
(Image by SewCreamStudio / Shutterstock)
In A Nutshell
- Scientists have agreed on the first widely adopted definition of gut health: normal digestive function without active disease or symptoms that affect quality of life.
- Having a gut disease like Crohn’s or celiac does not automatically mean poor gut health; remission counts.
- At-home microbiome tests cannot yet reliably diagnose disease or predict health risk in individual patients.
- Chronic, recurring symptoms that disrupt daily life are the threshold for concern, not the occasional stomach ache.
“Gut health” has been plastered across probiotic labels, wellness blogs, and supplement ads for years, used to sell everything from yogurt to fermented teas. It has become one of the most frequently invoked phrases in health marketing. There’s just one problem. Until now, there has been no widely adopted consensus definition of what gut health actually means.
A consensus statement published in Nature Reviews Gastroenterology & Hepatology changes that. An international panel of 13 leading experts worked toward what is likely the most authoritative definition of gut health ever produced: “a state of normal gastrointestinal function without active gastrointestinal disease and gut-related symptoms that affect quality of life.”
Simple enough, but the work behind that sentence carries real consequences for how people understand their own bodies. It draws a sharp line between having a digestive disease and experiencing poor gut health. Someone with celiac disease or inflammatory bowel disease can still achieve gut health during periods of complete remission. The reverse is also true: poor gut health can exist even when there is no diagnosable disease and no obvious abnormalities in lab tests.
Surveys cited in the paper put the stakes in perspective. A study of more than 53,000 Danish blood donors found that 68% reported at least one gastrointestinal symptom in the previous four weeks. Bloating, abdominal pain, constipation, and heartburn are so common that most people treat them as a normal part of life. Whether those symptoms signal something worth addressing, or just the routine noise of a working digestive system, has long been unclear. Now there is a framework for answering that question.
How Experts Arrived at a Definition of Gut Health
Convened by the International Scientific Association for Probiotics and Prebiotics (ISAPP), a nonprofit academic organization, the panel gathered in London for a full-day workshop after preparing written perspectives from their individual fields. Panelists came from institutions across the United States, Europe, and Australia, representing gastroenterology, microbiology, immunology, endocrinology, family medicine, pediatrics, and nutrition. The group refined the definition across two additional online meetings, and all 13 panelists approved the final wording in an online vote.
Before this consensus, different researchers measured gut health in entirely different ways. One study might define it by changes in the gut microbiome, another by a reduction in constipation. That inconsistency made it nearly impossible to compare findings or build reliable clinical recommendations.
What the Science Says About Gut Health and Your Microbiome
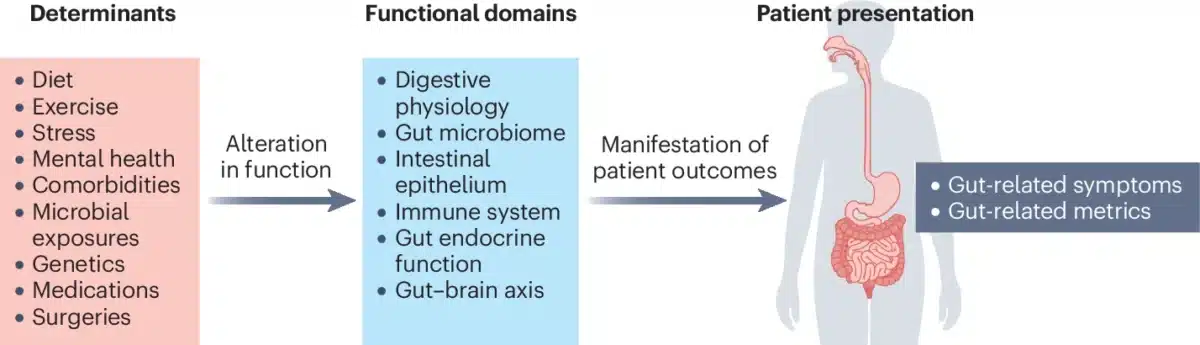
The panel identified six functional areas that together make up gut health: digestive physiology, the gut microbiome, the intestinal barrier, the immune system, gut hormone function, and the gut-brain connection. Each one is distinct, but they operate as an integrated system. Disrupt one, and the others often follow.
Digestive physiology is the most intuitive piece. It covers how well the body breaks down food, absorbs nutrients, and moves waste through the system. The gut secretes roughly seven liters of fluid daily to keep that process running. Disruptions in muscle movement or fluid secretion are among the most common drivers of bloating, nausea, and constipation.
The gut microbiome, the vast community of bacteria and other microorganisms living in the digestive tract, has attracted enormous scientific attention over the past two decades. The panel was candid about the limits of current knowledge, though. Despite an explosion of consumer-facing microbiome tests, “At present, the analysis of an individual gut microbiome cannot provide a meaningful index of health or a prediction of disease presence or risk,” the paper states. Microbiome testing has real scientific promise, but it has not yet reached the point where results can be reliably used to diagnose disease or assess individual risk.
The gut-brain connection is one of the more surprising pieces of the puzzle. Stress triggers gut symptoms. Early life stress shapes gut health in adulthood. Anxiety and depression are commonly tied to gastrointestinal symptoms. Communication between the gut and brain runs in both directions, and disruptions in that relationship can amplify pain, alter mood, and affect digestion at the same time.
The paper also addresses “leaky gut,” one of wellness culture’s most loaded terms. As the authors put it, “All guts are ‘leaky’; it is only when the selective permeability (or ‘leakiness’) that characterizes the normal epithelium is compromised that it can potentially lead to disease.” For many conditions that “leaky gut” has been blamed for, “evidence is scarce or non-existent.”
When Gut Health Symptoms Are Actually Worth Worrying About
Not every stomach ache signals trouble. One of the most practical contributions of the paper is its guidance on distinguishing ordinary gut symptoms from those that indicate a real problem.
The key question is whether symptoms are frequent, persistent, and disruptive to daily life. Occasional heartburn, a bout of constipation while traveling, or bloating after a heavy meal fall within the range of normal. Symptoms that are chronic, recurring, and interfere with quality of life are the ones that warrant medical attention.
Diet is among the most actionable levers. Higher fiber intake has been associated with better bowel regularity, lower colon cancer risk, and a more diverse microbiome. Mediterranean and plant-based diets are associated with lower inflammation markers in the blood. High-fat meals tend to worsen symptoms in people who already struggle with recurring stomach discomfort or irritable bowel syndrome. Individual responses vary widely, though, meaning no single eating pattern works for everyone.
Regular physical activity is linked to a more diverse microbiome, better outcomes for constipation and irritable bowel syndrome, and faster digestion. Smoking, by contrast, is tied to microbiome shifts that raise the risk of gastrointestinal disease.
Most products that claim to support gut health are backed by loosely defined science, and the claims vary in quality. What this consensus statement offers everyday people is a clearer filter: a grounded, expert-tested definition that makes it easier to separate what the research actually supports from what is simply good marketing.
Disclaimer: This article is based on a peer-reviewed consensus statement and is intended for general informational purposes only. It does not constitute medical advice. If you have concerns about your digestive health, consult a qualified healthcare professional.
Paper Notes
Limitations
The consensus panel used a semi-structured discussion format rather than a more rigorous methodology like the Delphi process, which uses anonymous, iterative rounds of expert input. The authors acknowledge that their approach may have reduced anonymity and potentially encouraged conformity among panelists. The group of 13 scientists, while drawn from multiple continents and disciplines, did not represent all geographies or professional perspectives that might affect conclusions. The panel also notes that risk factors for future gut health are not yet well enough understood to be incorporated into the definition, and that many proposed biomarkers still require long-term validation before they can be used reliably in clinical practice.
Funding and Disclosures
ISAPP, a nonprofit academic organization, funded travel support for panelists to attend the initial meeting. Several authors disclosed consulting relationships, advisory board memberships, or research funding from food, supplement, and pharmaceutical companies, including Danone, Nestle, Fonterra, and Janssen, among others. The paper states that industry funding did not direct the organization’s activities or constrain the review’s content. Two authors, Stephan Bischoff and Marlies Meisel, declared no competing interests.
Publication Details
Authors: Maria L. Marco, Marla Cunningham, Stephan C. Bischoff, Gerard Clarke, Nathalie Delzenne, James D. Lewis, Marlies Meisel, Daniel Merenstein, Paul W. O’Toole, Heidi M. Staudacher, Hania Szajewska, Jerry M. Wells, and Eamonn M. M. Quigley | Journal: Nature Reviews Gastroenterology & Hepatology | Title: “The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of gut health” | Published: February 18, 2026 | DOI: https://doi.org/10.1038/s41575-026-01176-x







