
(© Feng Yu - stock.adobe.com)
In a Nutshell
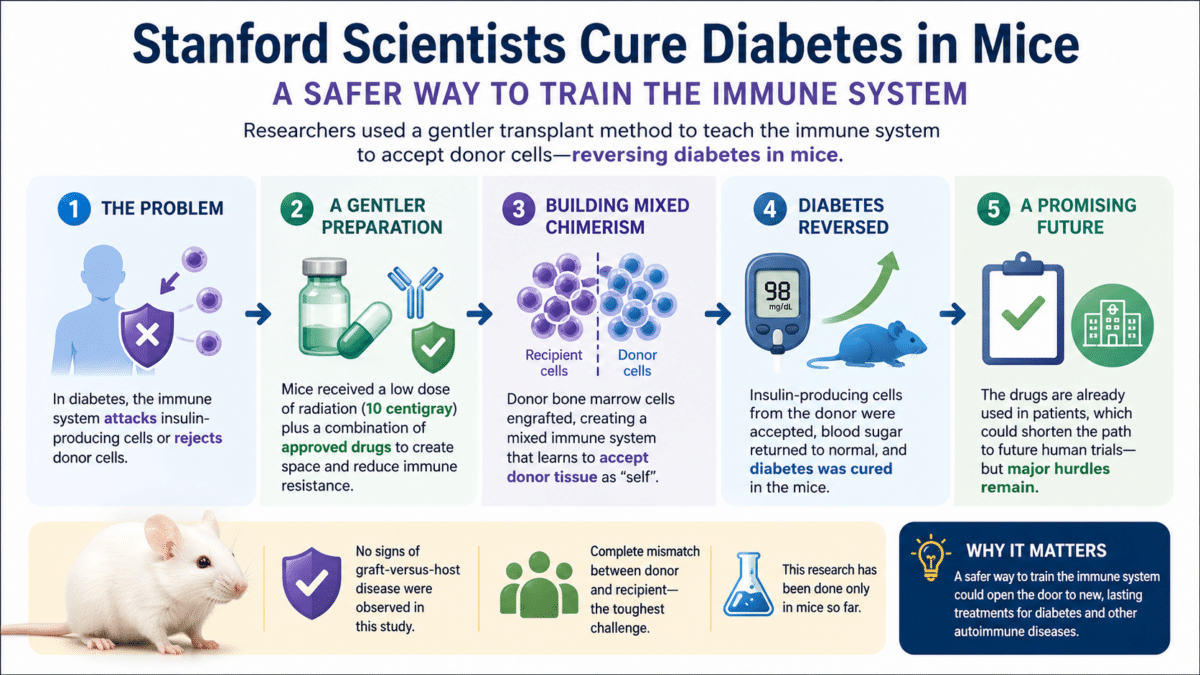
- Stanford researchers cured diabetes in mice by training the immune system to accept donor insulin-producing cells, without the toxic high-dose radiation or chemotherapy typically required before a transplant.
- The key was achieving “mixed chimerism,” a state where the recipient’s immune system learns to treat donor tissue as its own, using a combination of drugs already approved for other conditions plus an ultra-low radiation dose of just 10 centigray.
- Mice treated with this approach showed no signs of graft-versus-host disease, one of the most dangerous complications of bone marrow transplantation, suggesting the method may be safer than traditional conditioning.
For the millions of Americans living with diabetes, the dream of a lasting cure has always felt close yet frustratingly out of reach. Transplanting insulin-producing cells from a donor can, in theory, replace what the disease destroys. But the body’s immune system attacks those foreign cells, and the treatments used to prevent that rejection can be nearly as dangerous as the disease itself. Now, a team at Stanford has found a way to cure diabetes in mice by dramatically reducing the toxic preparation the body needs before a transplant, while still convincing the immune system to accept donor tissue as its own.
The study, published in JCI Insight on April 21, 2026, takes on one of the most stubborn problems in transplant medicine. When doctors transplant cells or organs from one person to another, the recipient’s immune system recognizes the new tissue as foreign and launches an attack. One powerful strategy to prevent this is to first transplant bone marrow cells from the same donor, creating a state where the recipient’s blood and immune system becomes a mix of their own cells and the donor’s, a condition called “mixed chimerism.” In that scenario, the immune system essentially learns to treat the donor’s tissue as “self,” a state known as tolerance. It’s an elegant solution, but it requires harsh preparation of the patient’s bone marrow beforehand, often involving high doses of radiation or chemotherapy, that carries serious risks of its own.
The Stanford team, led by senior author Seung K. Kim from the Department of Developmental Biology at Stanford School of Medicine, wanted to know whether they could achieve that same tolerance using a gentler recipe. Their answer, demonstrated in a mouse model of diabetes, appears to be yes. It is important to emphasize that this work has been done only in mice so far, and significant hurdles remain before it could be tested in people.
A Gentler Recipe for Curing Diabetes in Mice
To understand what sets this work apart, it helps to know what normally happens before a bone marrow transplant. The patient’s existing bone marrow, the factory where blood and immune cells are made, needs to be partially or fully cleared out to make room for the donor’s cells. Traditionally, this “conditioning,” the medical preparation that readies the body to receive new bone marrow, involves large doses of full-body radiation or powerful drugs that wipe out immune cells. These treatments work, but they also cause damage to organs and raise the risk of infections, among other serious side effects. That toxicity is a major reason why bone marrow transplants aren’t more widely used as a strategy to train the immune system to accept donated tissue.
The Stanford researchers tested a combination of agents, many of which are already in clinical use for other conditions. Their cocktail included baricitinib, a drug that blocks certain immune signaling pathways; venetoclax, a drug that targets a protein helping certain immune cells survive; and an antibody against CD47, a molecule sometimes called a “don’t eat me” signal on cells. They combined these with an antibody against CD117, a protein found on blood stem cells, along with temporary removal of immune cells called T cells.
The real breakthrough was in how little radiation they needed. The team used just 10 centigray of full-body radiation, an extraordinarily low dose. Traditional conditioning for bone marrow transplants can use doses many times higher. By pairing this minimal radiation with their drug combination, the researchers achieved lasting mixed chimerism across a complete mismatch in the molecular markers that the immune system uses to tell “self” from “foreign.” A complete mismatch in these markers represents the toughest immunological barrier, comparable to transplanting between two entirely unrelated individuals.
Curing Diabetes in Mice Without Dangerous Immune Reactions
Once the researchers established mixed chimerism in their diabetic mice, they transplanted insulin-producing cells from donor mice that matched the bone marrow donor. The transplanted cells were accepted by the recipient’s immune system, and the mice’s diabetes was reversed. Blood sugar levels returned to normal, effectively curing the disease in these animals.
Just as important was what didn’t happen. One of the most feared complications of bone marrow transplantation is graft-versus-host disease, a condition where the donor’s immune cells turn against the recipient’s own body, attacking organs like the skin, liver, and gut. In this study, the researchers did not observe signs of that reaction in the treated mice. Their gentler conditioning approach worked, and it worked safely, at least in this animal model.
The transplanted insulin-producing cells came from donors completely mismatched in their immune markers, which matters because it shows tolerance can be achieved even under the most difficult conditions. The team carefully tracked the proportion of donor versus recipient blood cells to confirm that the mixed state lasted over time, rather than fading quickly.
Why Existing Drugs Make This Approach Especially Promising
One of the most practically significant aspects of this research is that the agents used are not experimental compounds sitting in a laboratory freezer. Baricitinib is prescribed for rheumatoid arthritis and other inflammatory conditions. Venetoclax is used in certain blood cancers. The anti-CD47 antibody has been explored in cancer treatment. The antibody against CD117 has also been studied in clinical settings for conditioning before bone marrow transplants.
This means the path from laboratory discovery to potential human trials could be shorter than it would be for entirely new drugs. Of course, “could be” carries a lot of weight in that sentence. Mouse studies don’t always translate to humans, and there are good reasons for caution. The specific mouse strains, diabetes model, and transplant conditions used in this study may not fully replicate the complexity of the human immune system or human diabetes.
While the individual drugs have established safety records, their combined use in this specific conditioning regimen has never been tested in people, and unforeseen interactions or side effects could emerge. Significant safety and effectiveness testing in larger animals and eventually in clinical trials would be needed before this approach could be tried in people with diabetes, a process that typically takes many years.
Beyond diabetes, this work speaks to the broader field of transplant medicine. The central problem in any transplant, whether it’s a kidney, a liver, a heart, or insulin-producing cells, is convincing the recipient’s immune system not to destroy the donated tissue. Currently, most transplant recipients must take immune-suppressing drugs for the rest of their lives, medications that dampen the entire immune system and leave patients vulnerable to infections and cancers.
Mixed chimerism offers a different path: rather than suppressing the whole immune system indefinitely, it retrains the system to specifically accept the donor’s tissue. If conditioning for chimerism could be made safe enough, it could change how transplants are done, not just for diabetes but for a wide range of conditions. The Stanford team’s work is a step toward making that conditioning safe enough to be practical.
Diabetes affects roughly 37 million Americans, and for those whose disease involves the immune system destroying their own insulin-producing cells, the promise of a transplant-based cure has been a decades-long pursuit. This study doesn’t deliver that cure to the clinic. Not yet. But it offers a clearer, less toxic road map for how the field might get there, while underscoring how much work remains to turn promising results in mice into treatments for people.
Paper Notes
Limitations
This study was conducted entirely in mice, and results from animal models do not always translate directly to human patients. The specific mouse strains, diabetes model, and transplant conditions used may not fully replicate the reality of human diabetes or the human immune system. The paper is presented as an in-press preview, meaning the full peer-reviewed details of sample sizes, long-term follow-up duration, and safety data may not yet be fully available in the published version reviewed here. Additionally, while the individual drugs tested are in clinical use, their combined use in this specific conditioning regimen has not been tested in humans, and unforeseen interactions or side effects could emerge in clinical translation.
Funding and Disclosures
No specific funding sources or conflict-of-interest disclosures were detailed in the content available for this article.
Publication Details
Title: Improved conditioning for hematopoietic chimerism induces islet tolerance to cure diabetes
Authors: Stephan A. Ramos, Preksha Bhagchandani, Diego M. Burgos, Xueying Gu, Richard Rodriguez, Nadia Nourin, Martin Neukam, Shiva Pathak, Judith Shizuru, and Seung K. Kim
Affiliations: Department of Developmental Biology and Division of Blood and Marrow Transplantation, Stanford School of Medicine, Stanford, United States of America
Journal: JCI Insight | DOI: 10.1172/jci.insight.194491 | Published: April 21, 2026







