
Nagoya University Hospital Center for Maternal-Neonatal Care (Neonatal Intensive Care Unit / Growing Care Unit) Bright lighting and colorful art create a child-friendly atmosphere for the neonatal care unit. (Credit: Yoshiaki Sato)
Protein found in stem cells could fuel the brain’s ability to generate neurons, leading to improved movement, memory, and learning.
In A Nutshell
- Stem cells from baby teeth improved movement, memory, and learning in rats with cerebral palsy, even after symptoms had already developed.
- The cells travel to the brain after a simple vein injection and appear to kick-start the growth of new neurons in regions responsible for motor control and memory.
- A specific protein secreted by baby tooth stem cells, called HGF, was identified as the key driver of the brain’s renewed ability to generate neurons.
- No approved treatment currently exists to reverse cerebral palsy after symptoms set in, making this line of research particularly worth watching.
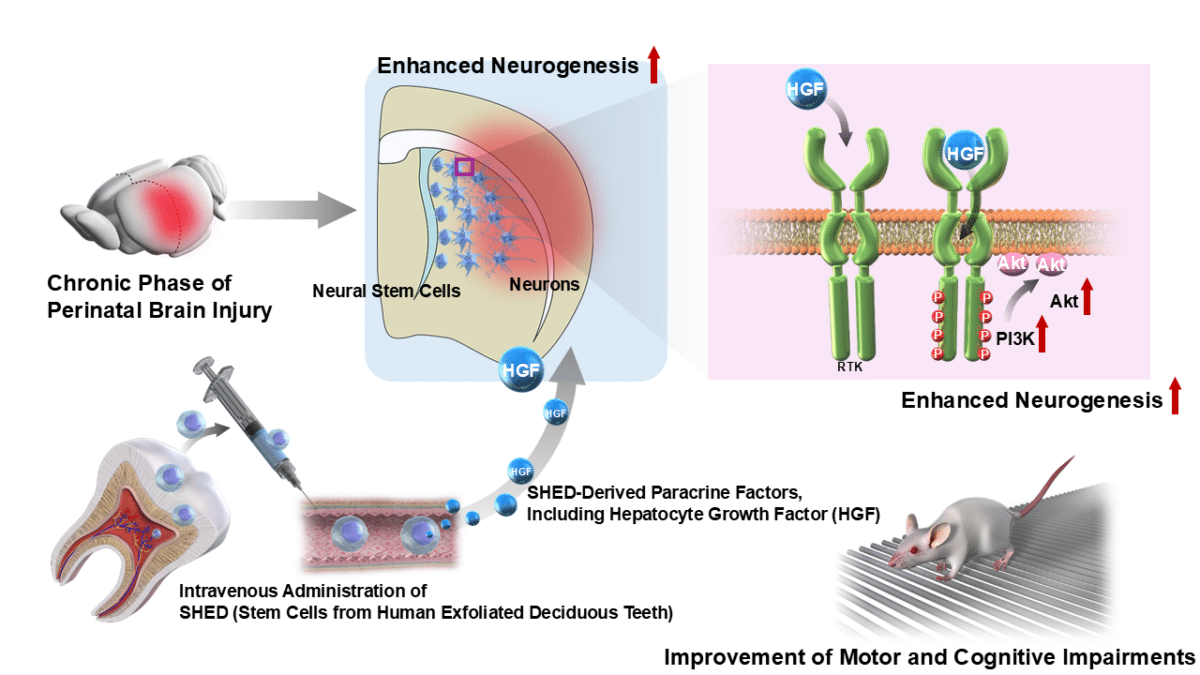
Most parents tuck their child’s lost baby teeth away as keepsakes, maybe leaving them for the tooth fairy. A new study out of Japan suggests those tiny teeth may carry medical value far beyond nostalgia. Stem cells harvested from the dental pulp of baby teeth appear capable of improving movement, memory, and learning in animals with cerebral palsy, even when treatment begins well after neurological symptoms have already set in.
Cerebral palsy affects an estimated 2 to 3 children per 1,000 live births worldwide. It most often stems from a brain injury called hypoxic-ischemic encephalopathy, or HIE, which occurs when a newborn’s brain is deprived of oxygen around the time of birth. Doctors currently rely on therapeutic hypothermia, which cools the body shortly after birth to limit initial damage, as the standard treatment for moderate to severe HIE. Once neurological symptoms settle in, nothing has been proven to reverse them.
Researchers at Nagoya University Hospital set out to test whether stem cells from human exfoliated deciduous teeth, abbreviated as SHED, could change that. Their findings, published in the journal Stem Cell Research & Therapy, are encouraging, and go further than prior stem cell studies by pinpointing the biological mechanism behind the improvements.
Why Baby Teeth Stem Cells Stand Out
Baby teeth are the first set of teeth children grow and eventually lose. Inside each tooth sits a soft core of tissue called dental pulp, and buried within that pulp are stem cells. Researchers first identified SHED in the early 2000s and have studied them as candidates for regenerative therapies ever since.
What sets them apart is a combination of practical and biological advantages. Baby teeth are normally thrown away, raising few ethical concerns about collection. On the biological side, SHED produce unusually high levels of a protein called hepatocyte growth factor, or HGF, compared with bone marrow stem cells and other commonly studied types. HGF acts on the brain’s own repair cells, called neural stem cells, pushing them to multiply and develop into new neurons.
In laboratory dishes, neural stem cells placed near SHED divided at significantly higher rates than those grown alongside bone marrow stem cells or skin cells. When researchers blocked HGF with antibodies, that effect disappeared. When they engineered SHED with the HGF gene knocked out, neural stem cell growth dropped sharply, and adding HGF back restored it. That chain of evidence identifies HGF as the primary driver of the connection between baby tooth stem cells and brain repair.
Yoshiaki Sato)
Testing Baby Teeth Stem Cells in a Cerebral Palsy Model
To create a rat model of cerebral palsy, researchers restricted blood flow to one side of newborn rats’ brains while exposing them to low oxygen for an hour, mimicking the type of birth injury that leads to CP in humans. Rats that developed motor impairments were then confirmed using a ladder-walking test at four weeks of age, where each animal’s gait was scored as it crossed a ladder with rungs spaced at varying intervals.
Rats with confirmed impairments were split into two groups. One received three intravenous SHED injections at ages five, seven, and nine weeks, a window roughly corresponding to the preadolescent stage in humans. A control group received the carrier solution alone. At four months, SHED-treated rats scored significantly better on the ladder-walking test. A second test, which measured how much each rat favored its unaffected limb, showed less asymmetry in the SHED group, a sign of improved coordination. A memory and learning test, where rats were trained to move to the other side of a chamber to avoid a mild shock, showed that SHED-treated animals learned the task more effectively and retained it better across repeated sessions.
Tracking Baby Teeth Stem Cells Into the Brain
A central question for this type of therapy is whether the stem cells actually travel to the brain after being injected into a vein. To find out, researchers tagged SHED with quantum dots, tiny fluorescent particles that glow under imaging equipment, then tracked them after injection.
Within 24 to 48 hours, fluorescence increased in the brains of treated rats but not in controls. Three-dimensional imaging confirmed the cells had reached the injured cortex. Their presence was brief, peaking around 24 to 48 hours and fading within a week, yet the behavioral benefits lasted long after the cells had cleared. Something set in motion during that short window kept working.
How Baby Teeth Stem Cells Appear to Rebuild the Brain
Brain tissue collected two weeks after the final SHED injection contained more newly formed, immature neurons in the hippocampus and in the subventricular zone, two of the brain’s main regions where new neurons can form. Ten weeks after treatment, the increase in mature neurons was still measurable in both the hippocampus and cortex.
To check whether that growth reflected actual new neuron production or simply fewer neurons dying off, the team stained brain tissue for a marker of dying cells. No significant difference appeared between groups. New growth, not reduced cell death, explained the gain.
Protein analysis of the treated brain tissue pointed to neurogenesis, the brain’s process of making new neurons, as the most strongly affected biological change following SHED treatment. In plain terms, the baby tooth stem cells appear to kick-start the brain’s ability to generate new neurons, a process that normally winds down within weeks of the original injury, and to sustain it well into the chronic phase of the disease.
Cerebral palsy has no cure, and earlier clinical trials using other stem cell sources showed some promise but lacked clear explanations for why. “SHED is a promising candidate for postsymptom-onset treatment of cerebral palsy,” the authors wrote, adding that “further studies to confirm these findings and examine the clinical utility of SHED are warranted.” For families who have long been told that birth-related brain damage cannot be undone, a therapy sourced from something as ordinary as a lost baby tooth is a direction worth following closely.
Disclaimer: This article is based on preclinical research conducted in animals. The findings have not yet been tested in humans, and the treatment described is not currently available as a clinical therapy. Consult a qualified medical professional for guidance on cerebral palsy treatment options.
Paper Notes
Limitations
This study was conducted entirely in rats, and human immune systems may respond differently to SHED than rat immune systems did. Differences in brain architecture between rodents and humans mean the mechanisms observed here may not translate directly to human patients. Group sizes were relatively small and unequal, largely because some animals died during the injury model procedure and others failed to develop motor impairment. HGF, while beneficial for neural repair in this context, also has known cancer-promoting properties, so any clinical application would require thorough safety evaluation. Optimal dosing for human use has not been established. SHED can only be collected during a specific window of childhood, and clinical-scale preparations from multiple donors would introduce variability in cell quality that would need to be managed carefully.
Funding and Disclosures
This work was supported by JSPS KAKENHI Grants-in-Aid for Scientific Research (grant numbers 23K07248, 23K25192, 24K21339), Nagoya University Hospital Funding for Clinical Research, the JSPS MEXT Quantum Leap Flagship Program (JPMXS0120330644), the SEI Group CSR Foundation, the BRIDGE program, QST, and Kidswell Bio Corporation. Several authors disclosed conflicts of interest. Lead author Takahiro Kanzawa, corresponding author Yoshiaki Sato, and co-author Shinobu Shimizu have a collaborative research agreement with Kidswell Bio Corporation, which supplied SHED free of charge. Co-author Yasuyuki Mitani is a board member and holds stock options in Kidswell Bio Corporation. Kanzawa, Shimizu, Hiroshi Yukawa, Mitani, and Sato have filed a patent for the application of SHED in the treatment and prevention of cerebral palsy. The authors also disclosed use of AI for English proofreading in the manuscript.
Publication Details
“Novel stem cell therapy for cerebral palsy using stem cells from human exfoliated deciduous teeth” was authored by Takahiro Kanzawa, Atsuto Onoda, Azusa Okamoto, Xu Yue, Ryoko Shimode, Yukina Takamoto, Sakiko Suzuki, Kazuto Ueda, Ryosuke Miura, Toshihiko Suzuki, Naoki Tajiri, Shinobu Shimizu, Saho Morita, Hiroshi Yukawa, Hiroshi Kohara, Noritaka Fukuda, Yasuyuki Mitani, Hideki Hida, Yoshiyuki Takahashi, and Yoshiaki Sato. Corresponding author: Yoshiaki Sato, Division of Neonatology, Center for Maternal-Neonatal Care, Nagoya University Hospital, Nagoya, Japan ([email protected]). Published January 23, 2026 in Stem Cell Research & Therapy, volume 17, article number 44. DOI: 10.1186/s13287-025-04828-y. Open access under Creative Commons Attribution 4.0 International License.







