
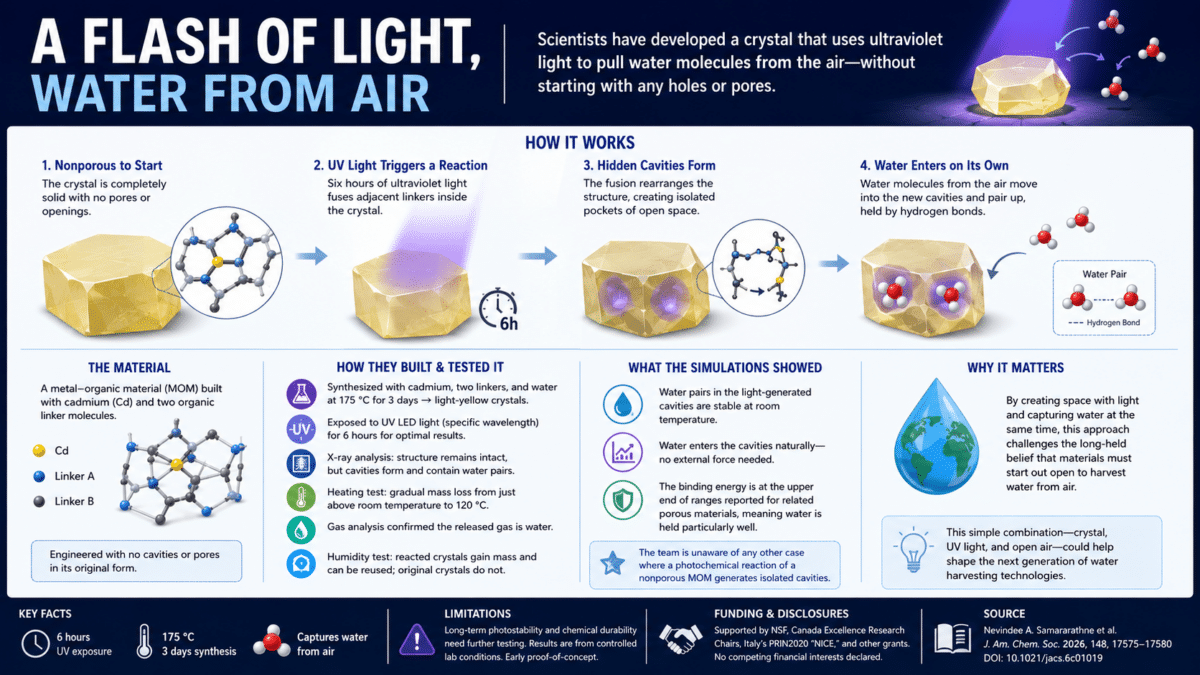
Chemists led by the University of Iowa have created a three-dimensional structure that captures water from the air and stores it. The box-like illustration in the center shows the structure before (left side) and after undergoing a chemical reaction involving sunlight (flames in center). The resulting lattice (right side) includes cavities that capture and store water molecules. The diagrams in the upper left and lower right show the chemical arrangement before and after the light-induced reaction. (Credit: Nevindee Samararathne and Leonard MacGillivray group at University of Iowa/Universite de Sherbrooke, Canada, Tamador Alkhidir and Sharmarke Mohamed group at Khalifa University of Science and Technology, Abu Dhabi, United Arab Emirates)
Researchers Used Light to Make a Sealed Crystal Absorb Water From the Atmosphere
In A Nutshell
- Scientists developed a crystal with no pores or openings that captures water directly from the air after just six hours of UV light exposure
- The light triggers a chemical reaction that creates cavities inside the crystal where none existed before, and water moves in on its own
- Lab tests confirmed the material can absorb moisture repeatedly, suggesting it may be reusable
- This is an early proof-of-concept, not a ready-made solution, and significant testing is still needed before any real-world application could be considered
A crystal sits under a beam of ultraviolet light. It has no holes, no openings, no internal structure capable of holding anything. Six hours later, it has quietly pulled water molecules straight out of the air, with no water source nearby. Scientists say this is not a trick. It is chemistry, and it may point toward a new way to think about addressing one of the world’s most pressing problems, though it is an early laboratory result, not a working device.
Researchers have developed a crystalline material that, when exposed to ultraviolet light, captures water molecules directly from the surrounding air. Before the light hits it, the material has absolutely no openings or pores. The light essentially carves out space inside the crystal where none existed before, and water moves in. A paper describing the work was published in the Journal of the American Chemical Society.
According to the United Nations, water stress is projected to affect nearly 5 billion people by 2050. Technologies that can pull water from the atmosphere are increasingly seen as one possible piece of the puzzle, especially if they can eventually work under dry conditions. Most materials designed to capture atmospheric water rely on having permanent, open chambers built into their structure from the start. This new research flips that idea entirely.
Still, this is an early proof-of-concept, and the material would need substantial safety, durability, and performance testing before anyone could think about real-world use.
Light-Activated Crystal Captures Water From Air With No Pre-Built Pores
At the heart of this research is a class of engineered solid materials called metal-organic materials, or MOMs, microscopic frameworks built from metal atoms connected by carbon-based molecular rods. Scientists have long used similar structures for capturing gases and water because they can be designed with precisely shaped internal chambers. This new material was intentionally built without any chambers at all.
The team synthesized a material built around the metal cadmium, linked together by two types of organic molecular connectors. In its starting form, the crystal is completely solid, with no gaps, no cavities, and no room for water. When hit with ultraviolet light for six hours, a chemical reaction occurs between the molecular connectors inside the crystal. Two adjacent connector molecules fuse together, and that fusion creates isolated pockets of open space within the crystal structure.
Those newly formed pockets then fill with water pulled from the surrounding air, even though no one added water to the system. Shorter or longer exposure times produced lower yields or cracked crystals, so six hours worked best under the study conditions.
How Scientists Confirmed the Water Was Really There
After UV exposure, the team used X-ray analysis to examine the crystals in detail. The crystal maintained its overall structure throughout the transformation, but two things changed: the internal geometry shifted, and isolated cavities appeared. Inside those cavities, the X-ray data revealed water molecules arranged in pairs, held together by the same type of attraction that holds ordinary liquid water molecules together.
To confirm the water was present, the team ran several additional tests. A heating test showed gradual mass loss beginning just above room temperature and continuing up to 120 degrees Celsius, consistent with water escaping the cavities. A gas analysis confirmed that what was leaving the crystal was indeed water. A humidity-exposure test on the reacted crystals showed a measurable mass increase when exposed to moisture, while the original unreacted crystals showed no change at all. That same test suggested the material could be reused, a factor that would matter for any practical water-harvesting technology, though real-world durability has not been tested.
Computer Simulations Show Why the Water Stays Put
To better understand the mechanism, the research team ran computer simulations and energy calculations. The simulations showed that water pairs inside the cavities remain stable at room temperature, staying bonded to each other and to the surrounding crystal framework rather than drifting apart.
Energy calculations showed that water moving into the light-generated cavities happens naturally without needing to be forced. The researchers also noted that they are “unaware of a case wherein a photochemical reaction of a nonporous MOM generates isolated cavities,” meaning, as far as they know, no one has ever reported a completely solid material spontaneously opening up and trapping water simply from exposure to light.
A New Starting Point for Water-Harvesting Material Design
What this research puts in question is a long-held assumption in the field: that to capture water, a material must start out open. Here, the act of capturing water and the act of creating space for it happen simultaneously, triggered by nothing more than light.
The material may not be ready to put on rooftops, but the principle it demonstrates could give scientists a new way to think about designing water-harvesting materials.
Paper Notes
Limitations
The authors acknowledged that photostability and chemical durability tests are essential for practical applications, meaning the material’s long-term performance under real-world conditions has not yet been characterized. The study was conducted under controlled laboratory conditions, and how the material behaves across varied humidity levels, temperatures, or light intensities was not reported. Water occupancy values in the crystal cavities were partial, meaning not every cavity was completely filled, which may reflect limitations in capture efficiency. The material also contains cadmium, a toxic metal, which raises questions about safety for any scaled or consumer-facing application that the paper does not address. The work is an early proof-of-concept, and the path from laboratory demonstration to scalable water harvesting involves many additional steps the study did not address.
Funding and Disclosures
Support came from the National Science Foundation under grant numbers DMR-2221086 and CHE-1828117, the Canada Excellence Research Chairs Program, and the Italian Ministry of University and Research through the PRIN2020 “Nature Inspired Crystal Engineering (NICE)” grant. One author received postdoctoral fellowship funding from Institut Courtois and Fonds de recherche du Québec. Computational work received support from Khalifa University of Science and Technology through a Research and Innovation Grant (KU-INT-RIG-2023-054) and the Khalifa University Center for Catalysis and Separations, with high-performance computing resources also provided by Khalifa University. The authors declared no competing financial interests.
Publication Details
Authors: Nevindee A. Samararathne, Davide M. Proserpio, Eric Reinheimer, Farshid Effaty, Tamador Alkhidir, Sharmarke Mohamed, and Leonard R. MacGillivray (corresponding author, University of Iowa and Université de Sherbrooke) Journal: Journal of the American Chemical Society, 2026, Volume 148, Pages 17575–17580 Paper Title: “Photo Capture of Water by Single Crystals of a Nonporous Metal–Organic Material” DOI: https://doi.org/10.1021/jacs.6c01019







