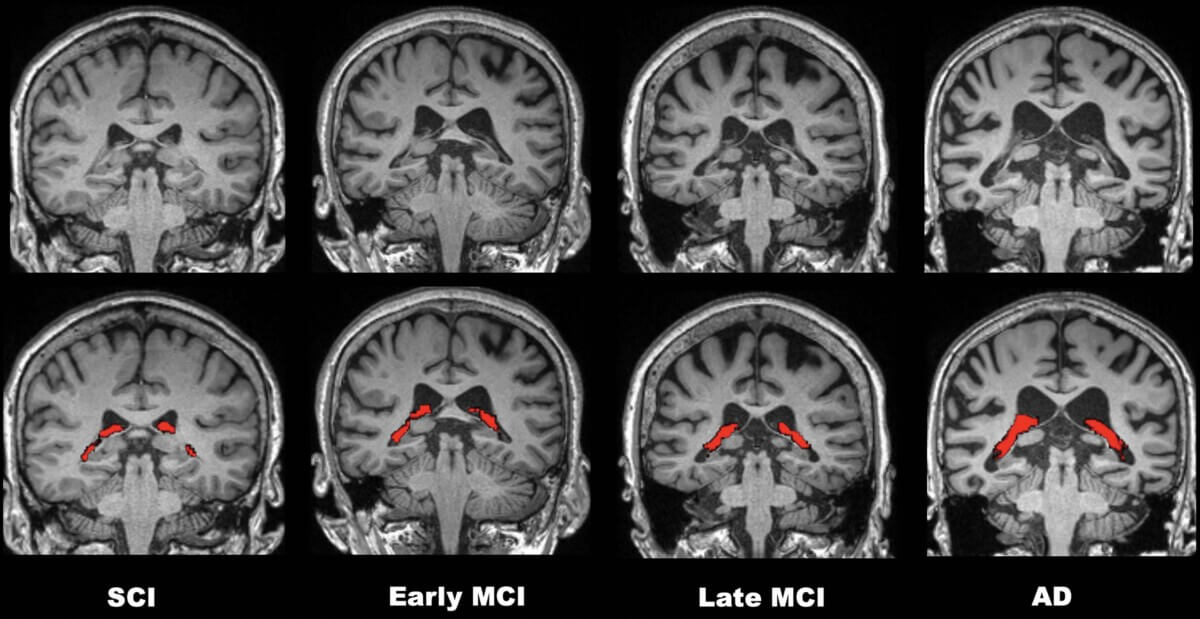
Brain scans show progression of cognitive decline from early mild cognitive impairment to Alzheimer's disease.
Microglia ‘stuff themselves with fat until they can barely function,’ study shows
In A Nutshell
- Brain immune cells called microglia clog with fat droplets in Alzheimer’s, losing their ability to clear toxic proteins.
- Female brains showed greater fat buildup in mice, helping explain higher Alzheimer’s risk in women.
- Blocking the fat-making enzyme DGAT2 restored microglia’s cleanup ability and shrank plaques by half in mice within a week.
- The findings suggest Alzheimer’s may be a metabolic disorder as much as a neurological one, opening new treatment possibilities.
WEST LAFAYETTE, Ind. — Deep inside the brains of Alzheimer’s patients, immune cells are drowning in their own fat. This buildup of fat droplets inside brain immune cells helps explain why the brain loses its ability to fight off the disease and points toward a surprising new treatment that worked in mice within a week.
Researchers from Purdue University and Cleveland Clinic have discovered that brain cells called microglia become packed with fatty deposits when exposed to toxic proteins that cause Alzheimer’s disease. As these cellular defenders accumulate more fat droplets, they transform from efficient cleanup crews into bloated, dysfunctional masses that can no longer protect the brain.
The discovery, published in Immunity, reveals that what scientists thought was simply brain inflammation may actually be a metabolic disease. Brain cells exposed to Alzheimer’s proteins don’t just become angry, they become “obese” at the cellular level, stuffing themselves with fat until they can barely function.
Researchers compared the effect to fatty liver disease: just as liver cells stop working when overloaded with fat, brain cells lose their protective abilities when they become lipid-laden.
The Microscopic Obesity Crisis
While we typically associate fat with an expanding waistline or clogged arteries, fat can also build up inside individual cells, stored in tiny compartments called lipid droplets. In healthy amounts, these droplets provide energy reserves and protect cells from damage. Too much, however, and cells become sluggish and dysfunctional.
Microglia normally patrol the brain like microscopic security guards, gobbling up cellular debris and toxic proteins before they can cause harm. But when researchers examined brain tissue from Alzheimer’s patients, they found these cells had become grotesquely swollen with fat.
Using advanced imaging techniques, scientists could peer inside individual brain cells and measure their fat content with precision. Microglia near Alzheimer’s protein clumps contained about 5.7 times more fatty deposits than cells in healthy brains. The closer cells were to toxic protein plaques, the fatter they became.
These cells looked completely different under the microscope. Instead of the lean, active cells found in healthy brains, researchers found bloated cells stuffed with fat droplets, barely able to move or function.
Why Female Brains May Be More Vulnerable To Alzheimer’s
Female mice showed particularly dramatic fat accumulation, with brain cells containing 58% more lipid droplets than males. This discovery may help explain why women face higher Alzheimer’s risk and often experience more severe symptoms.
The research examined both male and female mice at different ages, tracking how brain cell fat content changed over time. While both sexes showed increased fat accumulation with Alzheimer’s progression, female microglia consistently packed on more lipid droplets when exposed to toxic proteins.
Researchers found that human Alzheimer’s brains show the exact same cellular obesity found in mouse models.
The Cellular Fat Factory In The Brain
To understand how brain cells become obese, researchers tracked the molecular machinery responsible for fat production. When microglia encounter Alzheimer’s proteins, they undergo a dramatic metabolic shift that floods them with fatty acids, the building blocks of cellular fat.
Within just one hour of exposure to toxic amyloid-beta proteins, very long-chain saturated fatty acids spiked dramatically in brain cells. These are the same harmful fats linked to heart disease and diabetes, but now they were accumulating inside brain tissue.
Normally, cells protect themselves by converting dangerous fatty acids into safer storage forms and packaging them into lipid droplets. A specific enzyme called DGAT2 acts as the final step in this fat-storage assembly line, converting fatty acids into triacylglycerols, or essentially, cellular fat deposits.
DGAT2 functions as the foreman of a fat factory. When this enzyme goes into overdrive, cells become so packed with fat storage containers that they can’t perform their normal jobs anymore.
Brain tissue from both Alzheimer’s mice and human patients showed dramatically elevated levels of DGAT2, particularly in the fattest, most dysfunctional cells. DGAT2 enzyme activity kept driving fat production, even when cells were already overloaded.
Putting Brain Cells on a Diet
Researchers wondered whether they could reverse cellular obesity and restore brain function by targeting the fat-production machinery. They developed a compound that specifically degrades DGAT2 protein, essentially shutting down the cellular fat factory.
When they tested the treatment on isolated brain cells in laboratory dishes, the results were immediate. Fat-laden microglia that had lost their ability to clean up toxic proteins suddenly regained function when DGAT2 was blocked. They began moving again, extending cellular projections, and efficiently gobbling up Alzheimer’s proteins.
But the real test came with live animal experiments. Using tiny pumps, researchers delivered their DGAT2-degrading compound directly into the brains of elderly mice with advanced Alzheimer’s-like pathology. These animals had extensive brain damage and massive accumulations of toxic protein plaques.
After just one week of treatment, which is a remarkably short time in brain research, the results were dramatic:
- Brain cells lost 40% of their excess fat droplets
- Toxic protein plaques shrank by 51%
- Damaged neurons around plaques showed significant recovery
- The cellular cleanup machinery was restored to near-normal function
Scientists essentially put these brain cells on a crash diet. Once they lost their excess fat, they immediately got back to work protecting the brain.
Why Current Treatments Fall Short
The discovery helps explain why current Alzheimer’s treatments have been largely disappointing. Most drugs focus on removing toxic protein plaques from the brain, but they ignore the underlying problem: the brain’s natural defense system has become metabolically compromised.
Even when existing treatments successfully clear some protein deposits, they don’t address why brain cells stopped cleaning up toxins in the first place. Current treatments reduce plaques but don’t restore microglia’s cleanup ability. If the brain’s own cleanup crew is broken, removing some plaques won’t prevent new ones from forming.
The metabolic approach targets the root dysfunction. By restoring cellular metabolism, brain cells regain their ability to prevent plaque formation rather than just cleaning up existing damage. It’s the difference between fixing a broken immune system and just treating infections as they arise.
A Metabolic View of Brain Disease
The research suggests many neurodegenerative diseases might involve similar metabolic dysfunction. Brain cells in Parkinson’s disease, ALS, and other conditions also show signs of fat accumulation and cellular stress.
Scientists may have been thinking about these diseases all wrong. Instead of viewing them as purely neurological disorders, researchers might need to consider them metabolic diseases that happen to affect the brain.
This perspective opens new treatment possibilities. DGAT2 inhibitors are already being tested for fatty liver disease and diabetes, suggesting they might have favorable safety profiles. Rather than developing entirely new classes of drugs, researchers could potentially repurpose existing metabolic treatments for brain diseases.
The approach also offers hope for prevention. If cellular fat accumulation contributes to brain aging and disease susceptibility, maintaining healthy brain metabolism throughout life might delay or prevent neurodegenerative diseases.
Human Validation
To confirm their discoveries applied beyond mouse models, researchers analyzed brain tissue from deceased Alzheimer’s patients. The human data perfectly matched their animal studies: brain cells near protein plaques were massively engorged with fat droplets and showed elevated levels of fat-producing enzymes.
Using sophisticated 3D imaging, they could measure fat content in individual cells with unprecedented precision. The correlation was quite clear: the more fat a brain cell contained, the worse it became at clearing toxic proteins.
The team also discovered that different brain regions showed varying degrees of fat accumulation, which corresponded to areas most affected by Alzheimer’s pathology. The hippocampus, crucial for memory formation and among the first regions damaged in Alzheimer’s, showed particularly high levels of cellular fat deposits.
The Road to Treatment
While the results offer hope, researchers caution that human trials are still needed to confirm safety and effectiveness. DGAT2-targeting drugs will require extensive testing to determine optimal dosing and potential side effects in people.
However, the metabolic approach addresses several limitations of current treatments. Unlike drugs that broadly suppress immune function, targeting fat metabolism specifically restores cellular activity without compromising protective responses. The treatment also appears to work quickly, producing measurable improvements within a week in mice rather than months.
Perhaps most importantly, the discovery reframes how scientists think about brain aging and disease. Rather than viewing Alzheimer’s as an inevitable consequence of protein accumulation, the research suggests it may be a preventable metabolic disorder.
Brain cells don’t have to become fat and dysfunctional as we age. If scientists can find ways to maintain healthy cellular metabolism, they might be able to keep the brain’s natural defenses active throughout life.
The discovery that something as fundamental as cellular fat storage controls brain health offers a new paradigm for understanding and treating neurodegenerative diseases. After decades of focusing on protein plaques and tangles, the key to protecting the brain may lie in keeping its cells metabolically fit and lean.
Disclaimer: This article is for general informational purposes only and is not medical advice. Always consult a qualified healthcare provider for diagnosis or treatment of Alzheimer’s disease or any other medical condition.
Paper Summary
Methodology
Researchers isolated microglia from both mouse models of Alzheimer’s disease (5xFAD mice) and wild-type controls, along with human brain tissue from Alzheimer’s patients and healthy individuals. They used multiple reaction monitoring mass spectrometry to analyze over 1,370 lipid species and 700 metabolites. Flow cytometry measured lipid droplet content in living cells, while advanced microscopy techniques including stimulated Raman scattering provided label-free imaging of fat deposits. The team also developed a custom DGAT2 protein degrader and delivered it to mouse brains using implanted pumps.
Results
Microglia from Alzheimer’s mouse models and human patients contained significantly more lipid droplets than healthy controls, with female subjects showing particularly dramatic increases (58% more in mice). Fat accumulation correlated with proximity to amyloid plaques and impaired ability to clear amyloid-beta proteins. Exposure to amyloid-beta triggered rapid conversion of fatty acids to triacylglycerols via the DGAT2 enzyme. Inhibiting or degrading DGAT2 restored microglial cleanup function and reduced amyloid plaques by 51% in just one week of treatment.
Limitations
The study used relatively small sample sizes for human tissue analysis (6 patients per group) and short treatment durations in animal experiments. The research focused primarily on one mouse model of Alzheimer’s disease and didn’t extensively test other neurodegenerative conditions. Long-term safety and efficacy of DGAT2 targeting remain unknown, and the optimal delivery method for human treatment requires further investigation.
Funding and Disclosures
This work was supported by the US Department of Defense, NIH grants (RF1MH128866, R01NS112526, P50AA024333, R01AG081840), Eli Lilly and Company, and the Indiana Clinical and Translational Sciences Institute. Lead author Gaurav Chopra has financial interests in several companies and serves as director of the Merck-Purdue Center. No other significant conflicts of interest were reported.
Publication Information
Prakash, P., Manchanda, P., Paouri, E., et al. (2025). “Amyloid-β induces lipid droplet-mediated microglial dysfunction via the enzyme DGAT2 in Alzheimer’s disease,” published in Immunity on June 10, 2025. DOI: 10.1016/j.immuni.2025.04.029.







