
(© Antonio Gravante - stock.adobe.com)
In A Nutshell
- Researchers screened roughly 3,500 compounds using miniature lab-grown human intestines and identified glycyrrhizin, the natural compound that gives licorice its flavor, as a potential protector of gut cells against a key form of inflammatory damage linked to Crohn’s disease.
- Glycyrrhizin works by blocking a specific cell self-destruct pathway called apoptosis, and its protection disappears once gut bacteria break it down into a simpler form, raising important questions about how any future treatment would need to be delivered.
- In mice with chemically induced gut inflammation, glycyrrhizin reduced tissue damage, lowered cell death in the intestinal lining, and prevented colon shortening, though it did not produce a statistically significant improvement in body weight loss.
A substance that has been sweetening candy and herbal teas for centuries might have a second act nobody expected. Glycyrrhizin, the compound responsible for licorice root’s distinctive flavor, can block a form of inflammatory cell death linked to Crohn’s disease, according to researchers.
Crohn’s disease affects millions of people worldwide and remains stubbornly difficult to manage. The condition causes long-term inflammation in the digestive tract, destroying the cells that line the intestines and making it harder for the body to absorb nutrients. The most common front-line treatments use lab-made antibodies that neutralize TNF, a molecule the immune system produces during inflammation. But many patients either don’t respond to these drugs or stop benefiting over time, in part because the body learns to fight off the medication itself. And even when the drugs do work, they tamp down inflammation without necessarily repairing the damaged intestinal wall.
That gap in treatment is what pushed a team of researchers based primarily at The University of Tokyo and Chiba University to try something different. Rather than blocking the inflammatory signal, they went looking for compounds that could protect intestinal cells directly, giving gut tissue a kind of armor against TNF’s destructive effects. To do it, they built a drug-screening system using miniature, lab-grown versions of human intestines. Their paper is published in Stem Cell Reports.
How Lab-Grown Mini-Guts Changed the Search for Crohn’s Disease Treatments
The miniature intestines at the center of this work are called organoids: tiny, three-dimensional clusters of cells that organize themselves into structures resembling real organs. The researchers used human intestinal organoids grown from stem cells, which behave far more like actual human gut tissue than the cancer-derived cell lines scientists have traditionally used in lab experiments.
That distinction turned out to matter a great deal. When the team exposed standard lab cell lines derived from colon cancers to TNF, those cells barely reacted. But when organoid-derived intestinal cells got the same treatment, they died at concentrations matching or below levels found in the intestinal tissue of patients with active Crohn’s disease. In short, the lab-grown cells responded to TNF in a way that actually mirrors what happens inside a real patient, something the cancer cell lines simply could not do.
With this more realistic testing platform in hand, the researchers screened roughly 3,500 active compounds, looking for any that could prevent TNF from killing the organoid-derived intestinal cells. They ran the screen in a tray roughly the size of a paperback book, dotted with hundreds of tiny wells, each holding cells and a test compound.
How Glycyrrhizin Protects the Gut From Crohn’s Disease
Out of thousands of compounds tested, thirteen showed some protective activity. After follow-up testing at multiple doses, three stood out: thiostrepton (an antibiotic), PF-04691502 (a cancer research compound), and glycyrrhizin. Glycyrrhizin emerged as the most specific candidate: the only one that acted selectively against TNF-induced damage without simply accelerating cell growth.
The other two compounds boosted cell numbers even when TNF wasn’t present, suggesting they were simply making cells multiply faster rather than truly shielding them from inflammatory damage. Glycyrrhizin, by contrast, had no effect on cell growth by itself. It sprang into action only when TNF was actively killing cells, a sign that its protective effect was specific to the injury rather than a general growth-promoting trick. The compound also protected intestinal cells derived from multiple sources, including organoids grown from donated human tissue, with consistent effectiveness across different cell types.
One curious finding: glycyrrhizin did not protect a mouse cell line commonly used to study TNF-induced cell death. Rather than a flaw, this turned out to be an important clue.
TNF can kill cells through two distinct pathways. One is apoptosis, a tightly controlled self-destruct sequence. The other is necroptosis, a messier, more inflammatory form of death that sends damage signals rippling through surrounding tissue. The researchers found that intestinal organoid cells primarily die through apoptosis when exposed to TNF, while the mouse cells die through necroptosis. Glycyrrhizin specifically blocks the apoptotic pathway, which explains why it rescued the gut cells but not the mouse cells.
Digging further, the team pinpointed where glycyrrhizin acts within the cell’s self-destruct sequence. It acts downstream of an early trigger in that sequence, blocking later steps, including the cleavage of key proteins that carry out cell death, without shutting down the initiating signal itself.
The researchers also ruled out a popular theory about how glycyrrhizin works. The compound is widely known to interfere with HMGB1, a protein that dying cells release to ramp up inflammation. The team initially suspected this might explain the protective effect. But when they used genetic techniques to dramatically reduce HMGB1 levels in organoid cells, TNF-induced death continued at the same rate. The HMGB1 theory didn’t hold up in this model.
Another surprise emerged when the team tested glycyrrhetinic acid, the form that glycyrrhizin gets converted into by bacteria living in the gut. These gut bacteria strip away sugar groups from the glycyrrhizin molecule, fundamentally altering its structure. This breakdown product completely failed to protect intestinal cells. That means the whole, intact glycyrrhizin molecule, sugar groups and all, is necessary for the protective effect, and it raises real questions about how to deliver the compound effectively in a medical setting, since gut bacteria would naturally break it down before it could do its job.
From Lab Dish to Living Animal
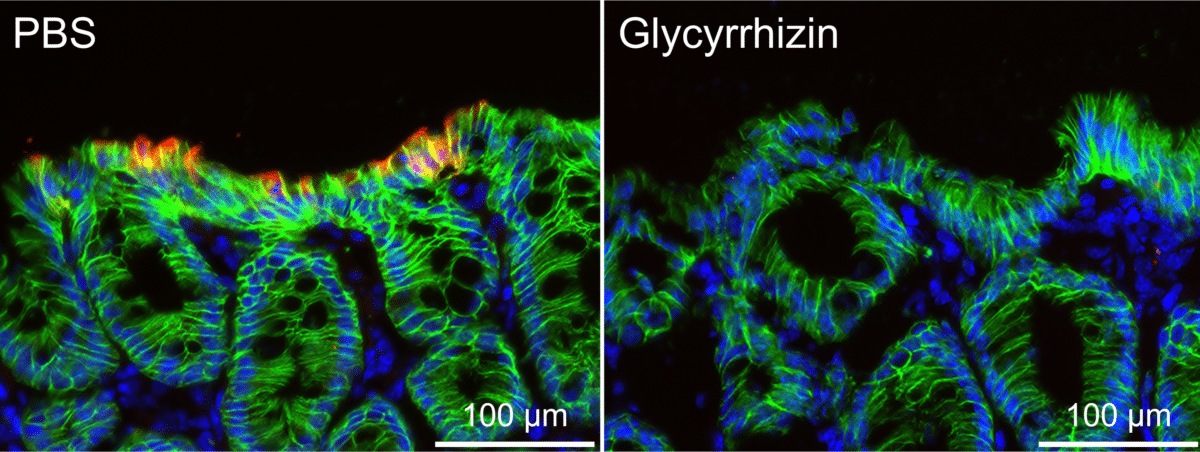
To see whether these findings held up outside a petri dish, the team turned to a well-established mouse model of intestinal inflammation. Mice received a chemical in their drinking water for five days to trigger gut inflammation. Some also received glycyrrhizin by mouth, starting three days before the inflammation was induced and continuing daily for seven days.
Mice that received glycyrrhizin showed less tissue damage and fewer immune cells flooding into their colons, based on microscopic examination. Colon shortening, a standard indicator of inflammation severity in this model, was significantly reduced in the treated group. Consistent with the lab results, the percentage of cells undergoing apoptosis in the intestinal lining was significantly lower in mice that received glycyrrhizin. Body weight loss, another common measure of disease severity, trended toward improvement in treated mice, though the difference did not reach statistical significance.
This study makes two contributions worth paying attention to. First, it shows that organoid-based screening can spot drug candidates that would be completely invisible using traditional cancer cell lines. If the researchers had relied on those older tools, they would never have detected TNF’s devastating effect on intestinal cells in the first place, and glycyrrhizin’s protective activity would have gone unnoticed.
Second, it identifies a naturally occurring, widely consumed compound with a specific and previously unrecognized way of protecting gut cells. Glycyrrhizin has been used as a sweetener and in traditional medicine in China and Japan for generations. The World Health Organization has established a safety threshold for it based on normal dietary consumption. This is not some exotic, untested molecule. It is something humans have been eating for a very long time.
None of this means people with Crohn’s disease should start loading up on licorice. The leap from a mouse model to a human therapy is long and uncertain, and the fact that gut bacteria convert glycyrrhizin into a form that lacks protective activity means figuring out how to get the intact molecule to the right cells is a real challenge.
The study demonstrates protective potential in lab and animal settings, but whether that translates to meaningful benefit in human patients remains to be seen. In a disease where available treatments fail too many patients and leave the intestinal wall unrepaired, a naturally derived compound that directly shields gut cells from inflammatory destruction is, at the very least, a lead worth following.
Disclaimer: This article is for informational purposes only and is not medical advice. The findings described are based on early laboratory and animal research and have not been tested in human patients. Glycyrrhizin is not an approved treatment for Crohn’s disease. Always consult a qualified healthcare professional before making any changes to your diet, supplements, or medical care.
Paper Notes
Limitations
The study’s authors acknowledge several important limitations. Human intestinal organoids consist solely of one cell type and lack other parts of the intestinal environment, including immune cells, connective tissue cells, and bacteria, all of which may influence how intestinal cells respond to inflammation and treatment. Whether glycyrrhizin’s protective effect translates to meaningful clinical benefit in humans remains undetermined. The mouse model used involved only four mice per group, limiting statistical power for some measures, and body weight recovery did not reach statistical significance. The authors also note that because intestinal bacteria convert glycyrrhizin into glycyrrhetinic acid, which lacked protective activity, delivery strategies that preserve the intact molecule at the intestinal lining may be needed to improve effectiveness in living systems. Testing glycyrrhizin in organoids derived from Crohn’s disease patients could help assess clinical relevance and individual variability.
Funding and Disclosures
This study was supported by the Ministry of Education, Culture, Sports, Science and Technology of Japan (Grant-in-Aid for Scientific Research C, 19K05928; Challenging Research Exploratory, 22K18342), the Kobayashi Foundation, the Platform Project for Supporting Drug Discovery and Life Science Research from AMED (JP20am0101086), JST FOREST Program (JPMJFR2354), the cell science research foundation, and the AMED NeDDTrim project (JP24ae0121040). The authors declare no competing interests.
Publication Details
Title: “Organoid phenotypic screening identified glycyrrhizin that confers protection against tumor necrosis factor-induced cell death” | Authors: Yu Takahashi, Zhongwei Zhang, Izumi Tanaka, I-Ting Lee, Jincheng He, Yurina Koura, Shintaro Sato, Hirotatsu Kojima, Takayoshi Okabe, Hiroshi Kiyono, Takashi Sasaki, Yoshio Yamauchi, Yosuke Kurashima, and Ryuichiro Sato | Institutions: The University of Tokyo (Graduate School of Agricultural and Life Sciences; Drug Discovery Initiative, Graduate School of Pharmaceutical Sciences); Chiba University (Graduate School of Medicine; Synergy Institute for Futuristic Mucosal Vaccine Research and Development; Chiba University Hospital); Wakayama Medical University (School of Pharmaceutical Sciences); Osaka University (Research Institute for Microbial Diseases); Chiba University–University of California San Diego Center for Mucosal Immunology, Allergy and Vaccine (CU-UCSD cMAV), Department of Medicine, School of Medicine | Journal: Stem Cell Reports, Volume 21, Article 102891, May 12, 2026 | DOI: 10.1016/j.stemcr.2026.102891 | License: Open access under CC BY-NC-ND 4.0, published by Elsevier Inc. on behalf of the International Society for Stem Cell Research







