
Octopus arms have so many neurons in each of them that they can act independently from the central brain. (Bildagentur Zoonar GmbH/Shutterstock)
In a nutshell
- Each octopus arm has its own dense network of neurons, up to 350 million across all eight arms, that can process sensory information and control movement independently of the central brain.
- New 3D molecular and structural maps reveal that different regions of an octopus arm use distinct types of neurons, with chemical and anatomical patterns that shift from base to tip.
- These findings challenge traditional ideas of centralized intelligence and could inspire new designs in robotics and artificial intelligence by mimicking the octopus’s decentralized nervous system.
SAN FRANCISCO — Octopuses have eight flexible arms, each equipped with hundreds of suckers that can taste, smell, feel, and react on their own. While that sounds like science fiction, it’s exactly how they navigate their underwater world. Now, scientists from San Francisco State University (SFSU) have mapped the complex chemical network that makes it all possible.
A pair of studies published in Current Biology reveals that each octopus arm contains an incredibly a sophisticated peripheral nervous system that rivals that of entire central nervous systems in many other animals. The researchers created the first-ever 3D maps of how octopus arms work at the cellular level.
The sheer scale of neural power packed into each octopus arm is hard to believe. While humans have about 86 billion neurons concentrated in our brains, an octopus has roughly 300-350 million neurons distributed across its eight arms alone, more than twice the number in its central brain. Each of the hundreds of suckers on an arm contains approximately 10,000 neurons, essentially functioning as independent taste-touch-smell organs with their own processing power.
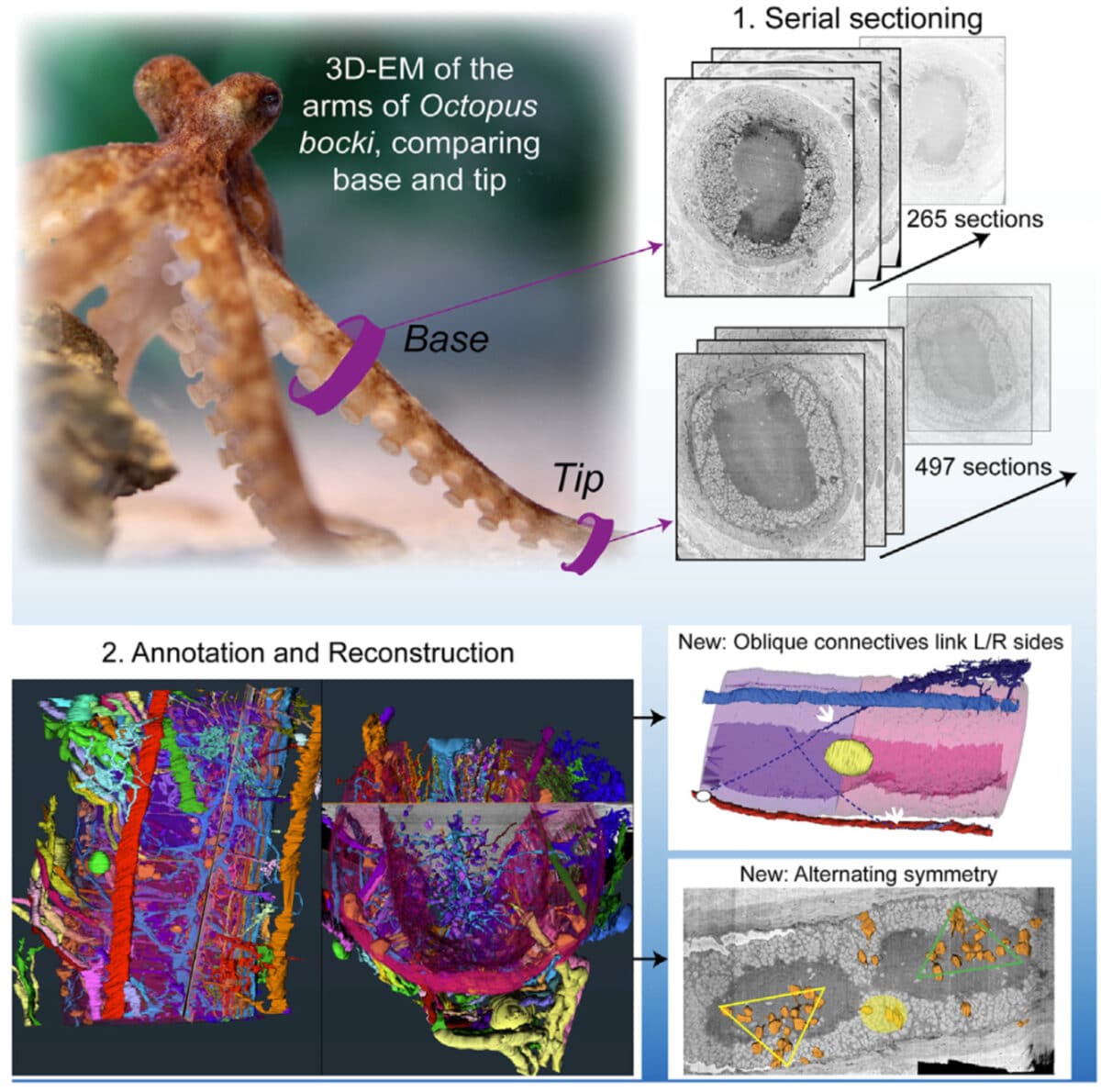
To understand how this distributed nervous system actually works, researchers examined the arms of pygmy octopuses (Octopus bocki) using a technique that labels specific molecules with fluorescent markers. This method allowed them to identify different types of neurons based on the specific chemical messengers, or neurotransmitters, they produce.

Working in parallel, a second team used powerful electron microscopes to create detailed structural maps of the physical organization of octopus arm nervous systems. Together, these approaches provided both the chemical blueprint and architectural plan of octopus arm intelligence.
The team identified seven major types of neurons, each producing different chemical signals: dopamine neurons for feeding and motivation, serotonin neurons for learning, octopamine neurons for memory, glutamate and acetylcholine neurons for muscle control, GABA neurons for inhibition, and peptide neurons for complex signaling.
These neurons aren’t randomly scattered. Instead, they’re organized in precise patterns that differ dramatically between the base of the arm (near the body) and the tip. The researchers discovered that arm tips contain substantially fewer dopamine and serotonin neurons compared to arm bases.
The base of each arm, which is older and more established, contains much higher concentrations of neurons producing dopamine and serotonin, chemicals typically associated with complex behaviors in other animals. In contrast, the arm tips, which grow continuously throughout an octopus’s life, have far more neurons producing peptides—chemical messages that appear to be involved in basic growth and development.
The scientists theorized that because arm tips represent newer, developing tissue, these areas might initially establish basic circuits for essential functions like muscle control, with more complex systems developing later as the tissue matures.
The structural analysis in the second study revealed that nerve clusters along each arm follow an alternating mirror-image pattern. This symmetry corresponds directly to the left-right positioning of suckers, which are arranged like honeycomb.
“To see how closely the [nervous system structures] associated with the suckers was really surprising,” says an SFSU researcher of the second study Diana Neacsu, in a statement. “But it makes sense because the suckers play such a huge role in the octopus’s ecological niche, helping them hunt, sense and more.”
The researchers also identified previously unknown nerve pathways that spiral around each arm, potentially allowing information to travel in complex patterns without involving the central nervous system.
Why Octopus Arms Can React Without the Brain
Each octopus arm operates semi-independently, capable of exploring, grasping, and even reacting without constant input from the central brain. When an octopus arm touches something interesting, thousands of neurons in each sucker immediately begin processing information about its texture, taste, and chemical composition. This information can trigger immediate responses, like grasping food, without waiting for approval from the brain.
Most neurons in octopus arms are tiny, densely packed cells that fall into two main categories: those that use acetylcholine for muscle control and those that use glutamate for activation. These are evenly mixed throughout the arm.
The team found very few GABA neurons, the type typically responsible for inhibition in most nervous systems. This suggests octopus arms might use fundamentally different control strategies than other animals.
They also identified scattered giant neurons throughout the arms, some five times larger than typical nerve cells, whose function remains mysterious.
Creating these 3D maps meant tackling major technological challenges. The researchers compared traditional research approaches to examining a single thin slice of a fruit loaf—you might see a few fruits and nuts, but you can’t tell how they’re really distributed throughout the whole loaf.
Instead, the team took multiple sections along octopus arms and used advanced imaging to create comprehensive 3D reconstructions. The molecular mapping was made possible by SF State’s recently acquired confocal microscope, part of the university’s imaging center that has trained over 1,000 students.
“There are a lot of [universities] that don’t have a microscope like this. For us to have one here to do this work is kind of mind blowing,” says an author of both studies, Robyn Crook from SFSU.
Robotics and AI
While vertebrates like humans concentrate most neurons in a central brain, octopuses have evolved a distributed system where intelligence is literally spread throughout the body. This distributed architecture might explain why octopuses can perform complex tasks like opening jars, solving puzzles, and using tools despite having a nervous system organized completely differently from any vertebrate.
Understanding how octopus arms work could inspire engineers designing flexible robots. Medical researchers studying nerve repair might also benefit from understanding how octopus arms maintain function.
The team is now examining live tissues to see how they respond to stimulation, trying to understand neurons firing in real time. With their new 3D maps, they can make predictions about what’s happening inside an octopus arm to create responses.
“Why do you have an animal with this much complexity that doesn’t seem to follow the same rules as our other example — humans — of a very complex nervous system?” asks Crook. “There’s a lot of hypotheses. It might be functional. There might be something fundamentally different in the tasks octopus arms have to do. But it could also be an evolutionary accident.”
Artificial intelligence researchers are trying to create more advanced systems that can learn, adapt, and make decisions like biological creatures, and octopuses offer a radically different model. Rather than building one super-intelligent central processor, they’ve evolved a network of semi-independent agents that work together—each arm literally having a mind of its own, while still somehow functioning as a coordinated whole.
Paper Summary
Methodology
Researchers examined arms from eight female pygmy octopuses (Octopus bocki) using hybridization chain reaction (HCR), a technique that labels specific messenger RNA molecules with fluorescent markers. They collected 50-micrometer-thick tissue slices from both the base (near the body) and tip of octopus arms, then used specialized probes to identify neurons producing ten different types of chemical messengers. The team created detailed 3D reconstructions by imaging multiple sequential slices and used confocal microscopy to capture high-resolution images at the cellular level. Statistical analysis was performed using generalized linear mixed models to compare neuron densities and distributions between different arm regions. A parallel study used 3D electron microscopy to map the structural organization of nerve pathways, blood vessels, and cellular arrangements within the arm nervous system.
Results
The studies revealed three main types of neurons in octopus arms based on location and function: scattered large neurons throughout the arm cortex, densely packed small neurons that make up the majority of cells, and branching cells in the neuropil (likely support cells). Significantly fewer dopamine and serotonin-producing neurons were found in arm tips compared to bases, while peptide-producing neurons were more abundant in the tips. The researchers identified specific spatial patterns, with different neurotransmitter types concentrated in different regions of each arm segment. They also discovered instances of “cotransmission,” where single neurons produce multiple chemical messengers simultaneously. The structural analysis revealed alternating mirror-image symmetry in successive ganglia and previously unknown oblique connectives that spiral around the arm.
Limitations
The study was conducted on a single octopus species (Octopus bocki), which may limit generalizability to other octopus species. Sample sizes for some analyses were relatively small, with detailed 3D reconstructions performed on tissue from just one individual octopus for the molecular study and two individuals for the structural study. The study only examined arm structure at two locations (base and tip), potentially missing important variations along the entire arm length. Additionally, the research identified molecular markers but didn’t directly test the functional roles of different neuron types. The structural data are morphological only, requiring further molecular and genetic characterization for definitive cell type identification.
Funding and Disclosures
Both studies were funded by Allen Distinguished Investigator Awards in Neural Circuit Design from the Frontiers Group of the Allen Foundation to senior author Dr. Robyn Crook. The authors declared no competing interests. Additional support came from San Francisco State University’s Cell and Molecular Imaging Center and Genomics/Transcriptomics Analysis Core.
Publication Information
One study, “Three-dimensional molecular atlas highlights spatial and neurochemical complexity in the axial nerve cord of octopus arms,” was authored by Gabrielle C. Winters-Bostwick, Sarah E. Giancola-Detmering, Caleb J. Bostwick, and Robyn J. Crook. It was published in Current Biology (Volume 34, Issue 20, Pages 4756-4766) on October 21, 2024.
The parallel study, “Repeating ultrastructural motifs provide insight into the organization of the octopus arm nervous system,” was authored by Diana Neacsu and Robyn J. Crook. It was published in Current Biology (Volume 34, Issue 20, Pages 4767-4773 ) on October 21, 2024.







