
Credit: Ground Picture on Shutterstock
In A Nutshell
- Korean researchers developed SC Fill paste, an injectable tissue filler made from processed human skin that could eventually help breast cancer patients who need reconstruction after tumor removal
- In six-month rat testing, the material promoted blood vessel growth and attracted tissue-building cells while showing inflammation that peaked early and then declined over time
- The paste showed some advantages over commercial products (better blood vessel formation, more cell recruitment) but also some drawbacks (thicker capsule formation at six months compared to one control product)
- This is early-stage animal research using subcutaneous implants in rats, not human trials or actual breast reconstruction surgery; clinical applications are still years away
Women who undergo breast-conserving surgery after a cancer diagnosis often face a difficult trade-off between removing enough tissue to ensure safety and maintaining the natural appearance of their breasts. For patients with smaller breasts or larger tumors, this balance can seem impossible to achieve. Korean researchers have developed what may be a better solution.
Surgeons usually rely on techniques that rearrange existing breast tissue, but these approaches frequently fall short when there isn’t enough tissue to work with. Borrowing tissue from other parts of the body is an option, but it comes with its own complications, including additional scarring and longer recovery times.
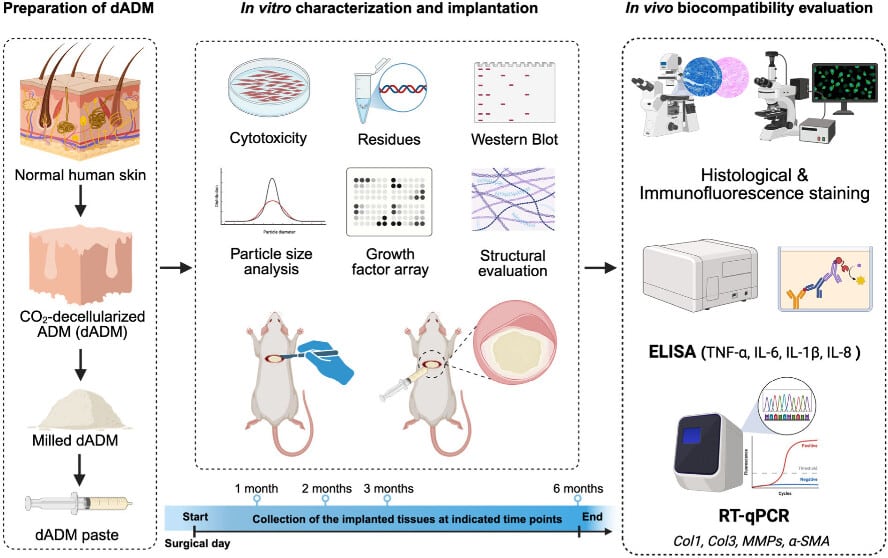
A team from Seoul National University Bundang Hospital created SC Fill paste, an injectable tissue filler made from processed human skin. The product could help surgeons fill the gaps left behind after tumor removal without requiring complex reconstructive procedures. The research, published in ACS Applied Bio Materials, shows this paste-like material demonstrates signs of integration with surrounding tissue over time in a rat model, with inflammation markers that peaked early (at the first measured time point) and then trended down over six months.
Injectable Filler Adapts to Individual Defects
SC Fill paste differs from the sheet-form materials surgeons currently use in breast reconstruction. Those sheets need to be shaped and sutured into place, which can be challenging when the defect has an irregular shape or extends into deeper tissue. The injectable format allows surgeons to adapt the material to the specific geometry of each patient’s defect.
Dr. Chan-Yeong Heo and his colleagues processed donated human skin through a series of steps that remove all cellular material while retaining some key extracellular-matrix components and measurable levels of several growth factors that help tissues rebuild themselves. They then ground the material into microscopic particles averaging about 190 micrometers in size and mixed it into a paste that can be delivered through a syringe.

Reduced Immune Response in Breast Reconstruction Material
The processed material showed dramatically reduced levels of components that typically trigger immune responses. Double-stranded DNA content dropped to 18 nanograms per milligram, well below the 50 ng/mg commonly accepted threshold for implanted materials. Laboratory analysis confirmed complete elimination of MHC-I proteins (one of the primary triggers for tissue rejection). At the same time, the paste retained more than twice the collagen content of PC-1, a commercial micronized acellular dermal matrix control product, and showed moderate retention of growth factors known to promote healing and blood vessel formation, though overall levels remained lower than native tissue.
Six-Month Testing Shows Promising Results
Testing the material in rats provided evidence about how it behaves after implantation. The researchers implanted small amounts of SC Fill paste under the skin of 80 animals and tracked what happened over six months, comparing the results against two commercial tissue products. The testing was performed in subcutaneous tissue on the rats’ backs, not in breast defect reconstruction.
SC Fill paste promoted robust blood vessel formation and attracted more fibroblasts (the cells responsible for creating new tissue). Blood vessel counts within the implanted material increased progressively, with SC Fill paste showing significantly higher vessel numbers at three and six months compared to PC-1. The material also attracted significantly more fibroblasts than the commercial products at the one-month mark.
Inflammatory markers revealed favorable responses. Levels of proteins like TNF-α, IL-1β, IL-6, and IL-8 peaked at the one-month mark (the first time point measured) but declined steadily over the following months. By the six-month mark, SC Fill paste samples showed significantly lower levels of IL-1β compared to the one-month time point, meaning the inflammatory response had largely resolved. SC Fill paste also maintained comparable or lower inflammatory marker levels than the commercial products throughout most of the observation period.
What Integration Looked Like in Rats
The material appeared to undergo appropriate remodeling. Expression of enzymes that break down and rebuild tissue scaffolds followed expected patterns. These enzymes were active early on but declined over time as new collagen accumulated. Type I collagen increased gradually throughout the six-month period, while type III collagen peaked early and then decreased (a pattern consistent with typical wound-healing phases). Researchers interpret this as evidence that the body treats the implanted material as a framework for new tissue rather than as foreign material requiring isolation.
For patients, these biological details could translate into better outcomes if the material performs similarly in humans. A filler that promotes blood vessel growth and attracts the right kinds of cells while showing declining inflammation should integrate more naturally than one that provokes persistent immune responses. The injectable format offers practical advantages, allowing surgeons to fill complex defects without additional incisions or complicated shaping procedures.
The study has limitations worth noting. Researchers conducted all animal testing in rats, whose immune systems and tissue healing processes differ from humans in important ways. The six-month observation period captured the acute and intermediate phases of tissue integration but may have missed longer-term issues. The authors note future optimization needs, including improving mechanical properties such as elastin content and how well the paste maintains form after placement.
Editor’s Note: This article reports on early-stage animal research conducted in rats over six months. The testing was performed using subcutaneous implants on rats’ backs, not in actual breast tissue or breast defect reconstruction. While the results show promise, significant additional research, including larger animal studies and eventual human clinical trials, would be required before this material could be used in patients. The study also showed mixed results, with the experimental material outperforming commercial controls in some measures (vascularization, cell recruitment) while showing thicker capsule formation compared to one control product at the six-month mark. Readers should not interpret this research as indicating an immediately available treatment option.
Paper Notes
Study Limitations
The study was conducted exclusively in male Sprague-Dawley rats over a six-month period, which may not fully replicate human tissue responses or long-term integration patterns. The research team noted that future work should focus on optimizing shape retention after implantation and improving elastin content preservation to better match native tissue properties. Larger animal models, such as porcine models, would provide more clinically relevant data before human trials. The study focused on subcutaneous implantation in the dorsal region rather than actual breast defect reconstruction, which may not capture all variables present in surgical applications.
Regarding capsule formation, the measured results showed that PC-1 had significantly thinner capsule formation than SC Fill paste at six months (p = 0.009). However, SC Fill paste demonstrated superior performance in other key measures including vascularization (significantly higher blood vessel counts at 3 and 6 months compared to PC-1), fibroblast recruitment (significantly higher at 1 month), and sustained reduction in inflammatory markers over time. The authors still describe the overall capsule profile as favorable in their Discussion, even though this specific PC-1 comparison favored PC-1 at six months.
Funding and Disclosures
This work was supported by the Technology Innovation Program (Project No. 20015793), funded by the Ministry of Trade, Industry, and Energy (MOTIE, Korea). Authors Thuy-Tien Thi Trinh, Young Hyun Lee, and Chan Yeong Heo were employed by the Korean Institute of Nonclinical Study Center (KINS). Authors Yong Woo Shin and Jin Woo Bae were employed by DOF Inc., a company that may have commercial interests in the development of acellular dermal matrix products.
Publication Details
Authors: Linh Thi Thuy Le, Thuy-Tien Thi Trinh, Nguyen Ngan Giang, Xin Rui Zhang, Yong Xun Jin, Yong Hyun Lee, Yong Woo Shin, Jin Woo Bae, Pham Ngoc Chien, and Chan-Yeong Heo
Affiliations: Department of Plastic and Reconstructive Surgery, Seoul National University Bundang Hospital; Department of Biomedical Science, College of Medicine, Seoul National University; Korean Institute of Nonclinical Study Center; DOF Inc.; Hai Phong University of Medicine and Pharmacy, Vietnam; Peking Union Medical College Hospital, China
Journal: ACS Applied Bio Materials | Title: “Development and Evaluation of an Injectable Acellular Dermal Matrix for Breast Reconstruction” | DOI: 10.1021/acsabm.5c01538 | Publication Date: December 4, 2025 (online); 2025, Volume 8, pages 10827-10838
Animal Ethics Approval: Institutional Animal Care and Use Committee of Seoul National University Bundang Hospital (BA-2110-329-008-01), conducted in compliance with ARRIVE guidelines and NIH Guide for Care and Use of Laboratory Animals
Human Tissue Ethics: Institutional Review Board of Seoul Asan Medical Center (IRB No. 20201305), conducted in accordance with Declaration of Helsinki principles







