguson, Kay M. Tye, Stephen D. Mague, Longgang Niu, Zhao-Wen Wang, Daniel Colón-Ramos, Rainbo Hultman, Nenad Bursac, and Kafui Dzirasa | Author Affiliations: Howard Hughes Medical Institute; Duke University Medical Center (Departments of Psychiatry and Behavioral Sciences, Neurobiology, Biomedical Engineering, and Neurosurgery); Yale University School of Medicine; Salk Institute for Biological Studies; University of Connecticut School of Medicine; Universidad de Puerto Rico; University of Iowa | Journal: Nature | DOI: https://doi.org/10.1038/s41586-026-10501-y | Received: August 9, 2023 | Accepted: April 7, 2026







