
Different time, different mind. (© Leo - stock.adobe.com)
The human brain is anything but static, shifting neural activity depending on time of day.
In A Nutshell
- Scientists mapped active neurons in mouse brains at four different times of day and found the same physical brain creates vastly different activity patterns depending on the time
- Deep layer 5 cortical neurons become significantly more active at the end of the wake period, suggesting rising sleep pressure
- The balance between excitatory and inhibitory neurons shifts throughout the day, especially in the thalamus, while the cortex stays relatively stable
- Brain network hubs move from subcortical wake-promoting regions in early evening to cortical default-mode-like areas by late night
The brain you have at midnight is not the same brain you have at noon. Research that mapped exactly which neurons fire up during different times of the day found that, despite having the same cells and physical wiring, the brain’s activity patterns and network organization change depending on the time.
Scientists at the University of Michigan and University of Zurich developed a way to capture snapshots of brain activity across four time windows in freely behaving mice. The communication highways between brain regions reorganize across the day.
The researchers explain in their paper published in PLOS Biology that while the brain’s physical structure and the molecular profiles of its neurons stay stable, the strength of connections and how active individual neurons are can change significantly throughout the day.
How Scientists Captured Brain Activity Throughout the Day
The research team tagged active neurons at four time points: early morning when mice settle in for sleep, late morning during rest, early evening when they wake up, and late night near the end of their active period. They used a genetic system that marks neurons expressing c-Fos, a gene that switches on when neurons activate. This approach captures an indirect marker of recent activity, with each snapshot aggregating roughly four hours of activity rather than second-to-second spikes.
After making the brains transparent using a tissue-clearing technique called CUBIC, the team photographed the entire organ in three dimensions with light-sheet microscopy. Machine learning algorithms identified individual neurons, capturing about one percent of all neurons—specifically the most active ones during each time window.
Numbers varied across the day. Activity was higher late at night, but location mattered more than quantity.
Deep Brain Layers Take Over at the End of the Day
The cortex, the brain’s outer layer for higher-level processing, showed a counterintuitive pattern. Active cortical neurons were more common during early morning rest than during early evening waking. Breaking down the cortex by layers revealed why.
Layer 5 neurons sit in the deeper part of the cortex and coordinate communication between cortical regions. These neurons became significantly more active at the end of the dark period, right when mice prepare to sleep.
Layer 5 neurons help coordinate signals between different cortical areas and play a role in generating the slow brain waves seen during sleep. The increased activity late in the active period may reflect rising sleep pressure.
The pattern appeared across multiple cortical regions, from visual to retrosplenial to temporal association areas. Most regions showed no major changes when analyzed as a whole. Breaking them down by layer revealed substantial reorganization beneath the surface.
The Brain’s Excitation-Inhibition Balance Shifts at Night
The researchers matched their active neurons to a spatial transcriptomics dataset, determining what type of neurotransmitter each neuron releases. This revealed a time-dependent shift between excitatory neurons (which use glutamate to activate other neurons) and inhibitory neurons (which use GABA to quiet activity).
From early evening to late night, active excitatory neurons increased while inhibitory neurons decreased in several regions, especially the thalamus—a deep structure that relays sensory information and regulates sleep and wakefulness. The cortex remained comparatively stable across these multi-hour windows.
This pattern contrasts with some previous studies that reported dynamic cortical changes, though those typically measured electrical activity rather than counting neurons.
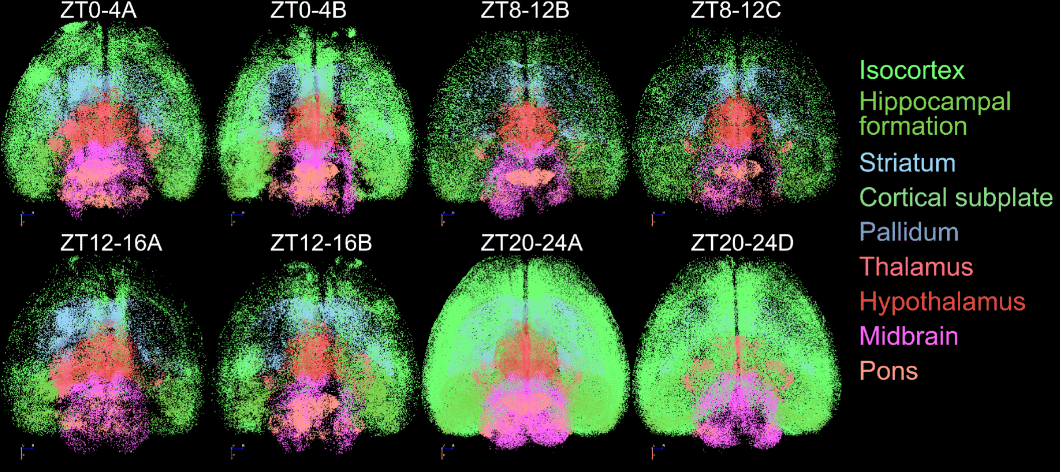
Brain Network Hubs Shift Location Throughout the Day
The researchers constructed “active connectivity,” mapping which brain regions can communicate based on both structural wiring and whether neurons in those regions are turned on.
The team calculated betweenness centrality for each region, identifying hubs that bridge different network parts. These are the brain’s major communication waypoints—regions that information must pass through.
Hub locations shifted dramatically over 24 hours. During early evening when mice become active, major hubs concentrated in subcortical regions like the posterior hypothalamic nucleus, which promotes wakefulness and arousal.
By late night, the network had reorganized. Many hub roles shifted toward cortical regions, particularly in default-mode-like networks, interconnected areas in mice that resemble networks originally discovered in humans through brain imaging.
The posterior hypothalamic nucleus ranked among the most central hubs during early evening but dropped substantially by late night as cortical regions climbed. Network modularity (how well it divides into distinct communities) appeared to decrease from early evening to late night, which would mean the brain became more globally integrated.
What This Means for Understanding Brain Function
The brain operates more like a subway system than a static electrical circuit. The tracks stay the same, but which trains run, which stations open, and which routes are available changes hour by hour.
This matters for understanding neurological and psychiatric conditions that show strong time-of-day patterns. Sleep disorders, mood disorders, and seizure susceptibility all vary with circadian rhythms. Knowing how the brain’s networks shift throughout the day could point toward treatments that target specific times when particular circuits are most active or vulnerable.
The study’s framework combines genetic tagging with tissue clearing, 3D microscopy, spatial transcriptomics, and network analysis. Functional MRI captures interactions over seconds to minutes. This method reveals integration of activity over hours, showing how the brain organizes itself over longer timescales.
The approach can be adapted to study neurodegenerative diseases, behavioral interventions, or sleep deprivation. By identifying which specific neurons and networks activate during different experimental conditions, researchers can move beyond regional brain maps to understand the cellular players in specific behaviors or brain states.
The physical brain atlas is essentially complete. The next frontier is understanding how that static anatomy gives rise to dynamic function: how the same structure produces. This study provides one of the first whole-brain, cellular-resolution views of that transformation.
Paper Notes
Limitations
The experiment captured only about 1% of the brain’s neurons (specifically the most active ones), meaning the vast majority of neurons with lower activity levels were not included in the analysis. The olfactory areas, medulla, and cerebellum were excluded from some analyses due to incomplete preservation during tissue processing. The matching of neurons to the spatial transcriptomics dataset relied on spatial proximity within brain regions, which may be less accurate in areas where different neuron types are tightly mixed, such as cortical layers 2/3. The method integrates activity over approximately 4-hour windows, so faster dynamics are not captured. The active connectivity analysis used an early version of the Allen Brain Atlas connectivity data and did not incorporate more recent layer-specific and cell-type-specific projection information, which is currently limited to the thalamocortical system.
Funding and Disclosures
The research was supported by the Human Frontier Science Program (HFSP grant RGP0019/2018 to DBF, SB and HU), National Science Foundation Division of Mathematical Sciences (NSF DMS 2052499 to DBF), Army Research Office Multidisciplinary University Research Initiatives (ARO MURI W911NF-22-1-0223 to DBF), and Velux Stiftung (grant 1812 to KK). Co-author H.R. Ueda is a co-inventor on patent applications covering the CUBIC reagents used in the tissue clearing process and is a co-founder of CUBICStars Inc. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Publication Details
“A framework to determine active neurons and networks within the mouse brain reveals how brain activity changes over the course of the day” by Guanhua Sun, Tomoyuki Mano, Shoi Shi, Alvin Li, Koji L Ode, Alex Rosi-Andersen, Erica Pedron, Steven A. Brown, Hiroki R Ueda, Konstantinos Kompotis, and Daniel B. Forger. Published November 13, 2025 in PLOS Biology, volume 23, issue 11, article e3003472. DOI: 10.1371/journal.pbio.3003472. The paper is distributed as open access under the Creative Commons Attribution License. The study was conducted at the Department of Mathematics and Gilbert S. Omenn Department of Computational Medicine and Bioinformatics at the University of Michigan (USA), the Chronobiology and Sleep Research Group at the University of Zurich (Switzerland), the Department of Systems Pharmacology at the University of Tokyo (Japan), and several other collaborating institutions. This paper is dedicated to the memory of Steven A. Brown, a senior co-author who tragically died in a plane crash during the research.







