
Dr. José-Alain Sahel holding the PRIMA implant. (Credit: UPMC)
In A Nutshell
- 81% of patients saw better after getting a tiny wireless chip implanted beneath the retina, with some able to read again after years of severe vision loss from advanced macular degeneration.
- How it works: Special glasses with a camera capture what patients look at, beam the image onto the chip using infrared light, and the chip converts it into electrical signals the brain can interpret.
- The catch: While eye chart scores improved and most patients reported reading at home, a standardized quality-of-life questionnaire showed no measurable change in daily functioning.
- Surgery risks: 26 serious complications occurred in 19 patients within a year, including elevated eye pressure, retinal tears, and macular holes—though most resolved within two months.
A wireless microchip implanted beneath the retina improved vision in 81% of patients with advanced age-related macular degeneration, allowing some to read again after years of severe vision loss. The device works with camera-equipped glasses that capture images and project them onto the chip using infrared light, which the implant converts into electrical signals the brain can interpret.
In a clinical trial involving 38 patients across Europe, most of those assessed at 12 months showed substantial improvement in their ability to see. Some went from profound vision loss to reading small print, marking what researchers describe as the first intervention to improve lost vision in this patient population rather than simply slow disease progression.
Age-related macular degeneration affects more than 5 million people worldwide and accounts for roughly 20% of all legal blindness cases in North America. Geographic atrophy, the advanced form of dry AMD studied in this trial, causes cells in the center of the retina to progressively die. As of this study period, no treatment existed to bring back vision once those cells were gone.

The PRIMA Eye Implant
The photovoltaic retina implant microarray, or PRIMA system, replaces the function of dead light-sensing cells with technology. Surgeons place a 2-millimeter square chip that’s thinner than a human hair beneath the retina in the area of vision loss.
Patients wear specialized glasses with a built-in camera that captures whatever they’re looking at. That visual information gets processed through a pocket-sized device, then projected back through the glasses onto the implant using infrared light (a type of light the eye can’t see naturally). The chip’s 378 tiny pixels convert this light into electrical pulses that stimulate the remaining healthy retinal cells, essentially restarting the flow of visual information to the brain.
Unlike other assistive devices that magnify images for healthy parts of the retina, PRIMA targets the blind spot directly while leaving natural side vision untouched. The transparent lenses mean users experience both artificial central vision and their remaining natural sight at the same time.
“All previous attempts to provide vision with prosthetic devices resulted in basically light sensitivity, not really form vision,” said Daniel Palanker, a Stanford professor of ophthalmology and a co-senior author of the New England Journal of Medicine paper, in a statement. “We are the first to provide form vision.”
Patients Gained Reading Ability After Years of Vision Loss
Among the 32 participants evaluated at one year, 26 showed improvement equivalent to gaining 10 or more letters on a standard eye chart. The average improvement was about 25 letters, while one patient improved by roughly 59 letters.
Participants could adjust zoom and brightness on their glasses during vision tests. Features like contrast adjustment and magnification helped users see more than the chip’s basic resolution would normally allow.
At home, 84% of participants reported using the device to read letters, numbers, and words. About 69% said they were moderately to highly satisfied with the system. Three participants from an earlier study maintained vision sharp enough to read small fonts four years after getting the implant.
However, there’s an important caveat. Despite better scores on eye charts and self-reported reading improvements at home, a standardized questionnaire measuring quality of life showed no change from before the surgery at 6 or 12 months. Researchers say the questionnaire may not be sensitive enough to detect changes in people with extremely poor baseline vision.
Study participants had been living with profound vision loss. The average age was 79 years, and all had geographic atrophy affecting their central vision. Most had lost the ability to read, recognize faces, or perform other tasks requiring sharp central vision.
“I was an avid bookworm, and I wanted that back. I was nervous, excited, all those things. There was no pain during the operation, but you’re still aware of what’s happening,” said Sheila Irvina, one of the patients in the study who underwent surgery at Moorfields Eye Hospital in London. “It’s a new way of looking through your eyes, and it was dead exciting when I began seeing a letter. It’s not simple, learning to read again, but the more hours I put in, the more I pick up.”
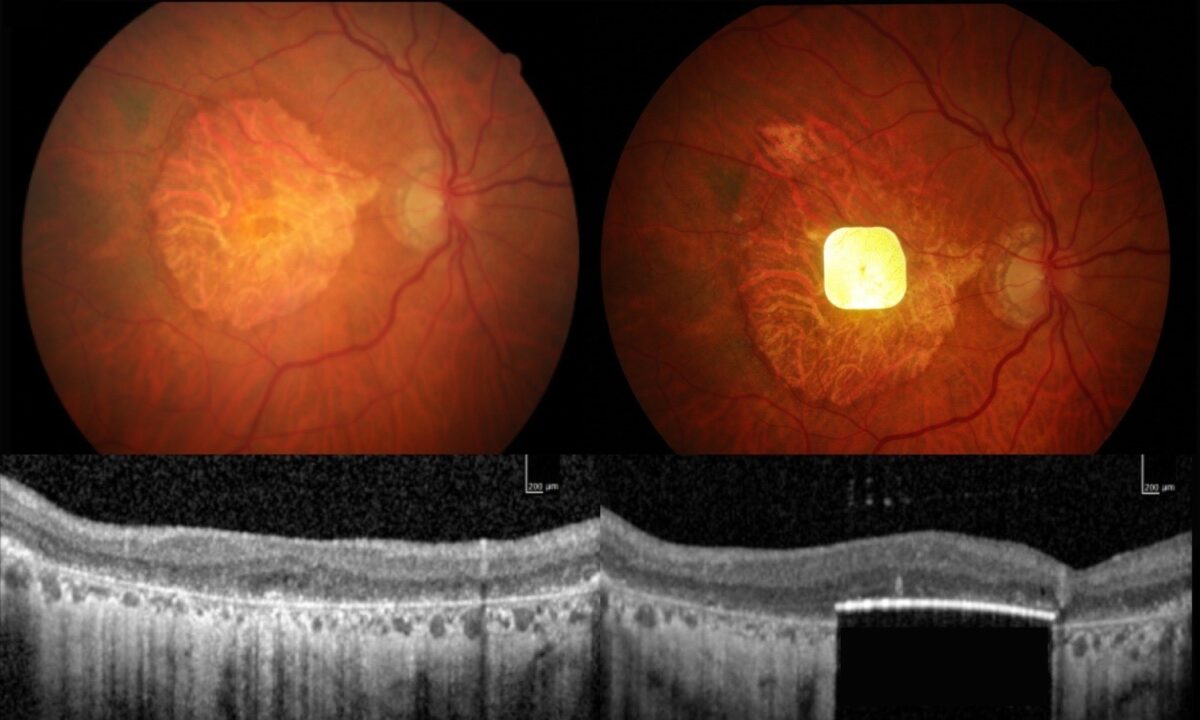
Surgery Carries Risks Common to Eye Procedures
Twenty-six serious complications occurred in 19 participants within 12 months of surgery, though researchers emphasize all were anticipated. Most complications happened within two months of the procedure, and 95% of early complications resolved within two months.
Elevated eye pressure affected six patients, appearing between day one and week three after surgery. All cases went away on their own. Five participants experienced small retinal tears during surgery, all repaired immediately without causing retinal detachment.
Three patients developed full-thickness holes in the macula (the central part of the retina). Two needed their implants repositioned away from the hole. One participant experienced multiple serious complications including retinal detachment, but was successfully treated with additional surgery.
Side vision after getting the implant remained just as good as before surgery, indicating the procedure didn’t damage the healthy retina surrounding the treated area.

Why This Approach Works Better Than Previous Eye Implants
Earlier attempts at artificial vision using different implant types didn’t work as well. An earlier device, Argus II, had a larger spacing between electrodes and stimulated cells farther from where they naturally receive signals, resulting in distorted vision and reduced sharpness.
PRIMA’s placement beneath the retina (rather than on top of it) and its wireless design were engineered to make surgery simpler than with earlier systems. The chip sits closer to the cells it needs to stimulate, which improves how well patients can see. Stimulating cells earlier in the vision pathway preserves more of the eye’s natural signal processing. Even small eye movements can help improve clarity beyond what the pixel size would suggest.
The wireless nature means no permanent openings in the eyeball, unlike older wired implants. The chip is extremely thin, about half the height of a natural light-sensing cell, which allows it to integrate with surrounding tissue without needing screws or tacks to hold it in place.
Current Limitations and Ongoing Research
Six participants weren’t evaluated at 12 months: three had died from causes unrelated to the study, one dropped out, and two weren’t available for testing.
The treated eyes showed greater disease progression than the untreated eyes. Researchers attribute this to the surgery itself rather than the device, since the progression occurred in areas where surgical instruments created temporary fluid pockets and small openings. Treated eyes showed an average disease progression of 8.5 square millimeters over the year, compared to 2.5 square millimeters in untreated eyes.
PRIMA’s wireless design could theoretically allow doctors to replace it with higher-resolution versions in the future, or even place multiple chips across a larger blind spot with minimal incisions. Participants using zoom and other digital features could read fonts smaller than the hardware alone should allow, suggesting the combination of chip and image processing extends useful vision.
Researchers plan to follow participants for up to three years after surgery. The first therapies recently approved for geographic atrophy work by slowing disease progression through monthly or twice-monthly injections. PRIMA takes a fundamentally different approach: rather than trying to keep cells alive, it replaces their function once they’re already dead.
For patients with geographic atrophy, these results offer something no previous treatment could. While drugs like pegcetacoplan and avacincaptad pegol can slow the spread of damage, they can’t reverse vision that’s already gone. PRIMA fills that gap by creating an artificial way to transmit visual information from areas where light-sensing cells have died.
“This is the first version of the chip, and resolution is relatively low,” said Palanker. “The next generation of the chip, with smaller pixels, will have better resolution and be paired with sleeker-looking glasses.”
Paper Summary
Methodology
Researchers conducted an open-label, multicenter study across 17 sites in five European countries. Participants aged 60 or older with geographic atrophy due to AMD in both eyes, visual acuity of at least 1.2 logMAR (Snellen equivalent 20/320) in the study eye, and foveal atrophy larger than the implant received the PRIMA device. Surgeons performed vitrectomy and created a subretinal space to position the photovoltaic array within the blind spot. Participants underwent training sessions to optimize use of the glasses and processor. Visual acuity was assessed at baseline and at six and 12 months using standard eye charts, both with and without PRIMA glasses. The primary end points were clinically meaningful improvement in visual acuity, defined as an improvement of at least 0.2 logMAR (corresponding to an increase of 10 or more letters) from baseline to month 12, and the number and severity of serious adverse events through month 12 after surgery that were considered related to the procedure or device.
Results
Of 38 implanted participants, 32 completed 12-month evaluation. Among those 32, 26 (81%) achieved clinically meaningful improvement of at least 0.2 logMAR. Statistical modeling accounting for six participants with missing data estimated 80% would have shown clinically meaningful improvement. Mean improvement from baseline to 12 months was 0.49 logMAR with PRIMA glasses and 0.51 logMAR with participant’s choice (whichever condition yielded better acuity). Maximum improvement reached 1.18 logMAR. At six months, 20 of 35 evaluated participants with available data showed improvement of at least 0.2 logMAR. Visual acuity without PRIMA glasses remained unchanged from baseline at both time points. Among 32 participants tested, 30 demonstrated central visual perception with the system at 12 months. The implant-resolution test showed mean visual acuity of 1.32 logMAR, matching theoretical resolution achievable with 100-micrometer pixels. In a post hoc analysis using a threshold of 0.3 logMAR improvement, 25 of 32 participants (78%) met this higher bar.
Limitations
The study used an open-label, single-group design without a control group, limiting ability to account for placebo effects or natural disease progression. Six participants weren’t assessed at 12 months due to death (3 participants), withdrawal (1), or unavailability (2). The Impact of Vision Impairment questionnaire showed no changes in self-reported daily functioning, possibly due to insufficient sensitivity for this extremely low-vision population. Atrophy increased more in study eyes (mean 8.5 mm²) compared to fellow eyes (mean 2.5 mm²), attributed to surgical trauma including retinotomy and bleb formation. The study wasn’t powered to detect changes in quality-of-life measures. Follow-up was limited to 12 months for the primary analysis, though longer-term data collection continues. The participant population was exclusively European, potentially limiting generalizability to other geographic populations. The study did not assess functional outcomes beyond visual acuity measurements and questionnaire responses.
Funding and Disclosures
The study was funded by Science Corporation (previously Pixium Vision) and the Moorfields National Institute for Health and Care Research Biomedical Research Centre (award NIHR203322). Multiple authors disclosed financial relationships with Science Corporation including employment, consulting fees, and equity ownership. Several authors reported grants, personal fees, or other relationships with pharmaceutical and medical device companies outside the submitted work. The sponsor participated in study design and data analysis.
Publication Information
The study “Subretinal Photovoltaic Implant to Restore Vision in Geographic Atrophy Due to AMD” was published in The New England Journal of Medicine on October 20, 2025 (DOI: 10.1056/NEJMoa2501396). The clinical trial was registered at ClinicalTrials.gov under identifier NCT04676854. The research was approved by local ethics committees at participating sites, and all participants provided written informed consent. A data and safety monitoring board reviewed all serious adverse events throughout the study period. First author Frank G. Holz can be contacted at the Department of Ophthalmology, University of Bonn, Germany.







