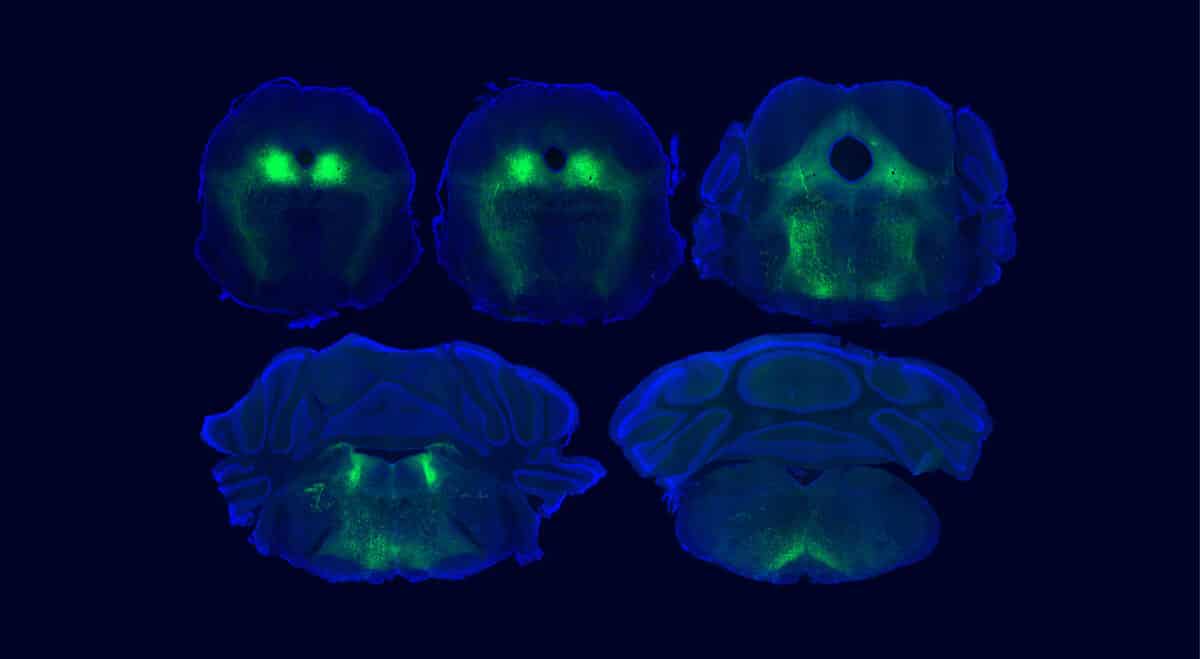
Fluorescent images of a key brain circuit involved in placebo pain relief in mice. Pain-regulating neurons located in the ventrolateral periaqueductal gray (vlPAG) are labeled in green, with their cell bodies visible as green spots and their wire-like axons extending to the brainstem to suppress pain. (Credit: Janie Chang-Weinberg)
Could Training Your Brain Before Surgery Replace Painkillers? Mouse Study Says Maybe
In A Nutshell
- Researchers identified the specific brain circuit that causes placebo pain relief in mice, tracing it from the thinking centers of the brain down to a deep brainstem hub that controls pain signals.
- The placebo effect in mice required a top-down command from the cortex to trigger the brainstem’s pain system, a step that morphine bypasses entirely, explaining a key difference between drug and placebo relief.
- Placebo pain relief generalized across different types of pain, meaning mice trained with heat pain also showed reduced sensitivity to mechanical pain, and vice versa.
- Conditioning healthy mice before an injury dramatically reduced their pain sensitivity afterward, raising the possibility that pre-injury placebo training could one day serve as a drug-free pain prevention strategy in humans.
Sugar pills contain no medicine, but they can still ease pain. That strange reality has puzzled doctors for decades. Now, a team of researchers has identified a specific brain circuit responsible for the placebo effect on pain in mice, tracing a pathway from the thinking part of the brain down to ancient circuitry that dials pain up or down. Even more surprising, training the brain to expect pain relief before an injury even occurs can dramatically reduce pain sensitivity afterward in mice, raising the possibility that with further research, placebo conditioning could one day offer a drug-free strategy against chronic pain.
Published in the journal Neuron, the study tackled a problem that has frustrated pain scientists for years. Brain scans in humans have shown which regions activate during placebo pain relief, but nobody could prove which circuits actually cause the relief. Human imaging can show correlation, but it cannot flip individual circuits on and off. So the research team rebuilt a well-known human placebo experiment in mice, where precision tools allowed them to manipulate specific brain cells one at a time.
What they found helps resolve a long-running debate about whether placebos work by changing how the brain interprets pain or by actually turning down pain signals at their source. The answer is both. And the mechanism requires a specific conversation between the brain’s prefrontal cortex, the region behind the forehead involved in thinking and decision-making, and a tiny structure deep in the brainstem, powered by the brain’s own natural painkilling chemicals.
How Scientists Trained Mice to Respond to Placebo Pain Relief
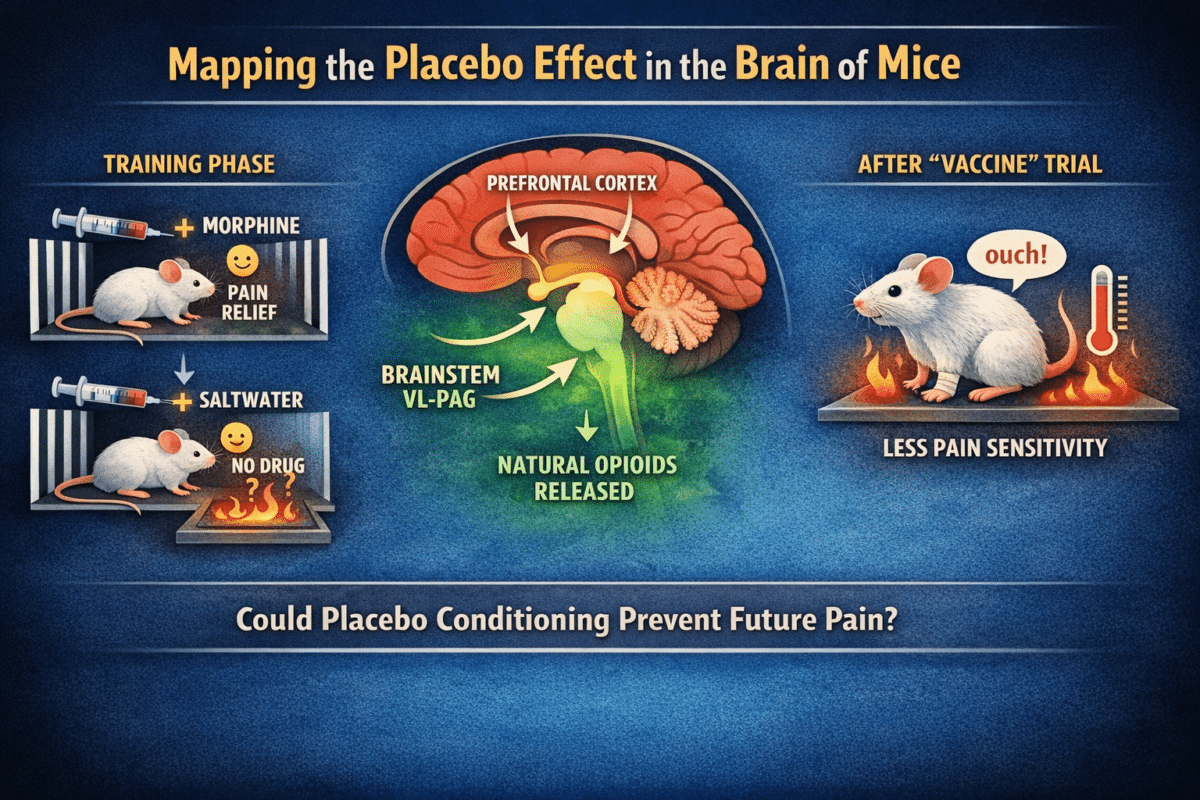
To create a placebo effect in mice, researchers paired morphine injections with a distinctive environment, a chamber with unique visual patterns and specific smells. After several rounds of receiving real pain relief from morphine in that setting, mice were given plain saltwater injections and placed back in the same chamber. Despite receiving no actual drug, the mice behaved as though medicated, tolerating a painful hot surface significantly longer than normal. The effect reached roughly 30 to 60 percent of the pain relief produced by real morphine.
The mice were not simply trained to hold still on a hot surface. When researchers tested escape behaviors never included in the training, placebo-conditioned mice showed reduced jumping and took longer to attempt their first escape, suggesting they were genuinely less bothered by the pain, not just suppressing a reflexive movement. Administering naloxone, a drug that blocks the brain’s opioid receptors, completely abolished placebo pain relief, confirming the brain’s own natural painkiller system was doing the work.
Mapping the Brain’s Placebo Circuit From Top to Bottom
With a reliable placebo effect established, the team began identifying which circuits were responsible. Using a genetic tool that permanently tags brain cells based on when they are active, the researchers labeled neurons in a key brainstem hub that fired during the placebo experience. Weeks later, they reactivated just those tagged neurons using a chemical switch, and the mice showed pain relief without any conditioning. Switching those neurons back on produced pain relief on its own.
When researchers silenced the neurons projecting from that brainstem hub to the next relay station, both morphine and placebo pain relief were completely blocked. This provided direct causal evidence that placebo pain relief runs through the same descending pain-control highway used by opioid drugs.
Using a modified virus that traces backward across brain connections, the team identified dense wiring into the hub from several frontal brain regions, areas associated with decision-making and expectation. Silencing those frontal pathways eliminated placebo pain relief entirely. Silencing the same pathways had no effect on morphine’s pain relief, which can bypass the cortex and act directly on lower brain structures. Placebo relief requires a top-down command from the cortex. Morphine does not.
To pinpoint exactly where those natural painkillers need to act, the team used a precision chemical tool that could be switched on inside a specific brain region with a flash of light. Triggering it in the brainstem hub just minutes before testing blocked the placebo effect entirely, confirming that local opioid release there is essential.
Placebo Conditioning as a Shield Against Post-Injury Pain
Among the study’s most surprising results was that placebo pain relief was not confined to the type of pain used during training. Mice conditioned with a hot surface showed reduced sensitivity to sharp mechanical pokes, and vice versa. All these generalized effects were blocked by naloxone, confirming they ran through the same mechanism.
Building on this, the team conditioned healthy, uninjured mice using the standard protocol, then injected an inflammatory agent into their hind paws to induce lasting pain sensitivity. Mice that had undergone placebo conditioning before the injury showed dramatically reduced pain sensitivity compared with controls. Pre-training the brain’s pain-suppression circuitry before injury created a lasting resilience against heightened pain developing later, though this approach remains untested in humans.
Chronic pain is among the hardest conditions to treat, and the opioid crisis has made clear the dangers of relying heavily on narcotic painkillers. If future studies confirm these mouse findings translate to people, deliberately engaging the brain’s placebo circuitry before surgeries or predictable injuries could one day offer a drug-free alternative. Understanding how that circuit gets activated in mice is a meaningful step toward finding out.
Disclaimer: This article is based on preclinical research conducted in mice. The findings have not been tested in humans and should not be interpreted as medical advice. Scientific results at this stage may or may not translate to human applications.
Paper Notes
Limitations
All mechanistic experiments were conducted exclusively in male mice because female mice showed weaker placebo responses during conditioning, a sex difference the researchers note mirrors findings from experimental pain studies in healthy human subjects. Whether the same circuits and chemical mechanisms operate identically in females remains unknown, which is a notable gap given that chronic pain disproportionately affects women. Additionally, while the conditioning approach was adapted from a validated human protocol, mice cannot receive verbal suggestions or conscious expectations the way human subjects can, meaning this model captures conditioning-driven placebo effects but may not fully represent the range of mechanisms underlying placebo pain relief in people. Activating the cortical inputs to the brainstem hub alone was not sufficient to produce pain relief, indicating the circuit requires additional signals, most likely the natural opioid release identified in the study, to function fully.
Funding and Disclosures
Support for this work came from the Rita Allen Foundation, the Esther A. & Joseph Klingenstein Fund and Simons Foundation, the Brain & Behavior Research Foundation, and several National Institutes of Health grants including RF1NS126073, R00DA034648, U01NS113295, U01NS120820, UM1MH136462, DP2GM140923, R01DA056581, R01DA056599, K99DA060979, and T32GM133351. Authors declared no competing interests.
Publication Details
Title: “Top-down control of the descending pain modulatory system drives multimodal placebo analgesia” | Authors: Giulia Livrizzi, Janie Chang-Weinberg, Desiree A. Johnson, Susan T. Lubejko, Jingzhu Liao, Blake A. Kimmey, Chunyang Dong, Yuan Li, Kevin T. Beier, Gregory Corder, Lin Tian, and Matthew R. Banghart. Livrizzi and Chang-Weinberg contributed equally as co-first authors; Banghart served as lead contact. | Affiliations: University of California San Diego, University of Pennsylvania, University of California Davis, University of California Irvine, and the Max Planck Florida Institute for Neuroscience. | Journal: Neuron, Volume 114, Pages 1–17, August 19, 2026. | DOI:https://doi.org/10.1016/j.neuron.2026.03.025 | Published by: Elsevier Inc. under a CC BY-NC open access license.







