
Liquid? Solid? Or something in-between? (Credit: Remigiusz Gora on Shutterstock)
The discovery challenges basic assumptions about how metals solidify
In A Nutshell
- Liquid metal contains stationary atoms: Some metal atoms remain fixed in liquid nanoparticles by bonding to defects in the graphene support, challenging the assumption that all atoms in liquids are constantly moving.
- Stationary atoms control how metal freezes: When fewer than 10 stationary atoms are randomly positioned, normal crystallization occurs. When stationary atoms form a ring around the liquid, they block crystal formation and create supercooled liquid metal.
- Supercooled liquid exists 1,000°F below normal freezing: Liquid platinum corralled by stationary atoms remains liquid down to 200-350°C, several hundred degrees below its typical solidification temperature, before transforming into an amorphous solid instead of crystals.
- Scientists can control freezing with electron beams: High-intensity electron beams create additional defects that trap more stationary atoms, allowing researchers to manipulate whether nanoparticles freeze into ordered crystals or disordered amorphous solids.
Using advanced electron microscopy, researchers have watched individual atoms transition from chaotic liquid motion to ordered solid structure, revealing that the boundary between liquid and solid metal is far more complex than previously understood. The breakthrough came from a new microscopy technique that captures metal nanoparticles freezing in real time, one atom at a time.
Scientists from Ulm University in Germany and the University of Nottingham observed platinum, palladium, and gold nanoparticles as they heated and cooled between 20 and 800 degrees Celsius. What they discovered challenges basic assumptions about how metals solidify: some atoms remain completely stationary within liquid metal, and these fixed atoms dramatically control whether freezing produces crystals or disordered amorphous solids.
“According to common understanding, the primary difference between a liquid and a solid metal lies in atomic motion,” the researchers explained in their study published in ACS Nano. “However, our research indicates that the boundary between these two phases is not as distinct as previously believed.”
Seeing Stationary Atoms in Liquid Metal
When platinum nanoparticles melted, most atoms moved so rapidly that they vanished from the microscope images. The liquid metal became nearly transparent, revealing the graphene support underneath. Yet scattered throughout this liquid, individual atoms remained visible as sharp points that stayed fixed in position for at least one second or longer.
These stationary atoms bond to vacancy defects in the graphene surface, trapping them in place through strong chemical bonds. The number and arrangement of these fixed atoms determined entirely different solidification pathways when the metal cooled.
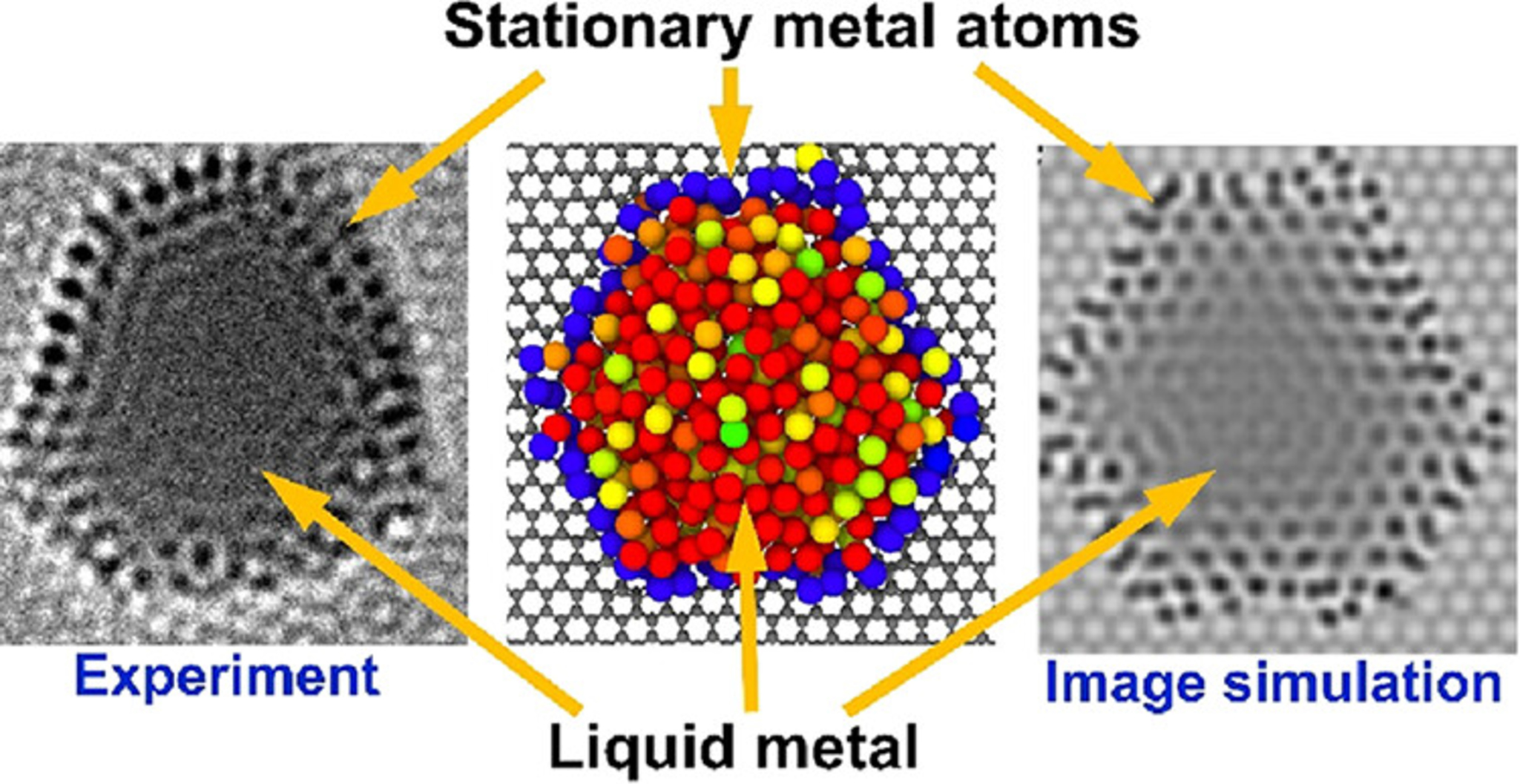
Three Different Ways Metal Freezes
The real-time observations revealed three distinct freezing mechanisms depending on the stationary atom configuration.
In nanoparticles with few randomly positioned stationary atoms, solidification followed a classical pathway. Small crystalline regions formed and dissolved repeatedly within the liquid around 500 degrees Celsius. Eventually, one survived and grew, transforming the entire particle into an ordered solid. The stationary atoms actually facilitated this process, serving as anchor points for crystal formation.
When stationary atoms formed a ring around liquid platinum, however, freezing followed a completely different route. These corralled nanoparticles remained liquid down to 200-350 degrees Celsius, which is several hundred degrees below the normal crystallization temperature. At these extremely low temperatures, the supercooled liquid finally solidified, but into an amorphous solid with randomly arranged atoms rather than an ordered crystal.
Some larger corralled particles displayed a third pathway: crystallization at the core while the edges remained amorphous, creating particles with both phases coexisting.
Controlling Freezing With the Microscope’s Beam
The research team discovered they could manipulate how particles froze by using the electron beam itself as a tool. At moderate intensity, the beam simply illuminated atoms for imaging. At high intensity, the beam knocked carbon atoms out of the graphene support, creating additional vacancy defects that trapped more metal atoms in stationary positions.
By irradiating individual liquid nanoparticles with intense electron bursts, researchers increased the number of stationary atoms surrounding them. This allowed direct comparison of how the same particle behaved with different numbers of fixed atoms.
Liquid particles could even be solidified instantly by high-intensity irradiation without changing temperature, transforming them directly into crystalline phases.
The team found platinum offered the best balance for observation. Palladium crystallized too quickly, solidifying within seconds. Gold remained liquid even under high-intensity irradiation because it bonds more weakly to carbon.
“Our experiments have surprised us as we directly observe the wave-particle duality of electrons in the electron beam. We visualize the material using electrons as waves. At the same time, electrons behave like particles, delivering discrete bursts of momentum that can either move or, surprisingly, even fix atoms at the edge of a liquid metal,” said study co-author Ute Kaiser, a professor at Ulm University, in a statement. “This remarkable observation has allowed us to discover a new phase of matter.”
Why Corralled Metals Won’t Freeze Normally
Two factors prevent corralled liquid nanoparticles from freezing into crystals.
First, the spacing between stationary atoms along the corral’s edge doesn’t match the atomic distances required for platinum’s crystal structure. This geometrical mismatch blocks crystal formation from the edges inward.
Second, solid platinum occupies less volume than liquid platinum, meaning crystallization requires shrinking. Corralled droplets face constraints from strong platinum-carbon bonds and the extreme stiffness of graphene. Creating the pressure needed for crystallization becomes unfavorable, raising an additional barrier.
Computer simulations supported these mechanisms. Calculations showed platinum atoms in corralled droplet centers moving significantly while edge atoms remained localized. As simulated particles cooled, they formed amorphous solids rather than crystals, precisely matching experimental observations.
What This Means for Catalysts and Materials
Metal nanoparticles on carbon supports serve as catalysts in numerous industrial chemical processes. Most current understanding assumes these particles behave uniformly based on size and composition.
The discovery that stationary atoms exist in varying numbers depending on local defects suggests catalytic activity may vary significantly between particles in the same sample, all experiencing identical temperatures but behaving differently based on their immediate atomic environment. This variability raises questions about measurements that may obscure important diversity in nanoscale behavior.
“The discovery of a new hybrid state of metal is significant. Since platinum on carbon is one of the most widely used catalysts globally, finding a confined liquid state with non-classical phase behavior could change our understanding of how catalysts work,” said co-author Dr. Jesum Alves Fernandes, an expert in catalysis at the University of Nottingham. “This advancement may lead to the design of self-cleaning catalysts with improved activity and longevity.”
The research demonstrates that atomic corrals can now confine liquids at the nanoscale, revealing exotic phase behavior. Whether this effect could be harnessed to design self-regenerating catalysts or advanced energy materials remains an open question, but the ability to watch and control solidification atom by atom opens new possibilities for materials engineering.
Paper Summary
Limitations
The study’s methodology works best for platinum nanoparticles on graphene. Palladium nanoparticles crystallize too rapidly under even moderate electron beam exposure to allow detailed observation of solidification dynamics. Gold nanoparticles remain liquid even under high electron beam flux because gold bonds more weakly to carbon than platinum, preventing the formation of sufficient stationary atoms to influence solidification pathways. The temperature range studied was limited to 20-800 degrees Celsius. Additionally, the embedded atom method (EAM) and modified EAM potentials used in simulations required elevated temperatures to observe phase transitions, introducing deviations from experimental conditions. The simulations also required constraining carbon atom movement to prevent instabilities at graphene edges.
Funding and Disclosures
The authors acknowledge financial support from the DFG (German Research Foundation), EPSRC (grant number EP/V000055/1), and the Royal Society. The authors declare no competing financial interests.
Publication Details
Leist, Christopher; Ghaderzadeh, Sadegh; Kohlrausch, Emerson C.; Biskupek, Johannes; Norman, Luke T.; Popov, Ilya; Alves Fernandes, Jesum; Kaiser, Ute; Besley, Elena; Khlobystov, Andrei N. “Stationary Atoms in Liquid Metals and Their Role in Solidification Mechanisms.” ACS Nano, December 9, 2025. DOI: 10.1021/acsnano.5c08201.
Authors are affiliated with: Central Facility Materials Science Electron Microscopy and Institute for Quantum Optics, Ulm University, Ulm 89081, Germany; and School of Chemistry, University of Nottingham, Nottingham NG7 2RD, United Kingdom.







