
(© Ivelin Radkov - stock.adobe.com)
In A Nutshell
- Up to 30% of older adults die with significant Alzheimer’s brain damage but no memory loss, and scientists still don’t know why their brains stay sharp.
- An AI tool analyzed thousands of human brain samples to build a genetic signature of Alzheimer’s, then used it to identify a mouse model that replicates this silent resilience.
- Male mice without the protein Chromogranin A showed Alzheimer’s-like brain changes but normal memory; female mice without it showed even broader protection, including far fewer toxic Tau tangles.
- The findings point to a new research direction: rather than trying to clear Alzheimer’s damage after the fact, it may be possible to strengthen the brain’s natural defenses before decline begins.
Up to three in ten older adults die with significant amyloid plaques and Tau tangles in their brains yet never showed a single sign of mental decline while alive. Scientists have now identified a new mouse model that mimics this puzzling resilience, and the discovery was guided by artificial intelligence.
For decades, Alzheimer’s research has been shadowed by a strange paradox. Autopsies of elderly people who were mentally sharp until the day they died sometimes reveal brains loaded with the same protein damage found in patients with devastating dementia. An estimated 20 to 30 percent of cognitively intact older adults carry this level of brain pathology yet never lose their memory or thinking abilities, a condition researchers call “asymptomatic Alzheimer’s disease.” Nobody fully understands why some brains hold up while others collapse under the same burden.
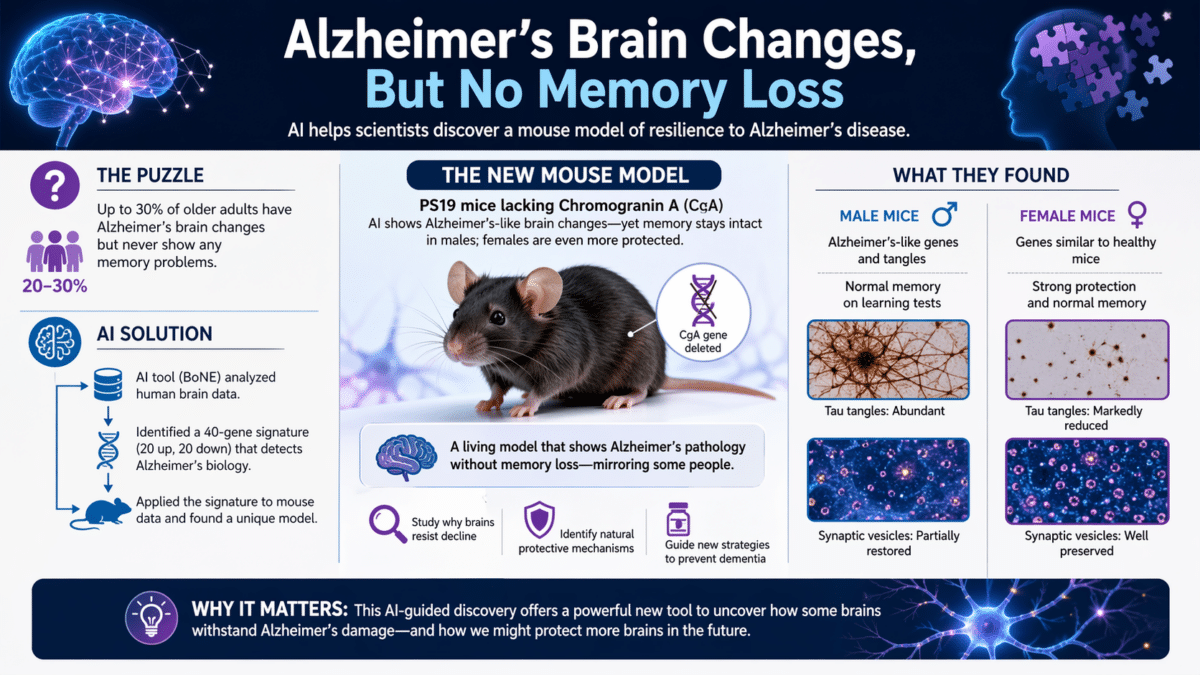
A team at the University of California San Diego and Weill Cornell Medicine identified a living laboratory mouse that carries the molecular fingerprints of Alzheimer’s in its brain but learns and remembers just fine. The discovery came from an AI system trained to decode Alzheimer’s at the genetic level, sorting through large databases of human brain tissue data to identify a reliable genetic signature of the disease. Researchers then applied that signature to established mouse strains, revealing one that could serve as a stand-in for this silent form of Alzheimer’s. Having such a model opens the door to studying resilience directly and to developing treatments that could protect the brain before irreversible damage sets in.
AI Identified a New Alzheimer’s Mouse Model Using Gene Logic
The study, published in Acta Neuropathologica Communications, used a computational tool called the Boolean Network Explorer, or BoNE. Rather than looking for loose statistical correlations between genes, BoNE identifies strict, directional relationships between pairs of genes. These logical rules hold more stable across datasets than many traditional approaches, making them far less likely to shift depending on which group of patients is studied.
Researchers fed BoNE data from 70 cognitively normal individuals and 219 Alzheimer’s cases, and the tool produced a network of gene relationships capturing the core biology of the disease. From this, the team distilled a signature of 40 genes: 20 that ramp up in Alzheimer’s and 20 that dial down. When tested against 35 independent datasets spanning multiple brain regions, it outperformed 24 previously published Alzheimer’s gene signatures in its ability to tell diseased brains apart from healthy ones. It also picked up disease signals earliest in brain regions known to be affected first, confirming the AI had latched onto genuine disease biology rather than statistical noise.
From Human Data to a Living Mouse With Alzheimer’s Resilience
With a validated genetic signature in hand, researchers applied it to gene activity data from five widely used genetically engineered mouse strains. The goal was to find a mouse whose brain looked like Alzheimer’s at the molecular level but whose behavior told a different story.
A protein called Chromogranin A, or CgA, proved to be the key. CgA is found in nerve cells and support cells in the brain, its levels are elevated in the spinal fluid of Alzheimer’s patients, and it has been found tangled up with the toxic nerve fiber knots that characterize the disease. In earlier work, the researchers showed that deleting the CgA gene in mice engineered to develop a brain-tangling disease reduced damage and improved survival.
When the AI signature was applied to brain tissue from these CgA-deleted mice, the results split along sex lines. Male mice lacking CgA but carrying the disease genes showed Alzheimer’s-like molecular and pathological features, yet performed normally on tests of spatial learning and memory. Their brains had the disease markers; their minds did not. Female mice with the same genetic setup showed something more dramatic: their genetic scores clustered with healthy mice, and their brains showed additional layers of protection at every level examined.
Female Mice Showed Near-Complete Protection From Tau Damage
At the tiny junctions between nerve cells where memories are formed, healthy mice showed dense clusters of small bubble-like packages that ferry chemical signals across the gap. Diseased mice had lost most of them. Males lacking CgA recovered some of that density, but females lacking CgA came closest to matching healthy animals, retaining the kind of robust signaling machinery that memory depends on.
When researchers examined toxic Tau tangles, the sex divide was sharper still. Males lacking CgA still showed substantial tangles. Females lacking CgA were largely free of these tangles across multiple brain regions, with roughly a 23 percent reduction in one memory-center region and about a 33 percent reduction in another, compared to diseased mice.
These sex differences mirror a complicated reality in human Alzheimer’s research. Women face a higher lifetime risk of the disease and at symptomatic stages often carry a heavier Tau burden than men. But some studies suggest women may show greater early cognitive resilience, an advantage that appears to fade as disease advances. The mouse model captures this dynamic, pointing to female-specific protective mechanisms that may operate early but could eventually be overwhelmed.
Removing CgA appears to weaken the link between brain pathology and cognitive decline without fully erasing the molecular signature of the disease. For a field that has spent billions trying to clear protein clumps from Alzheimer’s brains with limited success, the idea of bolstering the brain’s natural resilience represents a fundamentally different strategy, and this AI-guided mouse model offers a new path forward.
Disclaimer: This article is based on a peer-reviewed study that has been accepted for publication but not yet fully edited. Findings reflect early-stage preclinical research conducted in mice and should not be interpreted as medical advice. The research does not establish proven treatments or interventions for Alzheimer’s disease in humans.
Paper Notes
Limitations
The researchers acknowledge several limitations. Molecular and physical analyses focused primarily on the hippocampus and prefrontal cortex; other brain regions implicated early in Alzheimer’s were not examined. The biological mechanisms driving the observed sex differences in resilience remain unresolved, and future studies will need to investigate contributions from sex hormone signaling, sex chromosome differences, and sex-specific immune responses. The team also acknowledges limitations inherent to data-driven and AI-based approaches, including dependence on input data quality and the composition of patient groups. Gene activity data alone cannot fully capture the processes underlying cognitive resilience; integration with protein-level data, metabolic measurements, electrical recordings of brain activity, and cell-type-specific analyses will be necessary for a more complete picture. The paper is described as an unedited manuscript provided for early access, and the authors note that errors may be present that affect content.
Funding and Disclosures
This work was supported by multiple National Institutes of Health grants, VA Merit Review and RR&D grants, CDMRP awards, the Wu Tsai Human Performance Alliance, the Joe and Clara Tsai Foundation, and NIDDK core services. Several authors report potential conflicts of interest: a patent (PCT/US2024/053684) was filed in 2024 by two of the authors; one author is a co-founder of RNACheck; another is the founder of CgA Therapeuticals, Inc. and co-founder of Siraj Therapeutics. The authors disclosed these interests to UC San Diego.
Publication Details
Title: AI guided discovery of a murine model of asymptomatic Alzheimer’s disease | Authors: Suborno Jati, Sahar Taheri, Satadeepa Kal, Subhash C. Sinha, Brian P. Head, Sushil K. Mahata, and Debashis Sahoo | Affiliations: University of California San Diego; Veterans Medical Research Foundation, San Diego; Helen and Robert Appel Alzheimer’s Disease Research Institute, Feil Family Brain and Mind Research Institute, Weill Cornell Medicine, New York; VA San Diego Healthcare System | Journal: Acta Neuropathologica Communications (Article in Press, 2026) | DOI: 10.1186/s40478-026-02286-y | Received: December 20, 2025 | Accepted: March 23, 2026







