
Credit: maradon 333 on Shutterstock
In A Nutshell
- Researchers developed a light-activated method that breaks down a model “forever chemical” at the molecular level.
- Coating the catalyst with a specialized porous plastic nearly tripled its effectiveness under neutral water conditions.
- The coated catalyst remained effective across six reuse cycles, suggesting it could work in a practical treatment system.
- The catalyst responds to visible light, raising the possibility that future versions could run on sunlight.
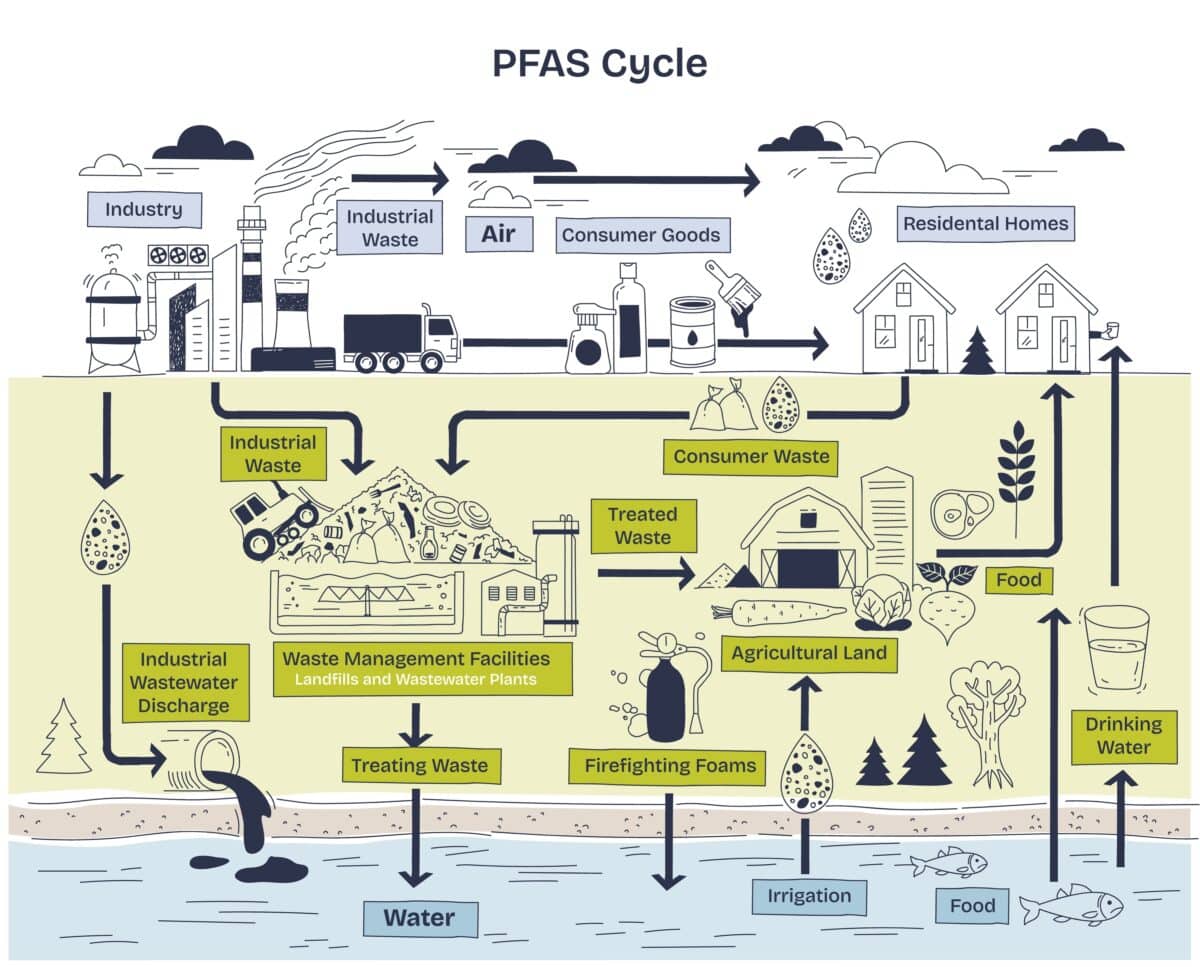
Somewhere in your body right now, there are almost certainly traces of PFAS. These man-made chemicals, used for decades in nonstick cookware, food packaging, and firefighting foam, have spread so thoroughly through the environment that researchers find them in arctic ice, in rainwater, and in the blood of people who have never worked anywhere near an industrial site. They earned the nickname “forever chemicals” because almost nothing in nature breaks them down. The question scientists have wrestled with for years is not whether PFAS are a problem, but how to actually get rid of them.
Researchers may have finally found a meaningful piece of the answer, and it involves light.
In a study published in the journal RSC Advances, researchers described a way to use a light-activated catalyst coated with a specialized porous plastic to break a model PFAS compound apart at the molecular level. Under neutral conditions, the combination worked nearly three times as effectively as the catalyst alone, and it did so under conditions far closer to what real water treatment systems actually operate in. That last part is what makes the result worth paying attention to.
Why “Forever Chemicals” Have Been So Hard to Kill
PFAS are built around one of the strongest chemical bonds in existence, the link between carbon and fluorine atoms. That bond is essentially what makes PFAS useful in the first place: it resists heat, water, oil, and almost everything else. It also resists the standard toolkit of water treatment. Many current approaches focus on removing PFAS from water rather than fully destroying them, filtering them out and concentrating them somewhere else, kicking the problem down the road rather than solving it.
Light-driven chemistry offers a different approach. Certain materials, when exposed to the right wavelengths of light, can generate enough energy to tear apart even stubborn chemical bonds. Graphitic carbon nitride, a yellow powdery catalyst that responds to visible light, has shown some ability to do this with PFAS. The catch is that in water, PFAS molecules do not stick around the catalyst surface long enough for the reaction to gain traction. The chemicals drift by, interact briefly, and move on before much damage is done.
That is the problem PIM-1 was brought in to solve.
The Porous Plastic That Changes Everything
PIM-1 is a polymer honeycombed with pores roughly one nanometer wide. Those pores are hydrophobic, which helps draw in water-repelling PFAS molecules. Since PFAS molecules share that same water-repelling quality, they migrate naturally into PIM-1’s structure, where they accumulate in high concentrations. When researchers coated the light-activated catalyst with PIM-1, they essentially created a trap: PFAS gets pulled in, held close to the catalyst surface, and exposed to the light-driven reaction long enough for real breakdown to occur.
Each PFAS molecule used in the study contained 17 fluorine atoms. When the reaction works, those atoms are stripped away and released as fluoride, giving researchers a precise way to measure how much destruction is actually happening.
What the Numbers Showed
In lab experiments, researchers used a 385-nanometer LED light source. That wavelength sits at the violet edge of the visible spectrum, the same range the catalyst responds to, which raises the possibility that future systems could be powered by sunlight rather than artificial lamps. With the catalyst alone, suspended in a strongly alkaline solution, the reaction released close to 30 percent of the fluorine locked inside the molecule over 16 hours. That is a reasonable result on its own. But the real interest was in what happened at neutral pH, the conditions that mirror actual water treatment environments.
At neutral pH, the uncoated catalyst produced a fluoride yield of roughly 1.9 percent. Add PIM-1, and that figure jumped to nearly 5.8 percent under the same conditions, almost a threefold increase. The porous coating kept drawing PFAS in and holding it against the catalyst regardless of pH, compensating for the less favorable chemistry. The coated catalyst also held up over six consecutive test cycles without losing meaningful performance, suggesting it could be reused rather than replaced after each treatment.
Notably, more catalyst is not always better. Piling on heavier amounts actually reduced effectiveness, likely because the thicker coating started blocking light and slowing the movement of molecules. A moderate amount hit the sweet spot.
A Step Toward Actually Eliminating PFAS
The study has limits. It tested one model PFAS compound under controlled lab conditions, not the complex mixture of dozens of different chemicals found in actual contaminated water. The full breakdown pathway was not mapped, and some intermediate products remain unidentified. More testing across longer timeframes and real-world water samples will be needed before this moves anywhere near practical application.
But the core concept is sound, and the neutral pH result matters. One major challenge with many light-driven PFAS experiments is that they perform best under strongly alkaline conditions, which are impractical and expensive to maintain in any real treatment system. A coated catalyst that works efficiently in ordinary water, powered by light and reusable across multiple cycles, clears several of those hurdles at once. This work suggests that destroying forever chemicals, rather than simply moving them around, may be closer to reality than it once seemed.
Paper Notes
Limitations
The study used a single model PFAS compound rather than the mixtures of dozens of different variants found in real contaminated water, and results may not translate directly across all PFAS types. The complete degradation pathway was not mapped, and some intermediate breakdown products were not identified or characterized. Hydrogen peroxide is generated as a byproduct and warrants further investigation. The reusability test covered six cycles, which does not constitute long-term stability testing. The system was not evaluated in actual contaminated water samples, which contain competing chemicals that could affect performance. Experimental measurement errors were estimated at plus or minus five percent.
Funding and Disclosures
Fernanda C. O. L. Martins and Wanessa R. Melchert received financial support from Brazilian Government agencies, including the Brazilian National Council for Scientific and Technological Development (CNPq, grants 305538/2022-5) and the Coordination of Superior Level Staff Improvement (CAPES, Finance Code 001). Akalya Karunakaran was supported by the National Overseas Scholarship-India. Frank Marken acknowledges initial funding from EPSRC (EP/K004956/1). Chris R. Bowen acknowledges support from the UKRI Frontier Research Guarantee (EP/X023265/1). The authors declared no conflicts of interest.
Publication Details
Authors: Fernanda C. O. L. Martins, Wanessa R. Melchert, Akalya Karunakaran, Chris R. Bowen, Nicholas Garrod, Philip J. Fletcher, Mariolino Carta, Dominic Taylor, Neil B. McKeown, and Frank Marken. Affiliations include the University of Bath, University of São Paulo, Swansea University, and the University of Edinburgh. | Journal: RSC Advances | Title: “Intrinsically microporous polymer (PIM-1) enhanced degradation of heptadecafluoro-1-nonanol at graphitic carbon nitride (g-C3N4)” | DOI: 10.1039/d5ra07284k | Published: January 2, 2026







