
(© I Viewfinder - stock.adobe.com)
BOSTON — The next frontier in the battle against respiratory infections isn’t a pill or a shot – it’s a spray. Scientists at Brigham and Women’s Hospital have developed PCANS, a nasal spray that could potentially shield us from a wide range of airborne threats, from everyday colds to pandemic-causing viruses.
It’s a breakthrough that would be music to the ears — and lungs — of those who find themselves battling colds and coughs frequently during flu season. This multi-modal nasal spray is designed to capture, neutralize, and prevent the transport of various respiratory pathogens.
The study, published in the journal Advanced Materials, details the creation of PCANS (Pathogen Capture and Neutralizing Spray). If successful, it could protect individuals from COVID-19, influenza, the common cold, and even pneumonia-causing bacteria.
“The COVID pandemic showed us what respiratory pathogens can do to humanity in a very short time. That threat hasn’t gone away,” says co-senior author Jeffrey Karp, PhD, distinguished chair in Anesthesiology at Brigham and Women’s Hospital, in a statement. “Not only do we have the flu to deal with seasonally, but we now have COVID, too.”
Indeed, influenza and COVID-19 infections continue to cause thousands of deaths and hundreds of thousands of cases of severe disease every year. Even milder infections result in significant discomfort and disruption to daily life, leading to missed work or school days.
While vaccines and masks have played crucial roles in combating these infections, they have limitations. Vaccinated individuals can still contract and spread infections, and masks, while helpful, are not foolproof and are often worn improperly or not at all.
“We need new, additional ways to protect ourselves and reduce the transmission of the disease,” Karp emphasizes.
PCANS takes a novel approach to preventing infection. Most respiratory pathogens enter our system through the nose, hitching a ride on tiny droplets of fluid that we inhale. PCANS aims to stop this process in its tracks.
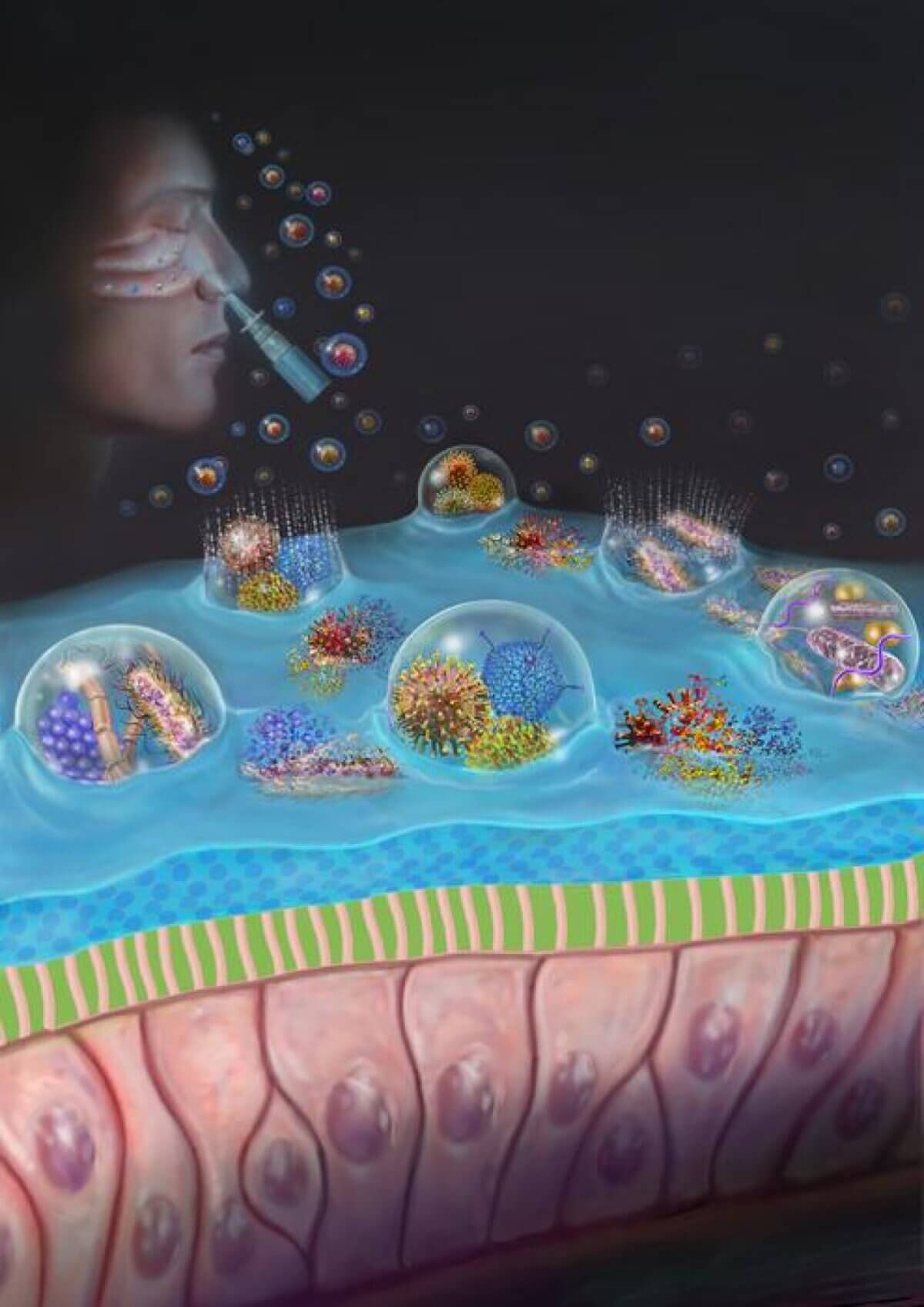
“The spray was developed using ingredients from the FDA’s Inactive Ingredient Database (IID), which have been previously used in approved nasal sprays, or from the Generally Recognized as Safe (GRAS) list of the FDA,” explains co-senior author Nitin Joshi, PhD, an Assistant Professor of Anesthesiology at Brigham and Women’s Hospital. “We developed a drug-free formulation using these compounds to block germs in three ways — PCANS forms a gel-like matrix that traps respiratory droplets, immobilizes the germs, and effectively neutralizes them, preventing infection.”
In laboratory tests, PCANS demonstrated impressive results. When sprayed into a 3D-printed replica of a human nose, it captured twice as many droplets as mucus alone. Even more remarkably, it blocked and neutralized almost 100% of all viruses and bacteria tested, including Influenza, SARS-CoV-2, RSV, adenovirus, and K. pneumoniae.
The effectiveness of PCANS was further demonstrated in animal studies. In mice, a single dose of the nasal spray effectively blocked infection from an influenza virus at 25 times the lethal dose. Virus levels in the lungs were reduced by more than 99.99%, and inflammation markers in the lungs of treated animals remained normal.
“The formulation’s ability to inactivate a broad spectrum of pathogens, including the deadly PR8 influenza virus, demonstrates its high effectiveness,” adds co-senior author Yohannes Tesfaigzi, PhD, AstraZeneca Professor of Medicine in the Field of Respiratory and Inflammatory Diseases at Brigham and Women’s Hospital. “In a rigorous mouse model study, prophylactic treatment with PCANS demonstrated exceptional efficacy, with treated mice exhibiting complete protection, while the untreated group showed no such benefit.”
One of the most promising aspects of PCANS is its potential for long-lasting protection. In tests on mice, the nasal spray showed a residence time of at least eight hours in the nasal cavity. This prolonged presence could potentially minimize the need for frequent reapplication, making it a convenient option for daily use.
While these results are undoubtedly exciting, it’s important to note that PCANS has not yet been tested in humans. The next steps would likely involve larger animal studies and, eventually, human clinical trials to confirm its safety and efficacy in people.
If proven effective in humans, PCANS could have far-reaching implications for public health. It could serve as a first line of defense against respiratory pathogens, particularly in situations where vaccines are not available or for individuals who cannot be vaccinated. The spray could be especially beneficial for high-risk populations, such as healthcare workers, the elderly, or those with compromised immune systems.
Moreover, the simplicity of PCANS’ formulation and its use of readily available ingredients make it a promising candidate for rapid scale-up and global distribution. This could be crucial in quickly responding to future pandemic threats.
The researchers are also exploring whether PCANS can block allergens, potentially opening up a new avenue for allergy relief. This versatility underscores the potential wide-ranging impact of this innovative nasal spray.
While there’s still work to be done before PCANS reaches our medicine cabinets, its development represents a significant step forward in our ongoing battle against respiratory diseases — and a world where we can all breathe a little easier.
Paper Summary
Methodology
The researchers employed a comprehensive approach to develop and test PCANS. They began by screening various biopolymers, surfactants, and alcohols for their ability to form physical barriers, neutralize pathogens, and capture respiratory droplets. They used techniques such as rheological measurements, trans-membrane assays, and plaque assays to evaluate these properties.
The team then optimized the formulation through iterative testing, focusing on factors like sprayability, mucoadhesiveness, and nasal retention. They used both in vitro and in vivo models to assess the efficacy of PCANS, including tests on cell cultures, 3D models of human nasal cavities, and mouse studies.
Key Results
The study yielded several significant results. In vitro, PCANS demonstrated over 99.99% reduction in the transport and neutralization of various pathogens, including influenza A virus, SARS-CoV-2, and E. coli. The spray showed a residence time of at least 8 hours in mouse nasal cavities. In the mouse influenza model, a single pre-exposure dose of PCANS led to 100% survival, compared to 0% in the control group, and resulted in undetectable levels of virus in the lungs. The formulation also showed excellent stability over 60 days under accelerated storage conditions.
Study Limitations
The primary limitation of this study is the lack of human trials. While the results in laboratory settings and animal models are promising, the effectiveness and safety of PCANS in humans remain to be established. Additionally, the study focused primarily on influenza and SARS-CoV-2, and while other pathogens were tested in vitro, more extensive testing against a wider range of respiratory pathogens would be beneficial. The long-term effects of regular use of PCANS on nasal health and the respiratory system also need to be investigated.
Discussion & Takeaways
The researchers emphasize that PCANS represents a novel, multi-modal approach to respiratory infection prevention. They highlight its potential as a complement to vaccines, especially for emerging pathogens or in situations where vaccines are not immediately available. The study suggests that PCANS could be particularly beneficial for high-risk populations or in settings with increased exposure risk.
The researchers also note the potential for rapid scale-up and global distribution due to the simplicity of the formulation and use of readily available ingredients. However, they stress the need for further studies, particularly in larger animal models and human trials, to fully validate the safety and efficacy of PCANS.
Funding & Disclosures
The study was supported by funding from the Gillian Reny Stepping Strong Center for Trauma Innovation at Brigham and Women’s Hospital, and the Department of Anesthesiology, Perioperative, and Pain Medicine at Brigham and Women’s Hospital. Several authors, including John Joseph, Helna Mary Baby, Yohannes Tesfaigzi, Jeffrey Karp, and Nitin Joshi, have a pending patent based on the PCANS formulation. Brigham and Women’s Hospital has licensed the technology to Akita Biosciences, which has commercialized PCANS as Profi Nasal Spray.
Joshi and Karp are paid consultants, scientific advisory board members, and hold equity in Akita Biosciences. The company, BWH, Joshi, and Karp may benefit financially if the intellectual property is further validated. Karp has also been a paid consultant or equity holder for multiple other biotechnology companies.







