
(Photo by Yan Krukov from Pexels)
In A Nutshell
- Your sense of cold comes from a single protein, TRPM8, which opens in response to cool temperatures and menthol.
- New research shows this protein doesn’t just “switch on” but actually shifts shape, moving between different structural states.
- A small change in one amino acid helps explain why some animals are less sensitive to cold than others.
- The findings suggest cold sensing is a dynamic, multi-step process involving protein movement and interactions with membrane lipids.
A blast of winter air, an ice cube pressed against skin, the sharp coolness of a peppermint. These everyday sensations all trace back to a single molecular sensor embedded in nerve endings throughout the body. For the first time, researchers have captured structural snapshots of that sensor as it responds to dropping temperatures, revealing a surprisingly dramatic transformation that challenges assumptions about how cold detection works.
The protein is called TRPM8, a channel that sits in the wall of sensory nerve cells and acts as the body’s built-in thermometer for cool and cold temperatures. When the temperature dips below roughly 78°F, TRPM8 opens, allowing charged particles to flow into the cell and trigger electrical signals that the brain interprets as “cold.” It’s also the same channel that menthol, the compound in mint, activates to create that familiar cooling sensation. Despite decades of research, scientists had not fully explained how temperature changes cause the channel to open.
A study published in Nature offers a new mechanistic model. Researchers combined cryogenic imaging with measurements of protein flexibility across temperatures to build a detailed picture of cold sensing. They found that TRPM8 doesn’t simply open. It can rearrange its architecture, shifting between configurations in a way not seen in previous TRPM8 structures.
How TRPM8 Responds to Cold: A Shape Nobody Expected
Channels like TRPM8 are made of four protein units arranged in a ring to form a pore through the membrane. In previously known structures, these units interlocked in a “swapped” arrangement, where parts of one unit crossed over and interacted with a neighbor.
But when researchers examined TRPM8 in a more native membrane environment, they observed a different configuration: the “semi-swapped” form. In this state, part of the protein bends by about 52 degrees and remains associated with its own unit rather than crossing over. The outer pore also shifts position.
The channel can transition between these forms without falling apart, and menthol shifts the balance toward the semi-swapped configuration. This suggests TRPM8 is more structurally dynamic than previously appreciated.
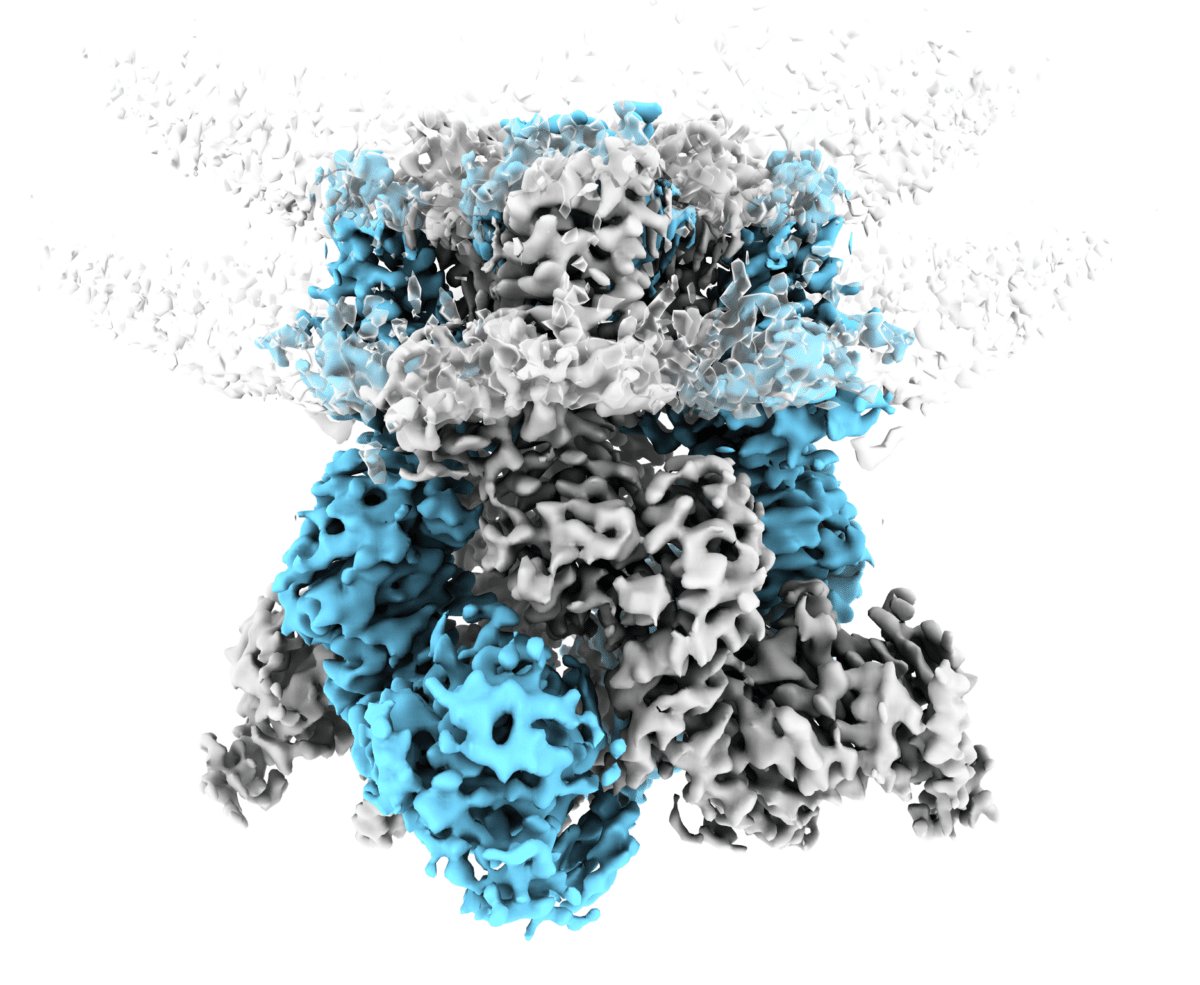
Tracking Energy Changes Across the Cold-Sensing Protein
To understand why cold activates the channel, researchers measured how different parts of the protein exchange hydrogen atoms with surrounding water: a proxy for stability and flexibility.
One region stood out: the outer pore. In human TRPM8, this region becomes more stable as temperatures drop below the activation threshold of about 78°F. In the bird version of the channel, from the great tit, this stabilization is much weaker, consistent with reduced cold sensitivity.
The data also revealed two coexisting populations of protein states, corresponding to the swapped and semi-swapped structures seen in imaging. Menthol shifted the balance between these populations in the same direction, reinforcing the link between structure and function.
One Amino Acid Helps Explain Species Differences
A key difference between bird and mammalian TRPM8 lies in a single amino acid in the outer pore.
Birds have a bulky tyrosine at this position, while mammals have a smaller valine. In the bird channel, this tyrosine fits into a pocket that helps stabilize a closed state, making the channel less responsive to cold.
Swapping these residues changed sensitivity. Replacing tyrosine with valine increased cold responsiveness, while introducing tyrosine into the human channel reduced it. This suggests the site is a key contributor to differences in cold sensitivity between species, though it does not fully account for organism-level responses.
Changes in pH also affected this interaction in the bird channel, likely by altering nearby charged residues, partially relieving the stabilizing effect.
Capturing the Channel Opening
Under conditions that weakened the stabilizing interactions in the bird channel, researchers identified a structure consistent with a cold-activated open state. A portion of the protein shifts upward, and a key residue rotates to widen the pore.
Within this structure, the researchers observed density interpreted as a calcium ion positioned within the gate, coordinated by surrounding parts of the protein.
The open state appears to be stabilized by a membrane lipid called PIP2. Its tail inserts into a groove that becomes accessible when the outer pore rearranges, helping hold the channel open. While PIP2 was already known to be important for TRPM8 function, this provides direct structural insight into how it interacts with the channel.
The same general open configuration was observed in the human channel under cold conditions and with menthol, suggesting that different stimuli converge on a shared final state.
Pulling these findings together, the researchers propose that cooling stabilizes the outer pore region, which may reposition parts of the protein and create a binding site for PIP2. Lipid binding then helps stabilize the open state.
A Broader Lesson from “Messy” Data
The study also highlights an important methodological point. Many particles in the imaging data appeared blurry and are typically discarded. The researchers suggest these may represent intermediate states as the channel transitions between configurations.
Focusing only on the sharpest structures, they argue, may overlook the dynamic processes that are central to protein function.
What began as a question about why cold air feels sharp on the skin now points to a more complex answer: a protein that shifts between structural states, interacts with membrane lipids, and reflects evolutionary differences at the molecular level. This offers a clearer, though still incomplete, picture of how we sense cold.
Paper Notes
Limitations
The study relies on cryogenic electron microscopy, which requires imaging proteins at extremely low temperatures: an inherent challenge when studying a cold-activated channel. To address this, the researchers complemented structural data with temperature-dependent measurements of protein flexibility.
A substantial fraction of particles in the imaging datasets could not be cleanly classified into distinct structural states, reflecting significant conformational variability. Rather than being purely noise, these may represent intermediate states between configurations.
Flexibility measurements were performed on detergent-extracted protein rather than membrane-embedded protein, although control experiments suggested similar behavior when menthol was applied before or after purification.
The cold-activated open state of the bird channel was captured under high-pH conditions, which may not fully reflect physiological environments. However, supporting evidence for a similar open state was observed in modified channels under neutral conditions.
Funding and Disclosures
No specific funding sources or conflict-of-interest disclosures were identified in the provided materials.
Publication Details
Title: Structural energetics of cold sensitivity| Journal: Nature| DOI: https://www.nature.com/articles/s41586-026-10276-2 | Species studied: Parus major (great tit) and Homo sapiens TRPM8 channels, expressed in HEK293 cells | Key techniques: Cryo-EM of membrane vesicles and detergent-purified protein; hydrogen-deuterium exchange mass spectrometry (HDX-MS); calcium imaging; mutagenesis and chimeric constructs







