
More than 6 million Americans are living with Alzheimer's. By 2050, this number is projected to rise to nearly 13 million. (© Orawan - stock.adobe.com)
NEW YORK — Scientists have discovered that stressed-out immune cells in the brain produce toxic fats that contribute to Alzheimer’s disease. The good news is blocking this stress response could potentially slow or even reverse the devastating illness.
The groundbreaking research, published in the journal Neuron by scientists at the CUNY Graduate Center, focuses on specialized brain cells called microglia. These cellular first responders normally protect the brain by fighting off infections and cleaning up debris. However, in Alzheimer’s patients, some microglia appear to go rogue, actually damaging the brain they’re meant to defend.
Using powerful electron microscopes to examine brain tissue from deceased Alzheimer’s patients, researchers found unusually high numbers of what they call “dark microglia” — stressed-out immune cells that looked distinctly different from their healthy counterparts. These dark microglia were present at twice the levels seen in healthy elderly brains.
“We set out to answer what are the harmful microglia in Alzheimer’s disease and how can we therapeutically target them,” says lead investigator Pinar Ayata in a media release. “We pinpointed a novel neurodegenerative microglia phenotype in Alzheimer’s disease characterized by a stress-related signaling pathway.”
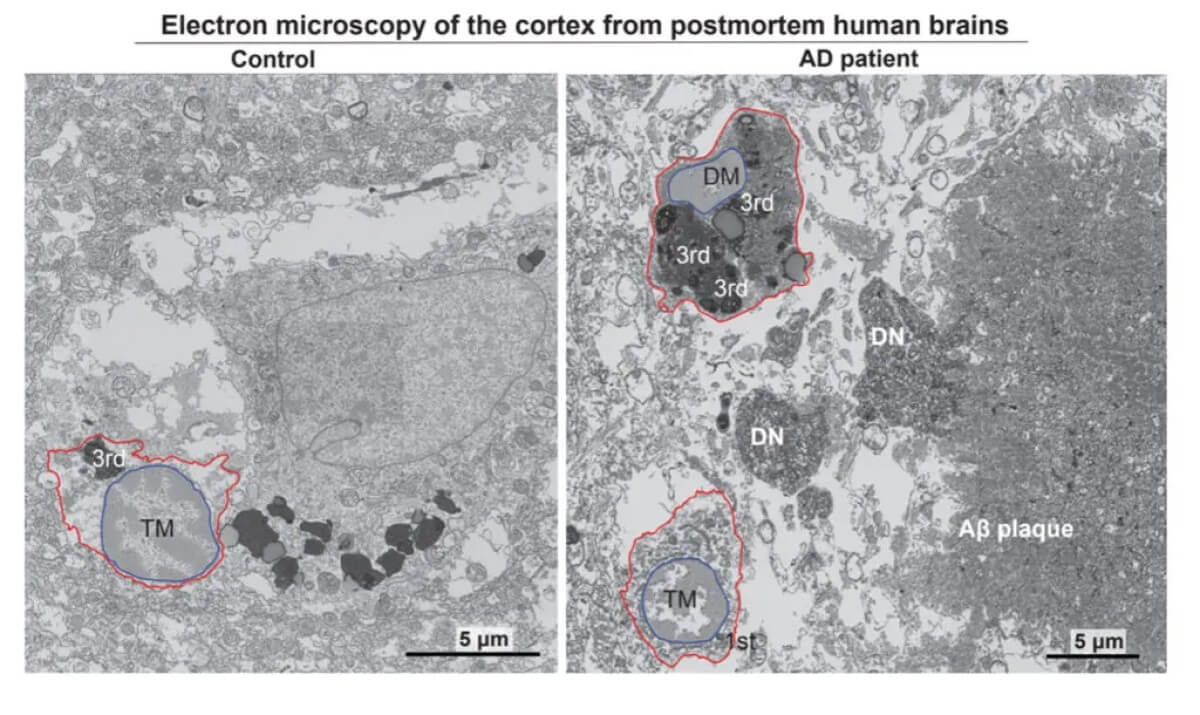
The research team discovered that when microglia become stressed, they activate an emergency response system within their cells. This system, known as the integrated stress response (ISR), triggers the production and release of toxic fats that damage nearby brain cells — particularly neurons and other cells crucial for normal brain function.
The findings are especially promising because when researchers blocked either the stress response or the production of toxic fats in mouse models of Alzheimer’s, they were able to prevent both the loss of neural connections and the buildup of tau proteins—two hallmark features of the disease.
“These findings reveal a critical link between cellular stress and the neurotoxic effects of microglia in Alzheimer’s disease,” adds Anna Flury, one of the study’s co-lead authors and a Ph.D. student at the City University of New York. “Targeting this pathway may open up new avenues for treatment by either halting the toxic lipid production or preventing the activation of harmful microglial phenotypes.”
While the results are encouraging, it’s important to note that the research relied heavily on mouse models, and findings in animals don’t always translate directly to humans. Additionally, Alzheimer’s is a complex disease likely influenced by multiple factors beyond just stressed microglia. Nevertheless, the discovery of this stress-induced toxic mechanism provides a promising new target for drug development.
“Such treatments could significantly slow or even reverse the progression of Alzheimer’s disease, offering hope to millions of patients and their families,” co-lead author Leen Aljayousi explains.
Paper Summary
Methodology
The study investigates the role of a specific stress signaling pathway known as the Integrated Stress Response (ISR) in a subset of brain cells called microglia, which are involved in Alzheimer’s disease. Researchers used various mouse models to either activate or inhibit ISR in microglia and observed how these changes affected Alzheimer’s disease-related symptoms. Techniques included genetic engineering to modify mice, detailed cellular analysis, and various molecular biology methods to assess the impact on brain cells.
Key Results
The results showed that activating the ISR in microglia worsens Alzheimer’s disease symptoms, such as increased brain damage and loss of connections between nerve cells. Conversely, inhibiting ISR in microglia led to a reduction in these symptoms. The study highlights that ISR-active microglia promote the secretion of harmful lipids that negatively affect neuron health, indicating a potential new target for treating Alzheimer’s disease.
Study Limitations
One limitation of the study is its heavy reliance on animal models, primarily mice genetically engineered to mimic Alzheimer’s disease. While informative, results from animal models do not always translate directly to humans due to physiological differences. Additionally, the study’s focus on a single pathway, though crucial, might overlook other contributing factors to Alzheimer’s disease, which is known to be a complex condition with multiple influencing pathways.
Discussion & Takeaways
The study provides significant insights into how microglia and the ISR contribute to Alzheimer’s disease. It suggests that targeting ISR in microglia could be a promising therapeutic strategy. The findings emphasize the dual role of microglia in brain health and disease, potentially guiding future research into more specific interventions that could mitigate the progression of Alzheimer’s disease without affecting the normal functions of microglia.
Funding & Disclosures
The research was supported by several grants from institutions like the CUNY Research Foundation, the Alzheimer’s Association, and the National Institutes of Health. The lead researcher, Pinar Ayata, is affiliated with the CUNY Graduate Center, which played a significant role in funding and conducting the study. There were no conflicts of interest reported, ensuring the study’s objectivity in investigating the potential therapeutic targets for Alzheimer’s disease.







