
Credit: MAXSHOT.PL on Shutterstock
In A Nutshell
- Researchers at Tokyo University of Science developed a method to create fluorescent, irregularly shaped nano-plastic particles from polypropylene, polyethylene, and PET, the same materials found in everyday packaging and clothing.
- When mouse cells were exposed to these particles for just 30 minutes, the plastics were observed entering the cells and clustering around the nucleus, likely through the same process cells use to absorb nutrients.
- Prior lab-based research on microplastic toxicity has mostly relied on perfectly spherical beads that don’t reflect the jagged, irregular shapes of real-world plastic particles, a gap this method begins to close.
- Whether these findings apply to human cells or living organisms remains an open question, but the tool gives researchers a more realistic way to study how nano-plastics behave inside living tissue.
Microplastics have already turned up in human blood, drinking water, table salt, seafood, and the air inside homes. Now, researchers have developed a way to watch in real time as these tiny particles slip inside living cells and cluster around the nucleus. What makes this worth paying attention to is which plastics the scientists used: polypropylene, polyethylene, and polyethylene terephthalate, the materials in water bottle caps, grocery bags, food packaging, and polyester clothing.
Published in Environmental Science: Advances, the study comes from scientists at Tokyo University of Science in Japan. By loading nano-sized fragments of those three everyday plastics with a fluorescent dye, the team created glowing particles that researchers then observed entering living mouse cells under a microscope, tracking where they ended up inside the cells.
This isn’t proof that grocery bags are breaking down inside human organs. It is, however, a new tool for studying what happens when cells encounter the same jagged, irregular plastic particles that shed from common products in the real world, rather than the perfectly round laboratory beads that have dominated this field for years.
Why Microplastic Shape Changes Everything
Nearly all prior research on microplastic toxicity used uniform, spherical polystyrene beads because they are easy to source and consistent to work with. But actual environmental microplastics are almost never spherical. Worn down by sunlight, mechanical abrasion, and microbial activity over years, they are fractured and rough-edged. Shape appears to matter more than researchers once assumed. One study cited in the paper found that irregularly shaped particles reduced swimming behavior in a marine fish compared to spherical ones. Fiber-shaped polypropylene particles also lingered longer in the digestive tracts of freshwater crustaceans, causing more harm than spheres did over time.
Preparing small, irregular plastic particles in the lab has historically been difficult, and most staining methods only work on one plastic type at a time. This study addressed both problems.
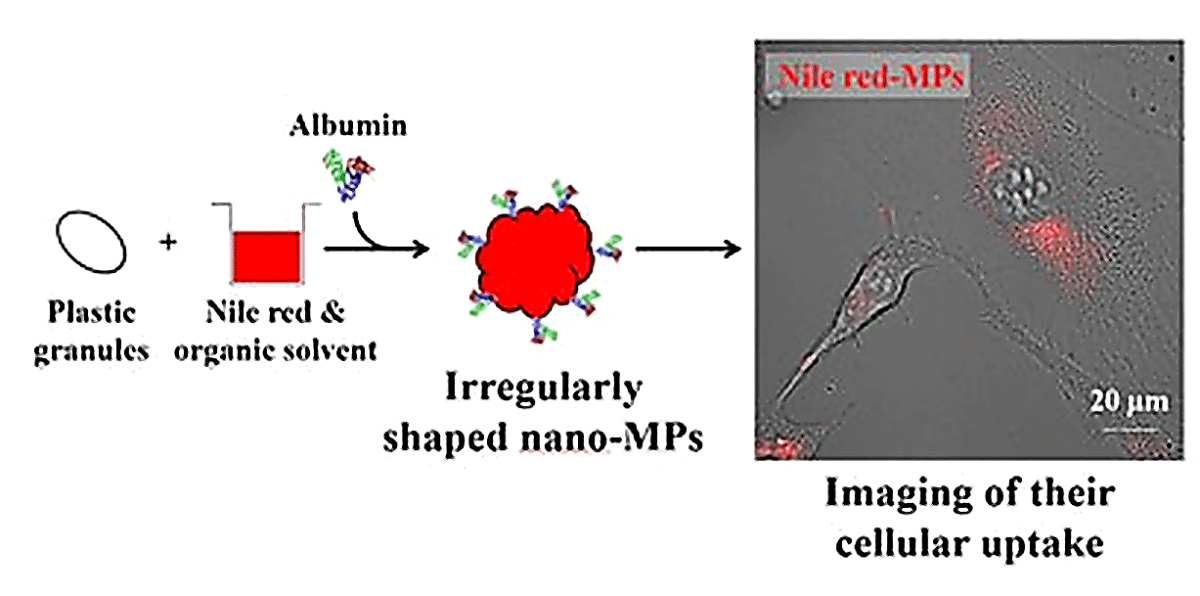
How Scientists Made Glowing Nano-Plastic Particles
To create trackable particles, the team dissolved granules of each plastic in a solvent matched to its chemical properties. This process broke the material down into particles just tens to hundreds of nanometers across, hundreds of times smaller than the width of a human hair. A fluorescent dye called Nile red, commonly used in biology to stain fats, was then mixed in. It bonds to plastic surfaces through water-repelling interactions, essentially hitching a ride on each fragment.
Next, the team added bovine serum albumin, a protein found in blood, to the mixture. It acted like dish soap, keeping fragments suspended in water rather than clumping into useless blobs. Once the solvents evaporated, stable fluorescent nano-plastic particles were left behind, ranging from roughly 80 to 250 nanometers, with uneven, jagged shapes visible under an electron microscope. Each plastic type fluoresced at a slightly different wavelength, letting researchers tell the materials apart visually.
Watching Nano-Plastics Enter Living Cells
Mouse fibroblast cells, a type of connective tissue cell found throughout mammalian bodies, were exposed to the fluorescent nano-plastics for 30 minutes, then washed and examined under a fluorescence microscope.
Rather than staying outside the cells or reaching the nucleus, the nano-plastics accumulated in the cytoplasm, the gel-like interior of the cell, clustered around the nucleus. “These images suggest that MPs are transported into the cells via endocytic pathways,” the researchers wrote. Endocytosis is a natural process cells use to pull in outside material by wrapping it in a membrane, the same mechanism used to absorb nutrients. Fluorescent signals held up for several months after the particles were first prepared, suggesting the dye remained bound without significant leaching.
What This Means for Everyday Plastic Exposure
Microplastics have been found in human blood, lungs, and digestive tracts, as well as in fruits, vegetables, bottled water, and seafood. Prior animal studies suggest plastic particles smaller than 200 nanometers may accumulate in organs including the liver, lungs, and kidneys, with smaller particles penetrating tissue more readily than larger ones. Polypropylene and PET alone account for a large share of microplastics measured in ocean sediments, coastal air samples, and indoor household dust. These are not obscure industrial materials. They are found in single-use packaging, synthetic textiles, and containers used every day.
Building a research tool from those exact materials, at the size range capable of crossing into cells, gives scientists a far more realistic baseline for studying what chronic plastic exposure might do to living tissue. Whether the same cellular uptake patterns occur in humans remains an open question, and this work was conducted in a lab dish, not in a living animal. But for a field that has spent years drawing conclusions from spherical beads made of materials barely found in the environment, that is a meaningful shift in direction.
Disclaimer: This study was conducted using lab-prepared nano-plastic model particles and mouse fibroblast cells in a controlled laboratory setting. Results do not establish that microplastics cause harm in humans, and findings should not be interpreted as direct evidence of health risk from everyday plastic exposure. Further research in human cells and living organisms is needed.
Paper Notes
Study Limitations
This study was conducted in a controlled laboratory setting using mouse fibroblast cells (3T3-L1), not human cells or living animals. Whether the observed uptake patterns apply to human tissue or cells from other organs remains to be determined. The method does not fully replicate the physicochemical history of environmental nano-plastics, which develop higher crystallinity through years of UV exposure and physical weathering; lab-prepared particles may therefore have a different internal structure than real-world fragments. Precise control of particle shape and size is difficult to reproduce consistently, which may limit the method’s use as a standardized reference material in toxicity testing. Particle concentration in cell experiments was not rigorously controlled, and potential dye leaching in aqueous suspension was not formally assessed, though fluorescence remained stable over several months. Nile red dye cannot be used to label all plastic types, only those with sufficient chemical affinity for it.
Funding and Disclosures
Work was supported in part by the Japan Society for the Promotion of Science (JSPS) KAKENHI program, under grant numbers 22H03335, 22K06565, and 25K02871. All authors declared no conflicts of interest.
Publication Details
Authors: Ryo Nagasawa and Sota Inoue are affiliated with the Department of Materials Science and Technology, Graduate School of Advanced Engineering, Tokyo University of Science, Tokyo, Japan. Takashi Miyano and Masakazu Umezawa are affiliated with the Department of Medical and Robotic Engineering Design, Faculty of Advanced Engineering, Tokyo University of Science, Tokyo, Japan. Umezawa is the corresponding author and led the overall research. Nagasawa and Inoue conducted the experiments and collected data; Miyano contributed to fluorescence microscopy; Nagasawa drafted the manuscript, which Umezawa edited. | Title: “Preparation of irregularly shaped, nano-sized, fluorescent microplastic particles for tracing cellular uptake” | Journal: Environmental Science: Advances (Royal Society of Chemistry) | Received: January 16, 2026 | Accepted: February 9, 2026 | Published: February 18, 2026 | DOI: 10.1039/d6va00031b | Data availability: Raw data are available at https://tus.box.com/s/1xbyqefq74zvlyunlaxx699ww5we3pg3 | License: Open access under Creative Commons Attribution-NonCommercial 3.0 Unported Licence.







