3D illustration of a brain tumor. (Credit: Kateryna Kon on Shutterstock)
One Brain Cancer Patient Is Alive Four Years After a Custom DNA Vaccine.
In A Nutshell
- Researchers built a personalized DNA vaccine from each patient’s own tumor and tested it in nine people with a hard-to-treat form of brain cancer that doesn’t respond to standard chemotherapy.
- One-third of patients survived past two years, well above the 10 to 15 percent typically seen in this group, though the trial was too small to prove the vaccine caused that outcome.
- One patient remains alive and recurrence-free more than four years after surgery, with new immune cell activity in both the bloodstream and tumor tissue detected after vaccination.
- A common brain-swelling steroid called dexamethasone appeared to blunt the vaccine’s immune response, a potential obstacle researchers say must be addressed in future trials.
For most people diagnosed with the deadliest form of brain cancer, surviving two years is considered a long shot. Glioblastoma, a fast-moving, treatment-resistant brain tumor, kills most patients within 12 to 18 months, even with surgery, radiation, and chemotherapy. A small clinical trial has now produced a result researchers are calling a meaningful step forward: a personalized DNA vaccine, built from each patient’s own tumor, was associated with longer-than-expected survival in some patients, including one who remains alive and recurrence-free more than four years after surgery.
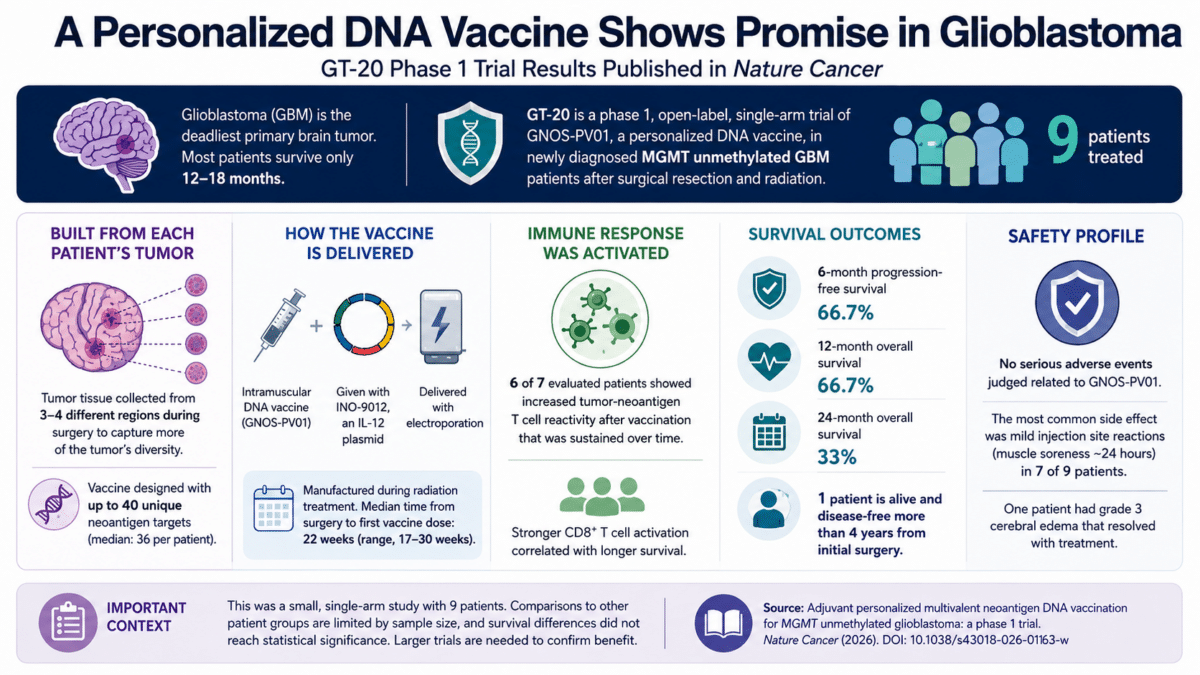
Nine patients enrolled in the trial, known as GT-20. All carried a specific genetic characteristic, called MGMT unmethylated, which means they don’t benefit from the standard chemotherapy drug used in treatment. Researchers designed the experimental vaccine, called GNOS-PV01, for each individual patient using up to 40 unique targets pulled from their own tumor tissue. It was injected into the muscle and activated with a mild electrical pulse, alongside a second DNA molecule designed to boost the immune response.
Results published in the journal Nature Cancer showed no serious adverse events judged related to the vaccine, though one patient had a brief episode of brain swelling, serious enough to require treatment, that resolved completely. Blood tests showed signs the vaccine was activating patients’ immune systems. One-third of the patients survived past two years, compared with the 10 to 15 percent researchers cited from earlier studies in the same patient population. Because the trial included only nine people and had no randomized control group, that finding is promising but far from proof.
Building a Personalized Glioblastoma Vaccine From Scratch
What sets GNOS-PV01 apart is how completely personalized it is. Glioblastoma tumors are notoriously inconsistent, with different parts of the same tumor carrying different genetic mutations, making it very difficult for the immune system to recognize and attack the cancer. Researchers collected tissue from three or four separate locations within each patient’s tumor during surgery, giving scientists a broader map of the tumor’s genetic makeup.
From those samples, they identified targets called neoantigens, protein fragments that appear on the surface of cancer cells but not healthy ones, essentially marking the tumor as foreign to the immune system. Because these markers are unique to each patient, the vaccine had to be custom-built for every person in the study. Manufacturing happened during the six weeks patients were already undergoing radiation therapy.
Sampling from multiple regions increased the number of identified mutations by 45 percent on average. A median of 36 targets were included per patient, and for seven out of nine, researchers included every candidate without cutting any.
Glioblastoma Vaccine Linked to Immune Response and Longer Survival
In six out of seven evaluated patients, blood tests showed a sustained rise in immune cell reactivity against tumor targets, beginning as early as the first measurement point after vaccination.
CD8 T cells, immune cells that can help kill abnormal cells, showed statistically significant increases in activation markers after exposure to the vaccine targets. When researchers examined whether the strength of that response correlated with survival, a pattern emerged: stronger CD8 T-cell activation was linked with longer survival in this small group. A similar pattern was not found for a related immune cell type.
One patient who did not generate a boosted immune response was receiving dexamethasone, a steroid commonly used to reduce brain swelling and known to suppress immune activity. Data from the trial suggest it may work against vaccine-based treatments, a conflict researchers say will need to be addressed in future studies.
In two patients who required a second surgery after vaccination, researchers examined the tumor tissue itself. One of those patients, not on steroids, showed a significant increase in immune cells infiltrating the tumor, along with new T-cell clones appearing in both the bloodstream and the tumor. Researchers also found a notable difference in a protein called B2M, which helps the immune system recognize tumor targets: one patient showed increased B2M expression after vaccination, while the other, on dexamethasone throughout, showed reduced B2M expression, making the tumor harder to detect.
What the Long-Term Survivor’s Data Revealed
Patient 8 is the most striking case in the trial: alive and recurrence-free more than four years after surgery, with no additional treatment after a diagnostic biopsy confirmed no active disease. Researchers found that new immune cell clones, apparently generated in response to the vaccine, had appeared in both the bloodstream and the tumor itself. Whether those immune changes are what kept the disease at bay is an open question, but researchers note the pattern looked markedly different from patients who fared worse.
Glioblastoma Vaccine Safety Holds Up as Trial Hits Its Goals
No patients stopped vaccination because of side effects. Mild injection site reactions were the most common complaint, and the brain swelling episode resolved fully. Including up to 40 targets rather than the 20 used in earlier trials did not make the vaccine less safe.
Logistical challenges remain. Getting each patient vaccinated within four weeks of completing radiation proved difficult, with the actual median closer to ten weeks. Researchers say speeding up manufacturing will be a priority.
With nine patients and no control arm, the trial was never designed to prove the vaccine works. But it met its pre-set goals, the immune signals were real, and one patient still alive four years later is the kind of data point this disease rarely produces.
Disclaimer: This article is based on a published peer-reviewed study. Individual results may vary. Cancer treatments discussed are experimental and not approved for general use. Consult a qualified medical professional before making any health-related decisions.
Paper Notes
Limitations
Authors explicitly acknowledge several limitations. Nine patients is a very small sample that prevents definitive conclusions about survival benefit or meaningful statistical comparisons with control groups. Contemporaneous comparison cohorts were also small, and survival differences between groups did not reach statistical significance. Several immunological analyses were restricted to subsets of patients due to insufficient samples. The feasibility endpoint was not fully met within the originally intended timeframe, and the authors note pipeline optimization will be required for future trials. Three patients had disease progression before receiving their first vaccine dose, complicating interpretation of outcomes for those individuals. The study also lacked a randomized control arm, meaning causality between the vaccine and survival outcomes cannot be established.
Funding and Disclosures
Several authors are current or former full-time employees of Geneos Therapeutics, the company that supplied the vaccine, adjuvant plasmid doses, and electroporation devices and provided research funding for the clinical trial. One author holds a consultant role for Geneos Therapeutics. Two authors hold consultant roles for the Jaime Leandro Foundation for Therapeutic Cancer Vaccines and Pathfinder Oncology. One additional author holds a consultant role for the Jamie Leandro Foundation. Additional funding came from the National Institutes of Health, the Mark Foundation for Cancer Research, the Nationwide Foundation Pediatric Innovation Fund, the Alvin J. Siteman Cancer Center Investment Program, and the Foundation for Barnes-Jewish Hospital. Readers should consult the full published article for a complete list of author affiliations and conflict-of-interest disclosures.
Publication Details
This article is based on the following peer-reviewed publication: Adjuvant personalized multivalent neoantigen DNA vaccination for MGMT unmethylated glioblastoma: a phase 1 trial, published in Nature Cancer. DOI: https://doi.org/10.1038/s43018-026-01163-w. Published online May 12, 2026. Manuscript received August 19, 2025; accepted April 7, 2026. Corresponding author: Tanner M. Johanns ([email protected]). Clinical trial registration: ClinicalTrials.gov identifier NCT04015700. A full list of authors and their institutional affiliations appears at the end of the published paper.







