(© egokhan - stock.adobe.com)
Quieting The Body’s Stress Alarm May Be The Secret To Longevity, Study Suggests
In A Nutshell
- Suppressing a key cellular stress protein called ATF4 significantly extended lifespan in fruit flies, while ramping it up shortened their lives.
- For years, the prevailing assumption was that activating this stress pathway was good for longevity, based on studies in yeast and roundworms. This study found the opposite in a more complex organism.
- A drug known to activate the same pathway, borrelidin, also shortened fly lifespan, while eliminating gut bacteria had no effect, pointing to the stress pathway as the driver.
- Results held across three independent experiments and two dietary conditions, but whether any of this applies to mammals or humans remains an open question.
For years, a popular idea has taken hold in aging research. The general notion is that revving up the body’s built-in stress defenses helps organisms live longer. If a little stress makes cells tougher, keeping those protective shields permanently raised should be a good thing. A new study in fruit flies has flipped that assumption, finding that silencing a major stress response pathway, rather than activating it, is what actually extends lifespan.
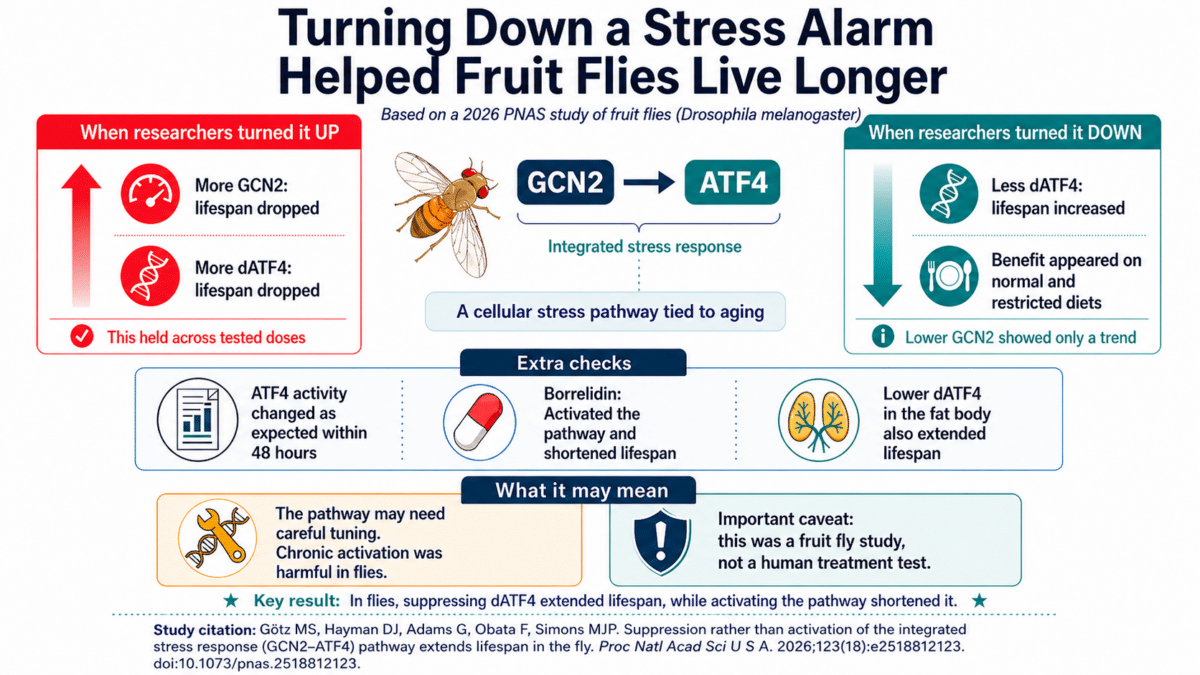
Published in the Proceedings of the National Academy of Sciences, the research is the first thorough test of a specific stress-signaling chain, known as the GCN2-ATF4 pathway, in fruit flies, an organism more biologically complex than the yeast and tiny roundworms where earlier work was done. GCN2 is a sensor protein that detects cellular stress, while ATF4 is the downstream protein that drives the stress response. Flies engineered to produce more ATF4 died significantly sooner, while flies engineered to produce less lived significantly longer. The results challenge a leading idea, built largely on studies in simpler organisms, that switching on this pathway promotes a longer life.
The authors write that their study “challenges the prevailing hypothesis that ATF4 activation is broadly beneficial for lifespan.” In the abstract, they add that ATF4 may have a “dual, dose-, and context-dependent role in aging.” How hard scientists push this alarm, and in which tissue, may matter as much as whether they push it at all.
Fruit Fly Stress Pathway Linked to Shorter Lifespan
Led by researchers at the University of Sheffield in the United Kingdom, the team used a genetic tool allowing them to flip specific genes on or off in adult flies at will, rather than altering them from birth. This approach isolates effects to adult life, the period most relevant to aging, and controls for the subtle genetic differences that can muddy lifespan studies.
Scientists targeted two genes: GCN2, the upstream sensor, and dATF4, the downstream workhorse. They tested what happened when each was cranked up or dialed down across the flies’ entire bodies, under both a normal diet and a restricted diet, since calorie restriction has long been linked to longer life and stress pathways have been suspected of playing a role in those benefits.
Sample sizes were large, with most experimental groups including several hundred flies and the largest exceeding 600 individuals. Cranking up GCN2 across the whole body drastically reduced lifespan. Cranking up dATF4 similarly shortened it. Dialing down GCN2 showed a trend toward longer life that fell just short of statistical significance. Dialing down dATF4 produced the headline result: a significant extension of lifespan that held regardless of diet.
How Scientists Confirmed the GCN2-ATF4 Lifespan Results
Because the findings ran counter to established thinking, the team went to unusual lengths to verify them. A glowing molecular tag inside the flies lights up when ATF4 is active. Within 48 hours of flipping the genetic switch, the tag confirmed that activity was shifting in the expected directions.
Researchers also tested whether different doses might reveal a sweet spot where activating the pathway could help rather than harm. Using three concentrations of the drug that triggers the genetic switch, they found that higher doses produced stronger effects. At every dose tested, however, activation still shortened lifespan.
To explore tissue specificity, they ran a separate experiment targeting dATF4 only in the fly’s fat body, an organ that works somewhat like a combination of the human liver and fat tissue. Cranking up dATF4 only there had no significant effect on lifespan. Dialing it down in just the fat body still extended lifespan, mirroring the whole-body results.
Stress Drug That Extended Life in Simpler Organisms Had the Opposite Effect in Flies
Researchers also tested a drug called borrelidin, which activates the same stress pathway by mimicking amino acid starvation inside cells. Previous studies in yeast and roundworms suggested borrelidin could extend lifespan. In flies, it significantly reduced lifespan, consistent with the finding that activating this pathway is harmful in this organism.
Borrelidin had almost no additional negative effect on flies already overproducing ATF4, suggesting those flies had perhaps already reached the limit of the damage the pathway could do. In flies where dATF4 had been dialed down, borrelidin removed the lifespan-extending benefit, suggesting the drug was pushing the pathway in a direction that countered the genetic manipulation.
To rule out borrelidin’s antibiotic properties as the cause, the team treated flies with a broad cocktail of antibiotics to eliminate gut microbes entirely. Removing those microbes had no significant effect on lifespan, supporting the idea that the observed changes were driven by the stress pathway, not by disrupted gut bacteria.
Using a technique that reads the full genetic messages produced by cells, researchers found that cranking up dATF4 was associated with suppressed energy production and DNA repair, while detoxification pathways ramped up, a profile resembling a cell in emergency mode. Dialing it down was associated with gene-activity patterns linked to protein quality control and DNA repair, pointing toward a shift into cellular maintenance.
ATF4 appears to be an important switch in aging biology, at least in flies. Whether dialing down this alarm in human tissues could one day slow aging remains an open question requiring extensive testing in mammals. But the simple story that activating stress defenses is always a recipe for longevity now has a conspicuous crack in it.
Disclaimer: The findings described in this article are based on a study conducted in fruit flies and have not been tested in humans or other mammals. The results should not be interpreted as medical advice or as evidence that any current treatment can extend human lifespan.
Paper Notes
Limitations
The study was conducted entirely in fruit flies, and the authors acknowledge that results may not directly translate to mammals or humans. All overexpression doses tested still shortened lifespan, so the researchers could not rule out the possibility that an even lower dose or more precisely targeted tissue manipulation might produce lifespan-extending effects from ATF4 activation. The gene activity analysis is correlative rather than causative: it suggests possible mechanisms but does not prove them. The overlap of genes significantly changed in both overexpression and knockdown conditions was not larger than expected by chance, though several known ATF4 target genes did change in the predicted directions. Borrelidin experiments were conducted at a single dose, and the authors note they cannot exclude that a lower dose or shorter exposure could extend lifespan in flies. Diet-by-genotype interactions on lifespan were not consistently observed, limiting conclusions about the relationship between dietary restriction and ATF4 in this system.
Funding and Disclosures
Mirre J. P. Simons is a Sir Henry Dale fellow supported by the Wellcome Trust and Royal Society (216405/Z/19/Z). Dan J. Hayman is a Vivensa Foundation ECR Fellow (ECRF24\13). A Royal Society ISPF International Collaboration award supported collaboration between Simons and Fumiaki Obata (ICA\R2\242197). Fly stocks were obtained from the Bloomington Drosophila Stock Center (NIH P40OD018537). Research was funded in whole, or in part, by the Wellcome Trust (216405/Z/19/Z). The authors declared no competing interest.
Publication Details
Title: Suppression rather than activation of the integrated stress response (GCN2-ATF4) pathway extends lifespan in the fly | Authors: Miriam S. Götz, Dan J. Hayman, Gracie Adams, Fumiaki Obata, and Mirre J. P. Simons | Affiliations: School of Biosciences, University of Sheffield, Sheffield, United Kingdom; RIKEN Center for Biosystems Dynamics Research, Kobe, Japan; Laboratory of Molecular Cell Biology and Development, Graduate School of Biostudies, Kyoto University, Kyoto, Japan | Journal: Proceedings of the National Academy of Sciences of the United States of America (PNAS), Volume 123, Number 18 | DOI: 10.1073/pnas.2518812123 | Published: April 28, 2026 | Corresponding Author: Mirre J. P. Simons ([email protected])







