
More than 6 million Americans are living with Alzheimer's. By 2050, this number is projected to rise to nearly 13 million. (© Orawan - stock.adobe.com)
NEW YORK — In a breakthrough that could eventually lead to new treatments for devastating conditions like Alzheimer’s disease, researchers have finally solved a puzzle that has stumped scientists for five decades: how a crucial fat molecule in the brain manages to survive while helping break down other fats.
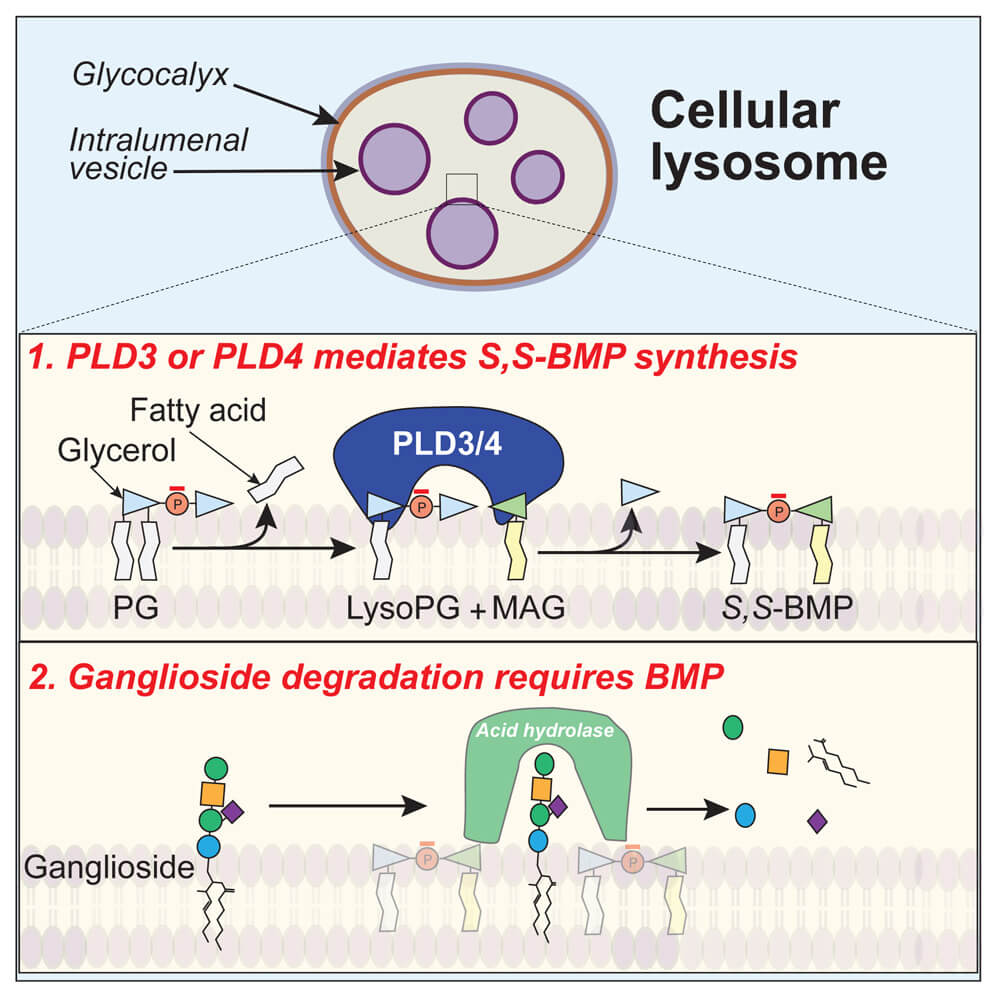
The molecule in question, known as BMP, works inside the brain’s cellular “garbage bins” called lysosomes. What makes BMP special is its unusual molecular structure – it’s essentially a mirror image of most other fats in the body. This unique characteristic allows it to survive in an environment where other fats are routinely destroyed, making it an essential player in maintaining brain health.
“BMP is a co-factor for degradation, but itself is very, very stable, and it has an unusual chemistry,” explains Howard Hughes Medical Institute Investigator Tobias Walther in a media release. “As a consequence, nobody had known how this is made.”
The discovery’s significance is particularly relevant for frontotemporal dementia (FTD), the condition that actor Bruce Willis was diagnosed with in 2023. FTD, which affects personality, judgment, and speech, is the leading cause of dementia in people under 60 and currently has no cure or treatment.
The research team, led by Walther and Robert Farese, Jr. at the Sloan Kettering Institute, found that two enzymes – PLD3 and PLD4 – are responsible for creating BMP. This discovery came after years of careful investigation, including experiments in laboratory tests, human cells, and animal models.
To understand why this matters, imagine BMP as a specially trained cleanup crew that helps dispose of potentially harmful fats called gangliosides in the brain. When BMP levels are low, as they discovered in FTD patients’ brains, these toxic gangliosides start accumulating, potentially contributing to the disease’s progression.
What makes this discovery particularly fascinating is how it solves the mystery of BMP’s unusual structure.
“Molecules have a pattern that is either like a left or right hand that is identical at one level, but one is a mirror image of the other,” Walther explains.
While most fats in our body are “right-handed,” BMP is one of the rare “left-handed” molecules.
The research team found that cells perform a remarkable swap meet at the molecular level to create this mirror image. Led by postdoctoral fellow Shubham Singh, they discovered that cells exchange parts between different molecules to create the unique form of BMP – a process called transphosphatidylation.
The findings, published in the journal Cell, have already revealed interesting connections to various brain disorders. Mutations in PLD3, one of the enzymes responsible for making BMP, are linked to both a rare neurodegenerative disease called spinocerebellar ataxia 46 and increased Alzheimer’s risk. When researchers disabled PLD3 in mice, they observed significant changes in brain lipids.
“The paper’s findings that these two related enzymes, PLD3 and PLD4, produce BMP fills in a significant piece in the BMP puzzle, and these enzymes do so in an elegant way that results in inversion of the stereochemistry, or handedness, of parts of the molecule,” says Jeremy Baskin, a cell biologist at Cornell University who did not take part in the study.
While it’s too early to talk about specific treatments, this fundamental understanding of how BMP is created could eventually lead to new therapeutic approaches for various neurodegenerative diseases.
“It really took us sitting down and drawing out the pathways with persistence and a little bit of serendipity to go after this. There are so many of these unturned stones and fundamental discoveries left to be made,” Walther concludes, highlighting the importance of basic scientific research.
Paper Summary
Methodology
The researchers were interested in how specific enzymes in the body, PLD3 and PLD4, play a role in breaking down certain fats (lipids) in the brain. To study this, they used human and mouse cells that either had or didn’t have the PLD3 and PLD4 enzymes.
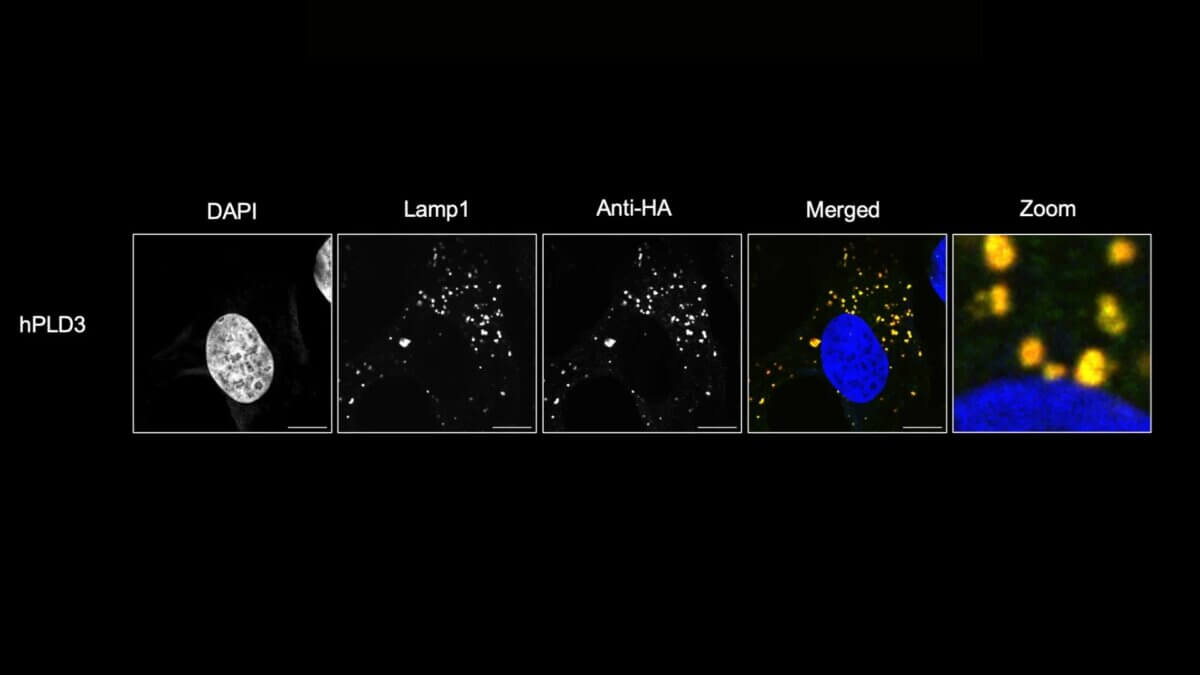
They also studied cells that had been modified to include mutations in PLD3, which are linked to neurological diseases. The researchers performed tests where they added specific fats to the cells and tracked how the cells responded by measuring the amount of a particular fat (S,S-BMP) that is necessary for breaking down other fats. They also used microscopes and advanced equipment to track how well the cells’ waste-processing system (lysosomes) was functioning.
Key Results
The researchers found that the enzymes PLD3 and PLD4 are essential for creating a special fat (S,S-BMP) that helps break down other fats inside the brain’s cells. Without these enzymes, the cells couldn’t properly manage the breakdown of fats, leading to an unhealthy buildup.
In mice missing these enzymes, especially in the brain, there was a buildup of fats that are known to cause problems in the brain, like gangliosides, which are linked to neurodegenerative diseases such as Alzheimer’s. The study shows that when these enzymes don’t work properly, it leads to big problems in how the brain handles fats.
Study Limitations
First, while the results were clear in lab conditions and mice models, we don’t know how these findings will fully apply to humans, especially in more complex real-world environments. Additionally, the study focuses heavily on specific types of fat and their breakdown, but there may be other factors or processes in the body that also contribute to these issues, which weren’t explored in this research.
Discussion & Takeaways
The big takeaway from this study is that the enzymes PLD3 and PLD4 are crucial for brain health because they help manage fat breakdown. When these enzymes don’t function correctly, fats build up in the brain, which may lead to neurodegenerative diseases like Alzheimer’s.
This research opens the door to potentially using therapies that target these enzymes or the fats they help produce to prevent or treat brain diseases. Future research will likely focus on figuring out exactly how to apply these findings in humans and whether boosting or fixing these enzymes could be used to fight brain diseases.
Funding & Disclosures
This work was supported by a grant from the Bluefield Project to Cure FTD, a Human Frontiers Science Program fellowship grant, and postdoctoral fellowship grants from the Bluefield Project to Cure FTD. The researchers involved have disclosed no conflicts of interest that would affect the integrity of the study.







