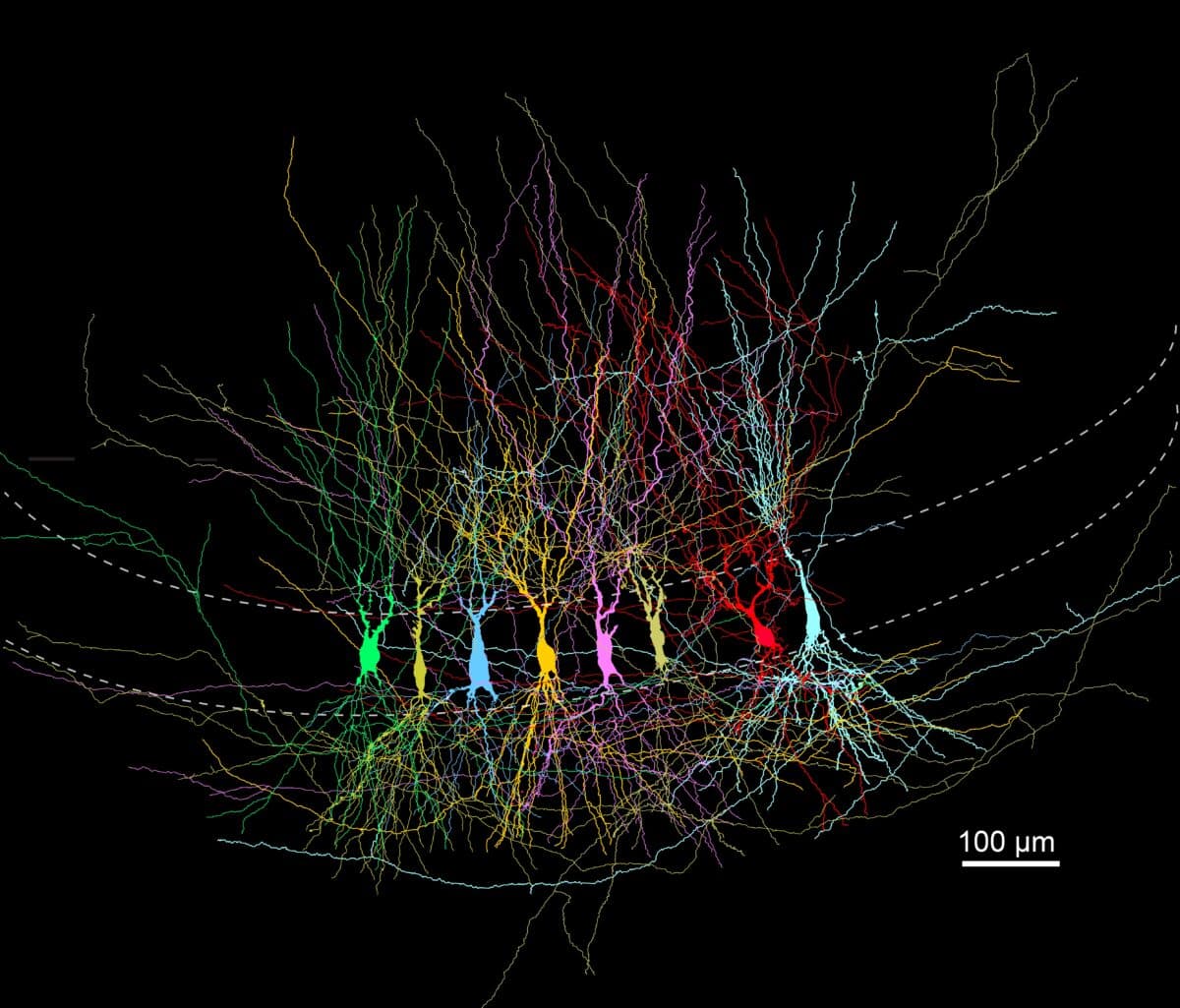
Neurons filled with biocytin—a tracer that labels them during recording—are fixed and stained to allow full reconstruction of their shapes. © Jose Guzman / Jonas group at ISTA
In A Nutshell
- A new study found that the brain’s memory circuitry is not fixed at birth but is dramatically reorganized during early development, becoming leaner and more structured over time.
- Researchers tracked changes in a key memory region of mouse brains from infancy to early adulthood, finding that neuron connections dropped sharply and became more selectively organized.
- Early in development, some brain connections were strong enough to trigger a chain reaction from a single signal; in mature brains, neurons required input from roughly 25 sources before responding.
- The rewiring likely involves a mix of built-in biological programs and experience-dependent processes, though the study did not directly confirm which experiences drive the changes.
A newborn mouse’s brain is a surprisingly chatty place. Neurons in a memory region fire signals to their close neighbors in a dense, seemingly random web of connections. But by the time that mouse reaches early adulthood, the wiring has been dramatically pruned and reorganized into a lean, efficient architecture, one shaped over time through a mix of built-in developmental programs and experience-dependent pruning.
That is the central finding of a new study published in Nature Communications by researchers at the Institute of Science and Technology Austria. What they found speaks to a longstanding question in neuroscience: is the brain’s memory circuitry hardwired from birth, or does it take shape over time? The answer points strongly toward a circuit that changes over time, rather than one that arrives fully formed.
Called CA3, the region studied is part of the hippocampus, the seahorse-shaped structure deep in the brain central to forming and retrieving memories. CA3 neurons connect back to each other in loops that allow the network to store patterns and fill in missing information. Someone who recalls an entire vacation from a single photograph is drawing on exactly this circuit.
How the Mouse Brain’s Memory Network Changes Over Time
To trace how this network develops, the research team simultaneously recorded electrical signals from up to eight CA3 neurons at a time in thin slices of mouse brain tissue at three stages: around one week after birth, three weeks, and seven weeks, roughly equivalent to infancy, the toddler stage, and early adulthood in mice.
What they discovered was a sweeping transformation. In the youngest mice, neurons were densely connected to their immediate neighbors in what appeared to be a random pattern. By seven weeks, the network had become something very different. Connections were sparser, more spread out, and organized in a structured, non-random way. In total, the team tested 7,736 potential connections. Connection probability dropped from roughly 6.4% in the youngest mice to about 2.2% in the most mature group.
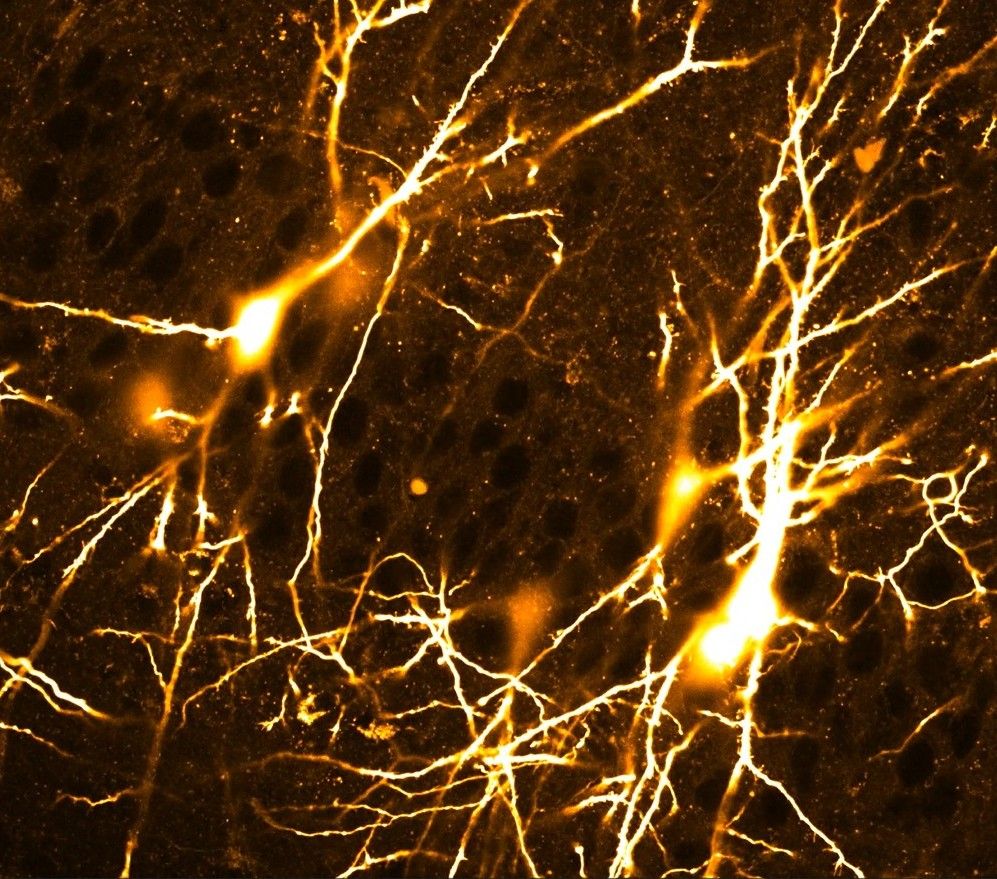
How Developing Brains Process Signals Differently
Perhaps equally surprising was what happened to the strength of individual connections as the mice grew older. In the youngest mice, some connections were strong enough that repeated firing from one presynaptic neuron could trigger the receiving neuron to fire, a near-detonation effect seen in roughly 10% of tested immature pairs. That behavior disappeared entirely by later stages. By early adulthood, neurons required input from an estimated 25 sources firing together before responding, up from roughly 7 in the youngest group.
This change reshapes how the brain processes information at different ages. In infancy, the network operates more like a chain reaction: one neuron fires and the signal can cascade easily to the next. That kind of setup could be useful for basic functions early in life, but it is also prone to runaway excitation, which may help explain why immature brains are more susceptible to seizure-like activity. In contrast, the mature arrangement, where neurons require agreement from multiple sources before responding, allows for much more precise computation.
Experience May Help Sculpt the Mature Memory Circuit
The researchers suggest this rewiring likely involves experience-dependent processes, with the animal’s interactions with its environment helping to bias which connections survive and which get eliminated. One possibility is that activity helps determine which connections are kept, weakened, or lost, though the study did not directly observe that process. The authors note the result “may result from the combination of pre-programmed and experience-dependent mechanisms.” Directly manipulating experience, such as through sensory deprivation, would be needed to confirm the causal link.
The team also used statistical methods to rule out the possibility that changes in connectivity could be explained by chance or by shifts in how neurons are physically positioned. Structured patterns in the mature network were genuinely non-random.
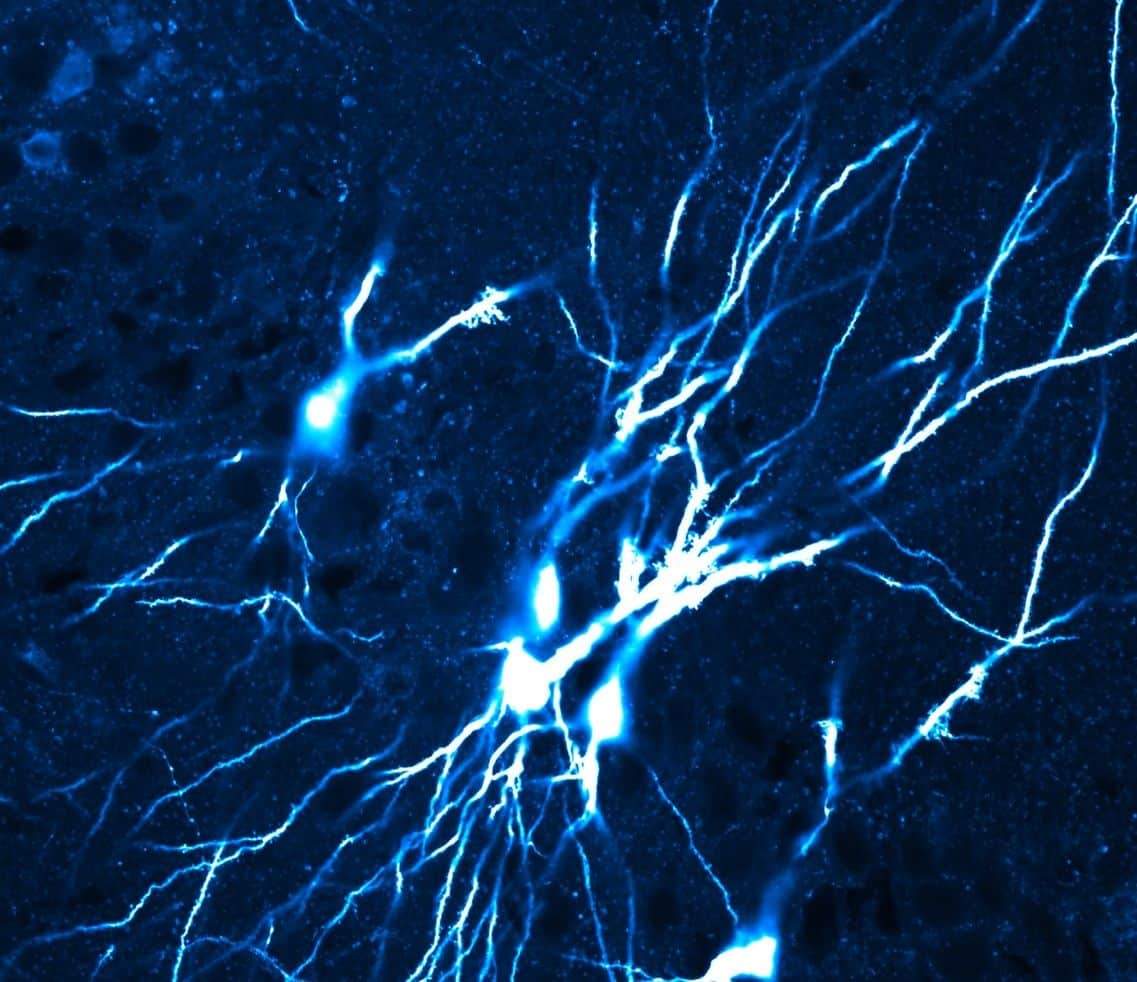
Why This Could Matter for Memory and Brain Development
When the researchers modeled a network with the properties of the immature brain, dense, strong, and random connections, it performed differently than one modeled after the mature brain. The mature version, with its sparse, weaker, and structured connections, showed clear advantages in memory functions. Switching from local to broadly distributed connectivity alone nearly doubled the model’s memory capacity.
Most previous studies of CA3 connectivity had focused on the adult brain, leaving the question of how that circuitry comes to exist largely unanswered. By showing that the mature network’s hallmark features emerge gradually over weeks of postnatal development, the study indicates that the memory system adult animals rely on, and that may offer clues about human memory development, is not a given at birth. It is assembled over time, through biological refinement that appears to depend on more than genetics alone.
Paper Notes
Limitations
The study was conducted in mouse brain slices rather than in living, behaving animals, which means the results reflect the potential of the circuitry rather than its real-time operation during actual memory tasks. The recordings captured snapshots at three developmental time points, so the precise timing and mechanisms driving the transition between states remain to be fully characterized. Additionally, while the authors suggest that experience-dependent mechanisms shape the mature network, the study did not directly manipulate experience to confirm this causal link, and the authors explicitly note the result may stem from a combination of pre-programmed and experience-dependent processes. The computer models, while biologically inspired, necessarily simplify many aspects of real neural networks.
Funding and Disclosures
The project received funding from the European Union’s Horizon 2020 research and innovation programme, including ERC Advanced Grants (No. 692692 and 101199096 to Peter Jonas), Marie Sklodowska-Curie Grants (754411 to Victor Vargas-Barroso and 101026635 to Jake F. Watson), the Fond zur Forderung der Wissenschaftlichen Forschung (multiple grants to Peter Jonas), the Nomis Foundation (fellowship to Andrea Navas-Olive), and a CONACyT fellowship (289638) to Victor Vargas-Barroso. Peter Jonas has an industrial collaboration with Leica Microsystems. The other authors declared no competing interests.
Publication Details
Authors Victor Vargas-Barroso, Jake F. Watson, Andrea Navas-Olive, Alois Schlogl, and Peter Jonas are affiliated with the Institute of Science and Technology Austria (ISTA) in Klosterneuburg, Austria. The paper, “Developmental emergence of sparse and structured synaptic connectivity in the hippocampal CA3 memory circuit,” was published in Nature Communications (2026). DOI: https://doi.org/10.1038/s41467-026-71914-x. Received September 3, 2025; accepted April 3, 2026; published April 21, 2026.







