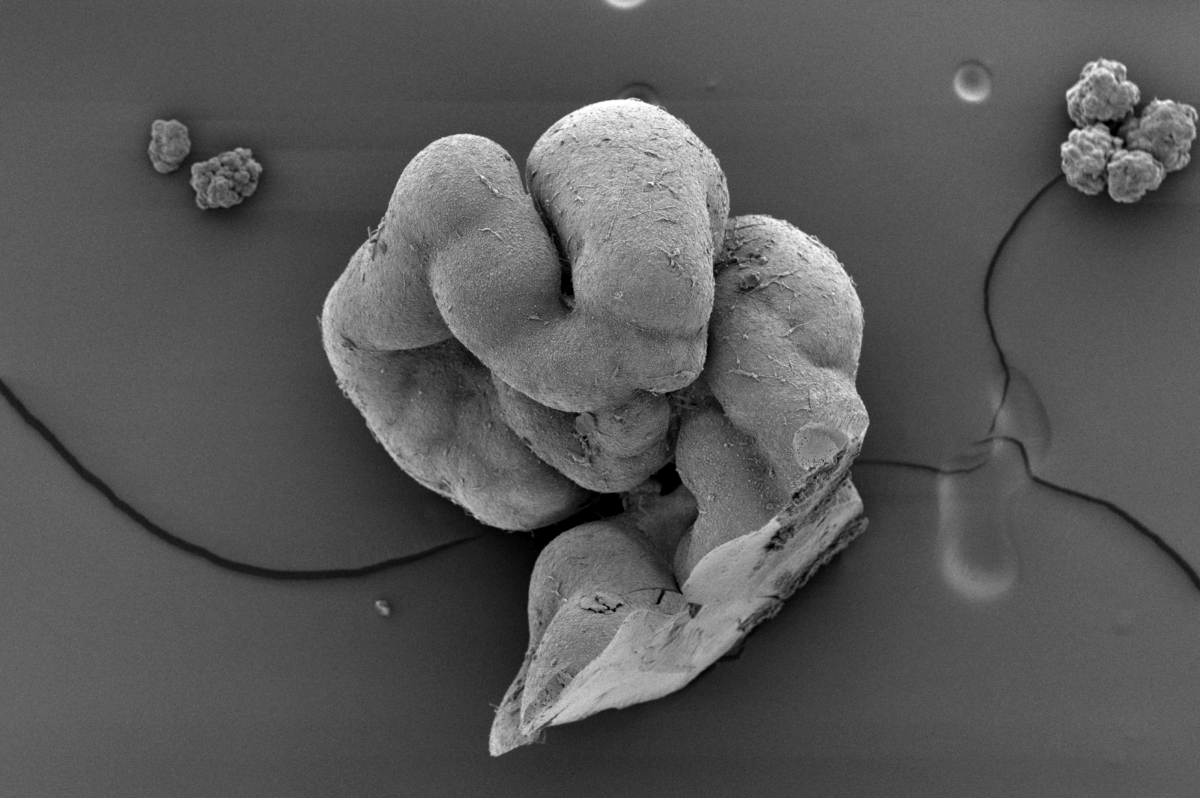
Missing link in stem cell research finally created after decades of attempts
LONDON — Scientists have achieved a major milestone in recreating human tissue development in the lab, successfully generating the notochord — a defining feature of vertebrates that acts like a biological GPS system, directing where and how our spine and nervous system should develop. This breakthrough by researchers at The Francis Crick Institute in London, brings us closer to understanding early human development and could accelerate research into spinal conditions and back pain.
Inside every human embryo, the rod-shaped notochord emerges during early development. While scientists have made significant progress in growing various human tissues in labs over the past decade, recreating the notochord has remained a persistent challenge, until now.
“It’s been difficult to generate this vital tissue in the lab, limiting our ability to study human development and disorders,” explains study senior author James Briscoe, Group Leader of the institute’s Developmental Dynamics Laboratory, in a statement. “Now that we’ve created a model which works, this opens doors to study developmental conditions which we’ve been in the dark about.”
The research team first mapped out the molecular signals that guide notochord formation during normal development. They studied chicken embryos to understand which genes were active in different cell types as tissues organized themselves, comparing their findings with existing data from mouse and monkey embryos. Their comprehensive analysis, published in the journal Nature, reveals that timing is critical. Cells need to be exposed to certain signals in a precise sequence to develop properly.
“Finding the exact chemical signals to produce notochord was like finding the right recipe,” notes Tiago Rito, the study’s first author. “Previous attempts to grow the notochord in the lab may have failed because we didn’t understand the required timing to add the ingredients.”
Armed with this knowledge, the scientists developed the framework for growing notochord tissue. They discovered that temporarily blocking two types of molecular signals — BMP and NODAL — at specific times was key. When these signals were inhibited 24 hours after initiating tissue development, many cells adopted notochord characteristics. However, blocking these signals too early or too late produced other tissue types instead.
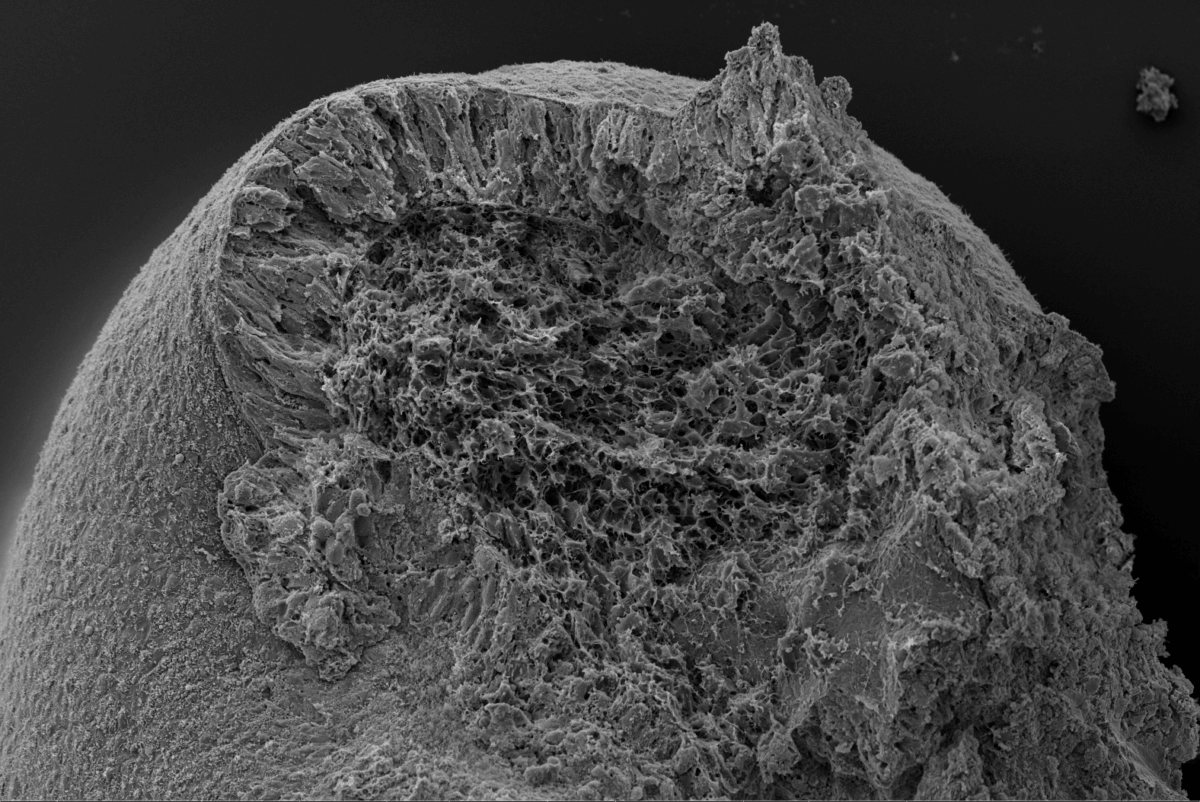
The resulting lab-grown tissues, which the team dubbed “notoroids,” developed elongated structures remarkably similar to those seen in developing embryos. These structures spontaneously elongated to 1-2 millimeters in length, containing a prominent stripe of notochord cells running through their center, surrounded by neural tissue and muscle precursor cells. About 70-75% of attempts successfully produced these structures, with the notochord cells expressing appropriate genes and exhibiting characteristic features like specialized cellular compartments and connections between cells.

“What’s particularly exciting is that the notochord in our lab-grown structures appears to function similarly to how it would in a developing embryo,” Rito explains. “It sends out chemical signals that help organize surrounding tissue, just as it would during typical development.”
The research also revealed new insights into the molecular dance that orchestrates tissue development. The team discovered that a protein called YAP helps coordinate when and where cells activate certain genes. This finding could have broader implications for understanding how complex tissues organize themselves during development.
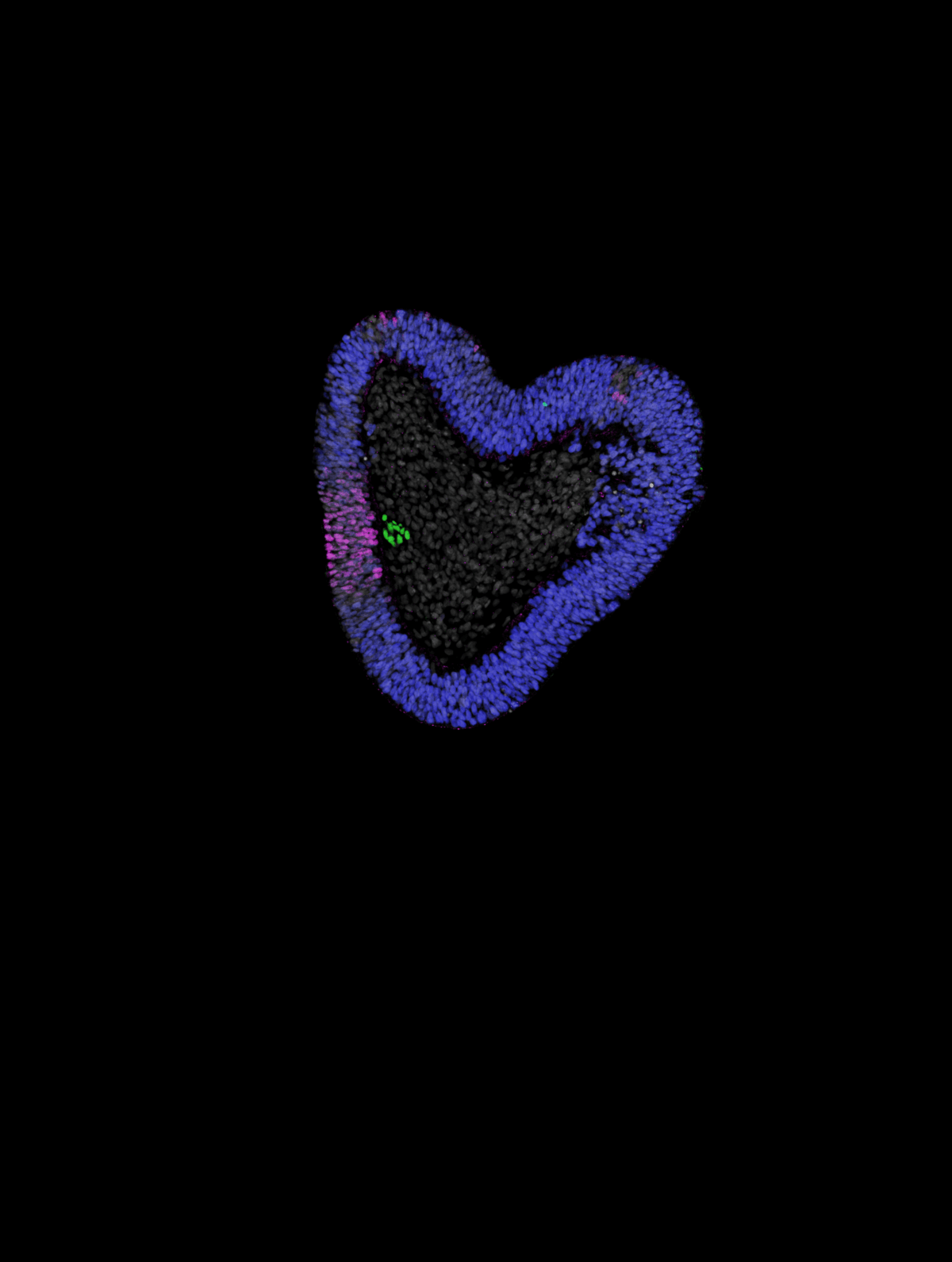
Beyond advancing our understanding of human development, this breakthrough could accelerate research into conditions affecting the spine and nervous system. The notochord plays a crucial role in these tissues’ proper formation, and abnormal notochord development has been linked to various disorders. The research might also shed light on conditions affecting intervertebral discs – the crucial shock-absorbing cushions between vertebrae that develop from notochord tissue. As these discs often degenerate with age, leading to back pain, better understanding their development could point toward new therapeutic approaches.
Looking ahead, the ability to grow notochord tissue in the lab opens new possibilities for regenerative medicine and drug development. While applications in treating patients directly are still years away, these tissues could provide invaluable testing grounds for new therapeutic approaches.
The future of developmental biology research just got a whole lot more spine-tingling.
Paper Summary
Methodology
The researchers used a systematic approach combining analysis of natural development with controlled lab experiments. They first studied chicken embryos using single-cell RNA sequencing to identify which genes were active in different cell types during development, comparing their findings with existing data from mouse and monkey embryos. This provided a molecular map of how tissues normally organize themselves. They then used this information to develop a protocol for growing human tissues from stem cells, testing different combinations and timings of molecular signals. The key breakthrough came from precisely controlling when certain signaling pathways were blocked using chemical inhibitors.
Results
The team successfully generated three-dimensional tissues containing notochord cells in about 70-75% of attempts. These “notoroids” spontaneously elongated to 1-2 millimeters in length, with notochord cells arranged in a central stripe surrounded by other tissue types. The notochord cells exhibited characteristic features visible under electron microscopes and expressed appropriate genes. Importantly, they influenced nearby neural tissue development similarly to natural notochord tissue, demonstrating functional capability beyond mere structural similarity.
Limitations
While the lab-grown tissues replicate many features of natural notochord development, they don’t perfectly match embryonic tissues. The structures are smaller and simpler than those in real embryos. Additionally, they lack some of the complex interactions with other tissues that occur during natural development. The research also relied heavily on chicken embryos for initial mapping, though key findings were validated across multiple species.
Discussion and Takeaways
This work represents a significant advance in our ability to study early human development and tissue organization. The success in generating functional notochord tissue opens new possibilities for studying developmental disorders and testing treatments, particularly for conditions affecting the spine and intervertebral discs. The research also revealed new details about how molecular signals coordinate tissue development, particularly the role of the YAP protein. These insights could have broader applications in developmental biology and regenerative medicine.
Funding and Disclosures
The research was primarily supported by The Francis Crick Institute, which receives core funding from Cancer Research UK, the UK Medical Research Council, and the Wellcome Trust. Additional support came from the Engineering and Physical Sciences Research Council and the Wellcome Trust. The authors declared no competing interests.







