
Credit: King's College London
BERLIN — Imagine looking in a mirror and seeing a version of yourself that’s almost identical, but with one crucial difference: everything is flipped. Your left hand is now your right, and vice versa. This mirror-image concept isn’t just a visual trick – it’s a fundamental principle in the world of molecules, known as chirality. And now, scientists have devised an ingenious way to detect these molecular mirror images using a revolutionary form of twisted light.
Researchers from the Max Born Institute, along with collaborators from King’s College London, Imperial College London, and Università degli Studi di Trieste, have unveiled a groundbreaking technique that could revolutionize how we develop life-saving drugs and detect early signs of disease. Their creation, dubbed the “chiral vortex,” is an entirely new structure of light that acts like a molecular detective, capable of distinguishing between nearly identical chemical twins.
At the heart of this breakthrough lies the concept of chirality, a property that gives molecules their “handedness,” much like our own left and right hands. These molecular twins, known as enantiomers, may look identical at first glance, but their subtle differences can mean the difference between a life-saving drug and a harmful substance. One version of a chiral molecule might be an effective painkiller, while its mirror image could be ineffective or even toxic. This is why drug developers need precise tools to identify and separate these molecular twins.
Until now, detecting these minute variations has been a challenge, often requiring large sample sizes and highly controlled conditions.
The new chiral vortex technique, detailed in Nature Photonics, works by creating a specially designed laser beam that twists as it travels, forming a complex 3D light pattern. When this extraordinary light interacts with chiral molecules, it produces unique patterns of emitted light – essentially, molecular fingerprints – that reveal the molecule’s handedness.
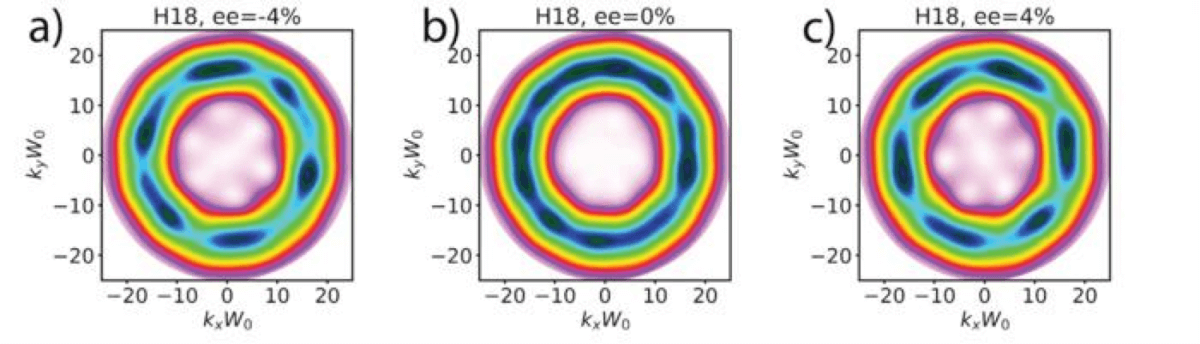
“Traditional measures of chirality have struggled to identify the concentration of right- and left-handed molecules in samples containing almost equal amounts of both. With our new method, a tiny excess in the concentration of either mirror twin can be detected, such as when the sample is 49% right-handed and 51% left-handed,” says Dr. Nicola Mayer, a postdoctoral researcher at the Max Born Institute and incoming Marie Skłodowska-Curie Actions Research Fellow at King’s College London, in a media release.
In the pharmaceutical industry, where the precise identification of molecular structures can make or break a new drug, chiral topological light offers a level of sensitivity that rivals and sometimes surpasses current gold-standard techniques. Environmental scientists could use this technology to detect trace amounts of pollutants with unprecedented accuracy, while chemists might unlock new insights into complex biochemical reactions.
What sets chiral topological light apart is not just its sensitivity, but its resilience. The topological properties embedded in the light beam ensure that the detection method remains stable even under less-than-ideal conditions — a crucial factor for real-world applications where perfect laboratory settings are a luxury.
“These signals can also provide a snapshot into how electrons move inside molecules at their natural speed, this understanding can lay the groundwork for shaping the behavior of electrons and even eventually influencing chemical reactions with light,” adds Dr. Mayer.
While the current study is based on theoretical models and computer simulations, it lays a solid foundation for future experimental work. The next step will be to build actual laser systems capable of producing this twisted “chiral vortex” light and test them with real molecular samples.
If successful, this technique could find applications across a wide range of fields. In drug development, it could ensure the purity and safety of medicines by accurately separating beneficial molecular versions from harmful ones. In medical diagnostics, it might enable earlier detection of diseases by spotting subtle molecular imbalances in the body. Even in basic scientific research, it could provide new ways to study how chirality emerges in chemical reactions or biological systems.
As we look to the future, this twisted tale of light and molecules reminds us that sometimes, the most illuminating discoveries come from seeing the world – and the molecules within it – in an entirely new light.
Paper Summary
Methodology
The researchers used a special type of light, called chiral topological light, which carries unique twisting properties that can interact differently with molecules based on their shape (left-handed or right-handed). They created this light by combining two light beams with opposite circular polarization—one spinning clockwise and the other counterclockwise. The light beams were focused in such a way that they generated a twisting motion in the electric field, forming patterns known as chiral vortices.
These vortices interact with the molecules, enabling the researchers to detect very small differences in the shape (chirality) of molecules. Using complex simulations and mathematical models, they observed how the light beams altered molecular responses, creating a highly sensitive method for detecting even tiny amounts of one type of molecule in a mixture.
Key Results
The researchers found that chiral topological light could detect small differences between left- and right-handed molecules, even when mixed together in very small proportions. The unique light created distinct patterns that could be seen when the molecules interacted with it. These patterns changed based on the amount of each type of molecule present.
The method they developed was able to detect these differences with extreme precision, making it possible to identify tiny concentrations of left- or right-handed molecules that other techniques might miss. They were even able to spot differences with just a small percentage difference in the mixture, showing how effective this method could be.
Study Limitations
First, the setup requires highly controlled light beams, and even small imperfections in the polarization or intensity of the light could affect the results. Additionally, the technique relies on sophisticated equipment that may not be readily available in all laboratories, making it challenging to implement in every research setting.
While the method works well for detecting small amounts of left- and right-handed molecules in specific controlled environments, it might be less effective in real-world scenarios where samples are more complex or contaminated. Finally, the simulations they used require high levels of computational power, which could limit its accessibility for some researchers.
Discussion & Takeaways
The findings from this study open new possibilities for detecting molecular chirality with unprecedented precision. Chiral topological light provides a robust way to observe differences between left- and right-handed molecules, which is crucial in fields like pharmaceuticals, where the shape of molecules can determine their effects.
This method offers a new tool for enantiosensitive spectroscopy, making it easier to detect small variations that could have been overlooked by traditional methods. By embedding topological properties into light, the researchers have demonstrated a novel way to enhance the accuracy and reliability of molecular detection. However, the implementation of this technique in everyday applications will require further development and overcoming current technical challenges.
Funding & Disclosures
This research received financial support from several prominent institutions. Notably, it was funded by the European Union Horizon 2020 program (Grant no. 899794) and the European Research Council (ERC, ULISSES, Grant no. 101054696). Additional support came from the Royal Society under various grant numbers. There were no competing financial interests declared by the authors, ensuring the objectivity and integrity of the study. The authors also acknowledged the Max-Born Institute for Nonlinear Optics and Short Pulse Spectroscopy for providing open access funding, ensuring broad dissemination of their work.







