
A centenarian blows out her 100th birthday candles. (Photo by Dan Negureanu on Shutterstock)
The Hydra Barely Ages. Scientists Want to Test What Its Genes Can Teach Us
In A Nutshell
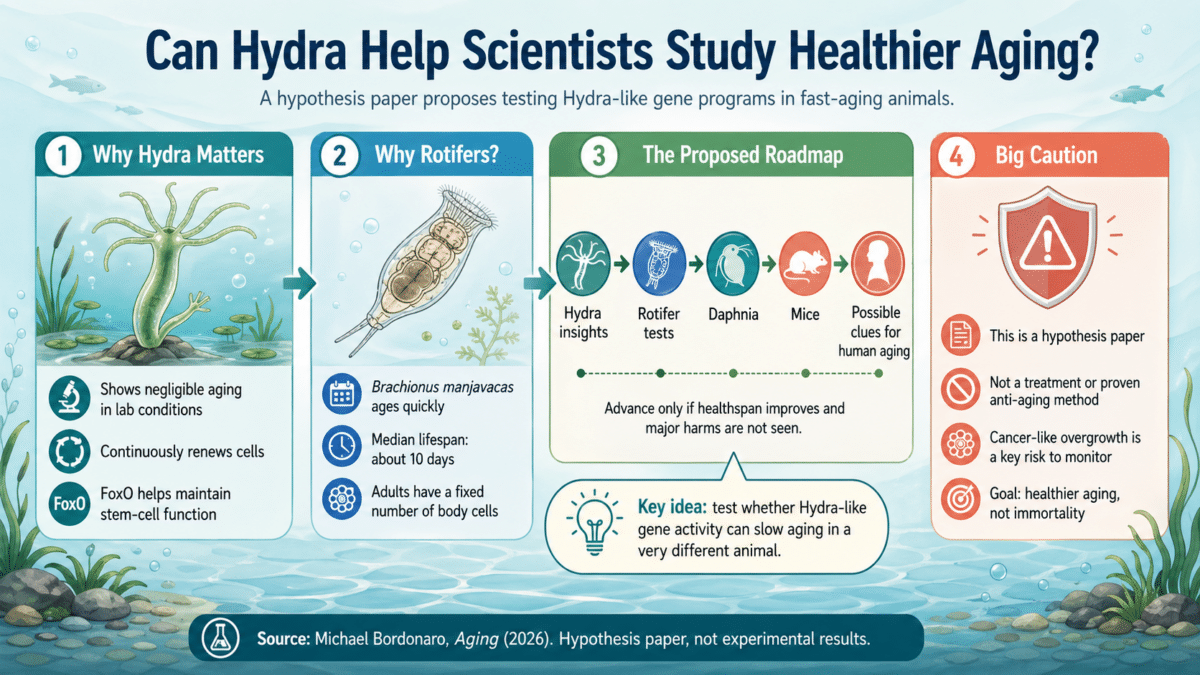
- Hydra, a tiny freshwater animal, shows no detectable increase in its risk of dying with age under lab conditions, making it one of the most studied organisms in aging research.
- A new hypothesis paper proposes using CRISPR to introduce Hydra’s anti-aging gene patterns into rotifers, microscopic animals that live just 10 days, as a first test of whether aging can be deliberately slowed.
- A key protein called FoxO appears to drive Hydra’s cellular staying power, and variations of the same gene family have been linked to longevity in humans.
- Slowing aging may raise cancer risk, since cellular aging also acts as a brake on tumor growth, and the proposed experiments include safety monitoring at every stage.
A freshwater animal no bigger than a fingernail has baffled biologists for decades. Hydra, a simple tube-shaped organism found in ponds and lakes, doesn’t appear to grow old. Its cells keep dividing, damaged tissue gets swapped out for fresh copies, and under laboratory conditions, scientists have not detected any increase in its risk of dying with age, though Hydra can still die from starvation, injury, predation, or other outside causes.
Now, a new hypothesis paper lays out a step-by-step plan to borrow parts of the Hydra’s anti-aging genetic playbook and test whether it can slow down aging in animals that do grow old, starting with one of nature’s fastest-aging creatures and eventually working toward strategies that might one day benefit people.
The proposal, published in the journal Aging, comes from Michael Bordonaro of Geisinger College of Health Sciences. Rather than cataloging what makes the Hydra special, Bordonaro outlines a practical experimental roadmap: take the genes responsible for the Hydra’s staying power and introduce their activity patterns into rotifers, microscopic animals that live a median of just 10 days.
If an animal hardwired for fast aging can be made to age more slowly using genetic tools borrowed from an animal that barely ages at all, that would be an important proof-of-principle that these pathways can be nudged in a very different animal.
Hydra Stem Cells Hold the Key to Its Ageless Biology
Hydra constantly churns out new cells from a reservoir of stem cells, replacing old or damaged ones without pause. Rotifers, by contrast, are born with a fixed number of body cells and never make more. One is essentially ageless. The other is a ticking clock. Bridging that gap, even partially, would suggest the anti-aging mechanisms at work aren’t exclusive to the Hydra’s unusual biology but could potentially be used in other animals, including mammals.
At the center of the Hydra’s agelessness is a protein called FoxO, a molecular switch that keeps stem cells in a youthful, self-renewing state. When researchers dialed down FoxO activity in Hydra, the animals’ growth slowed and their cells shifted toward a more “finished” state. When FoxO was cranked up, genes associated with stem cell identity switched on, even in cells that had already specialized for other jobs. Different versions of FoxO-related genes have also been linked to human longevity. Past attempts to manipulate FoxO in mammals and flies have produced mixed results, though, so the path from Hydra biology to human application remains long and uncertain.
Rotifers Carry More Human-Relevant Genes Than Most Lab Animals
One detail that strengthens the case for rotifers is their surprisingly human-relevant genetics. Unlike roundworms or fruit flies, rotifers have retained a more complete set of genes shared with vertebrates, including genes involved in stress responses, metabolism, and toxin resistance that are absent from worms and flies but present in people. Research has also shown that aging-related gene changes in rotifers begin surprisingly early in life, suggesting any intervention would work best if started young.
Hydra’s Anti-Aging Genes Could Reshape the Aging Clock in Rotifers
Bordonaro doesn’t propose a single experiment but rather a series of increasingly involved tests, each building on the last. Using CRISPR, a tool that allows scientists to precisely edit an organism’s DNA much like a find-and-replace function in a word processor, researchers would boost FoxO and a handful of other stem-cell-related genes in rotifers. Scientists would then track whether the modified rotifers live longer, stay more active, resist stress better, and maintain healthier cells compared to unmodified controls. The plan also works in both directions: introducing rotifer aging patterns into Hydra to see if that accelerates aging in the normally ageless creature, helping researchers identify which genetic changes are truly causing the anti-aging effects.
If rotifer experiments succeed, the next testing ground would be Daphnia, tiny crustaceans commonly called water fleas, followed by mice, including MRL mice, a strain known for unusual healing abilities, including efficient repair in multiple tissue types with little fibrotic scarring. Despite their superior healing, MRL mice actually have shorter lifespans than other strains, possibly because of elevated growth-promoting hormone levels. That paradox makes them a compelling test case.
The paper also points to the naked mole rat as a useful reference. These wrinkly rodents show unusual longevity and cancer resistance compared with many other mammals, a combination that makes their genetic profile a potential guide for managing the risks that come with suppressing aging.
Longevity, With a Catch?
Bordonaro is upfront about a serious concern: suppressing aging might come with a dangerous side effect. Cellular aging serves as a natural brake on cancer. Weaken that brake too much, or push cells toward a more youthful, dividing state in the wrong context, and damaged cells that would normally be shut down could instead keep multiplying, potentially becoming cancerous. Safety monitoring and genetic safeguards are built into the plan at every stage to manage this risk.
Human translation remains highly speculative and would require years of intermediate mammal studies. Still, as Bordonaro argues, without taking the initial step from Hydra to rotifer, nothing further would be possible. It’s a long road, but somewhere between a nearly immortal pond creature and a microscopic animal that’s dead in 10 days, the biology of aging may finally start to give up some of its secrets.
Disclaimer: This article is based on a hypothesis paper and does not describe completed experiments or proven results. The proposed research is in early theoretical stages, and any potential applications to human health remain highly speculative and years away from practical consideration.
Paper Notes
Limitations
The paper is a hypothesis and does not present original experimental data. The author acknowledges that the proposed experiments are “highly speculative” and that extending findings from rotifers to mammals faces substantial hurdles. The fixed adult cell number of rotifers fundamentally limits the potential for stem cell expansion, meaning any benefits would likely come from improved maintenance of existing cells rather than tissue renewal. Stable, heritable, multi-gene manipulations using CRISPR in microscopic organisms remain technically demanding, with risks including off-target effects, variable gene expression, and silencing of inserted genes across generations. Prior attempts to manipulate FoxO in mammals and flies have produced mixed results: some showed lifespan extension while others showed no benefit, accelerated aging, muscle wasting, or increased cancer-like growths. FoxO also regulates antimicrobial defenses and the microbiome, meaning manipulating it could have unpredictable side effects. Full organismal immortality in complex animals is described as not possible due to tissue complexity and evolutionary trade-offs.
Funding and Disclosures
The author declares no conflicts of interest. The work was supported by the Geisinger College of Health Sciences.
Publication Details
Author: Michael Bordonaro, Department of Medical Education, Geisinger College of Health Sciences, Scranton, PA, USA. Journal: Aging (2026, Vol. 18). Paper Title: “From Hydra to rotifer and beyond: implications for human aging and delayed senescence.” Published: April 8, 2026. Received: January 5, 2026. Accepted: March 20, 2026. DOI: not specified in source. Copyright: Open access under Creative Commons Attribution License (CC BY 4.0). This is a hypothesis paper, not a report of completed experimental research.







