
(© Prostock-studio - stock.adobe.com)
Electrical pulses may represent a new way to treat IBD without any medications.
In A Nutshell
- Soft wireless implant wrapping around the splenic nerve reduced gut inflammation in rats over seven weeks without medications
- Device uses gel-based electrodes powered wirelessly through the skin; no battery, no rigid parts, no daily pills required
- Treatment triggered norepinephrine release that rebalanced immune cells specifically in intestinal tissue
- Human trials are years away due to nerve size differences, surgical challenges, and need for long-term safety data beyond seven weeks
Scientists testing a potential new treatment for inflammatory bowel disease have built a soft wireless implant that wraps around a nerve connected to the spleen. In rats with severe intestinal inflammation, the device used electrical pulses to restore healthier gut tissue over seven weeks—without any medications.
The gel-based device gets power wirelessly through the skin, similar to how some phones charge on a pad. Rats that received daily 20-minute treatment sessions showed less weight loss, healthier colons, and reduced inflammation compared to untreated animals with the same condition. The device is battery-free and built around soft hydrogel-based electrodes.
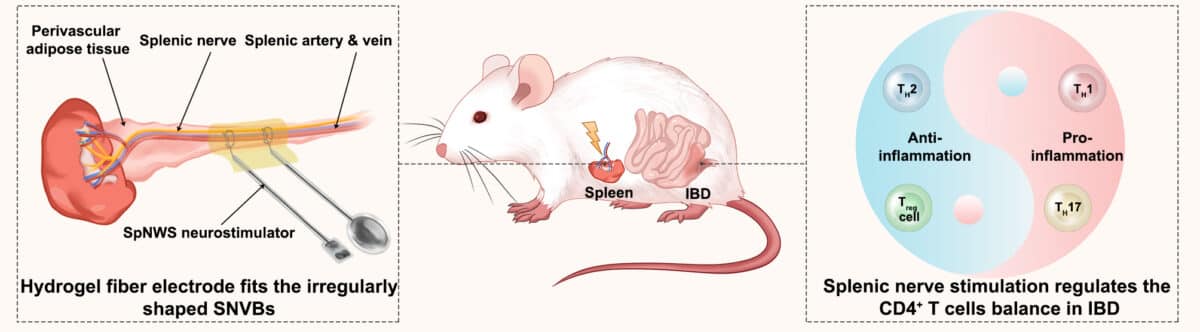
Researchers at Huazhong University of Science and Technology in China designed the device to stimulate the splenic nerve, which triggers norepinephrine release that can dial down inflammation. Unlike nerve stimulators used clinically that activate large bundles controlling multiple body functions, this approach targets a specific anti-inflammatory pathway.
Why Zap a Nerve?
The implant taps into a recently discovered connection between nerves and immune responses. When electricity stimulates the splenic nerve, it releases norepinephrine into the spleen, a major immune organ. This signal triggers changes that reduce inflammatory molecule production in the gut.
Nerve stimulators used clinically for other conditions activate the vagus nerve, a large bundle with around 100,000 different fibers controlling everything from heartbeat to voice. Stimulating all those fibers at once causes side effects like hoarseness and coughing. The splenic nerve pathway is more selective.
The wireless power system works like a contactless phone charger. A thin gel pad sits under the skin in the abdomen. When an external patch on the skin surface sends electrical signals, the implant receives them and relays pulses to the nerve. Rats needed twenty minutes of daily stimulation to see benefits.
Soft Enough to Live Inside the Body
Traditional metal electrodes can be much stiffer than soft tissue. That stiffness causes problems when organs move during breathing and digestion. The gel material in this device flexes and moves with the body.
The fibers wrap and knot around the nerve bundle and stick in place using a biological adhesive. After five weeks inside rats, thin tissue capsules formed around some components, but the material remained functional without causing significant damage.
What Happened in the Experiments
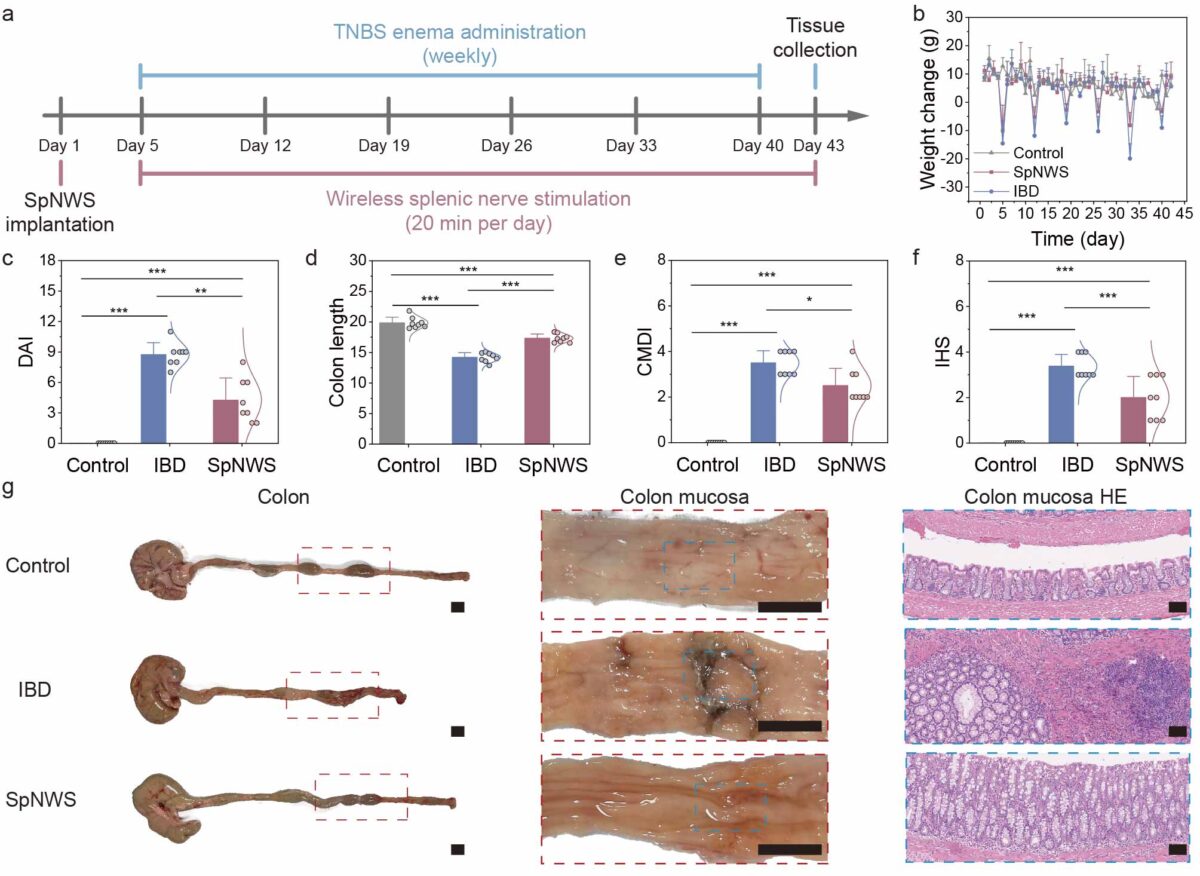
Researchers gave rats a chemical treatment that caused chronic intestinal inflammation. Some rats received the implant five days before the first TNBS enema, with daily electrical stimulation starting after that first enema. Others got no stimulation.
Treated rats maintained their weight better and had longer, healthier colons. When scientists examined intestinal tissue under microscopes, they saw less damage in the stimulation group.
Lab tests revealed changes in immune cell types within gut tissue. The treatment reduced certain inflammatory immune cells while increasing others that suppress inflammation. These changes, documented and published in National Science Review, occurred specifically in the intestines.
Getting From Rats to People
Human trials remain years away. The splenic nerve’s small diameter and anatomical complexity make stable interfacing challenging, and translating the approach to humans would require additional engineering and testing.
The experiments used chemicals to create bowel inflammation in rats. Human inflammatory bowel disease involves different immune processes that might respond differently. Seven weeks of testing is far shorter than the years of durability required for human devices.
Still, for people living with Crohn’s disease or ulcerative colitis who face limited treatment options, a drug-free approach could eventually prove valuable. Current medications either broadly suppress immune function or target specific inflammatory molecules but often lose effectiveness over time.
Disclaimer: This study was conducted exclusively in rats using chemically induced bowel inflammation. The device has not been tested in humans, and human inflammatory bowel disease involves different immune mechanisms. Clinical translation would require additional engineering, long-term safety testing, and regulatory approval before any potential human use.
Paper Notes
Limitations
This research used only rats with chemically induced bowel inflammation, not the autoimmune form of IBD affecting humans. The seven-week study duration is too short to assess long-term device durability or safety. Researchers could not track individual animals over time since all measurements required dissection. The treatment requires daily use of external equipment. Only eight animals were tested in each group. The study did not assess whether the treatment affected immune responses to infections or pathogens.
Funding and Disclosures
This work received support from the National Natural Science Foundation of China (32471387), the Key Research and Development Program of Hubei Province (2024BCB002), the Leading Innovative and Entrepreneur Team Introduction Program of Zhejiang (2023R01002), and the Distinguished Young Scientists Fund of Zhejiang (LR25H250001). The authors declared no competing financial interests.
Publication Details
Authors: Wenliang Liu, Qiong Wang, Renyuan Sun, Ming Yang, Ping Wu, Dingke Zhang, Kun Yang, Chong Ma, Chuan Gao, Nanxi Yi, Zhikun Li, Long Wen, Luyao Wu, Xiaokun Li, Jiexiong Feng, Zhouguang Wang, and Zhiqiang Luo
Affiliations: National Engineering Research Center for Nanomedicine, Research Center for Intelligent Fiber Devices and Equipment, State Key Laboratory of New Textile Materials and Advanced Processing, College of Life Science and Technology, Huazhong University of Science and Technology, Wuhan, China; Department of Pediatric Surgery, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China; The First Affiliated Hospital of Wenzhou Medical University, Wenzhou, China; Hubei Clinical Center of Hirschsprung’s disease and allied disorders, Wuhan, China
Journal: National Science Review (advance online publication) | DOI: 10.1093/nsr/nwaf557 | Publication Date: Published online January 5, 2026







