
If muscles can be controlled better, the patient's independence in everyday activities, such as holding a smartphone, also improves. (Image: Heidelberg University Hospital)
HEIDELBERG, Germany — In a groundbreaking development that could change the lives of thousands of people with spinal cord injuries, European researchers have discovered that a specialized antibody treatment may help patients regain better control of their arm and hand movements. The finding is particularly significant for those suffering from incomplete spinal cord injuries in the neck area, offering hope for greater independence in daily activities.
The research, published in The Lancet Neurology, focused on an innovative antibody called NG 101 which targets a protein in the body that normally prevents damaged nerves from healing. Think of it like removing a roadblock that’s preventing repair crews from reaching a damaged highway – when the obstruction is cleared, repair work can finally begin.
The study, spanning 13 medical centers across Germany, Switzerland, the Czech Republic, and Spain, involved 126 participants between the ages of 18 and 70. All patients had suffered acute spinal cord injuries in their neck region, a condition known as tetraplegia, which affects both arm and hand function. This type of injury can make everyday tasks, from using a smartphone to feeding oneself, incredibly challenging.
To test the treatment’s effectiveness, researchers divided participants into two groups: 78 received the antibody treatment through direct injections into the spinal canal, while 48 received a placebo. Neither the patients nor their doctors knew who was receiving which treatment – a standard practice in clinical trials to ensure unbiased results.
The findings revealed an important distinction: while patients with complete spinal cord injuries (where all nerve communication is severed) didn’t show improvement, those with incomplete injuries (where some nerve pathways remain intact) demonstrated significant progress in regaining control over their paralyzed muscles. This improvement translated into a better ability to perform daily tasks independently.
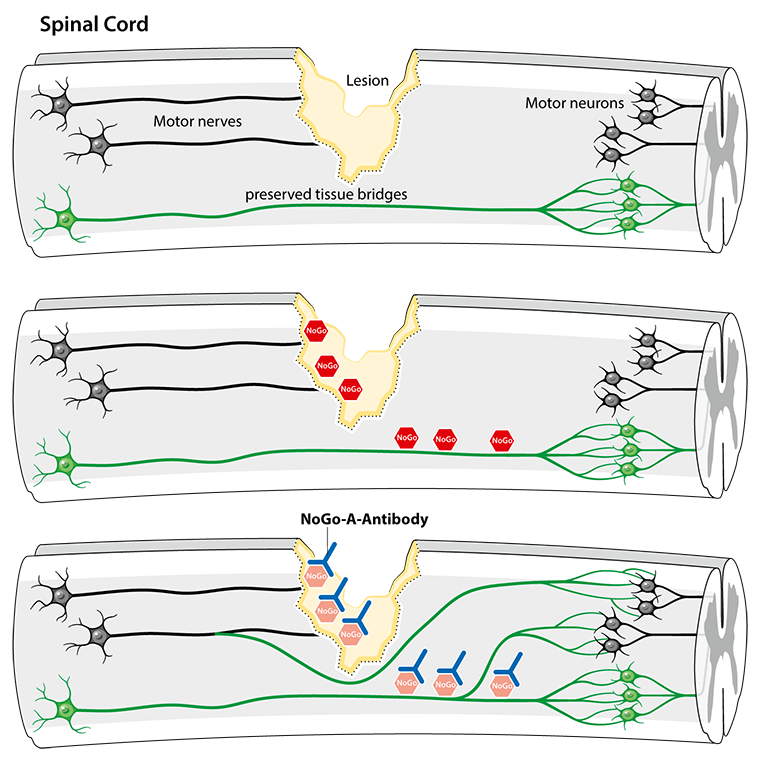
What makes this treatment unique is its approach to healing. The antibody works by neutralizing a protein called Nogo-A, which normally acts as a biological stop sign, preventing damaged nerve fibers from regenerating. By blocking this protein, the treatment essentially gives nerve fibers permission to grow and rebuild connections, potentially restoring lost function.
The treatment’s safety profile is particularly encouraging, with researchers reporting no significant side-effects so far. This positive outcome has prompted researchers to plan a follow-up study with an improved version of the antibody, scheduled to begin in December 2024.
For people living with spinal cord injuries, this development represents more than just a medical advancement – it offers hope for regaining independence and improving quality of life. As researchers move forward with their follow-up study, they’ll focus on patients most likely to benefit from the treatment based on these initial findings.
Paper Summary
Methodology
The study investigated the effects of a treatment called NG101 on patients with acute cervical spinal cord injuries. Conducted in multiple centers across several countries, it involved patients aged 18–70 who had suffered their injuries within 4–28 days prior to the start of the study. Participants were randomly divided to receive either NG101 or a placebo through intrathecal injections (administered into the spinal canal) six times over 4 weeks. Researchers, participants, and most of the study personnel did not know who received NG101 and who received the placebo, maintaining the study’s double-blind integrity. The treatment’s impact was primarily measured by changes in upper extremity motor scores after 6 months.
Key Results
The main finding was that NG101 did not significantly enhance motor function compared to the placebo when evaluating all patients together. Both groups showed similar changes in motor scores after 6 months. However, a closer look at different subgroups within the study revealed that patients with incomplete motor injuries (a less severe type of spinal injury) might have benefited from the treatment, showing potential improvements that could be explored in future research.
Study Limitations
The study’s size and duration might have restricted the ability to observe significant benefits from NG101. The selection of participants with a wide range of injury severities might have diluted the potential effects of the treatment. Variability in injury characteristics and responses to treatment among patients could have influenced the results and the ability to generalize findings.
Discussion & Takeaways
The results suggest that while NG101 was safe and well-tolerated, it did not provide a significant benefit over a placebo for all participants. This finding contrasts with some promising results in specific subgroups, indicating that NG101 might still hold potential for certain patients, particularly those with incomplete injuries. This calls for further studies to refine treatment targeting and to understand better who might benefit the most from such therapies.
Funding & Disclosures
The study received support from various European institutions, including EU program Horizon2020 and the Swiss State Secretariat for Education, Research and Innovation. None of the funders had any role in the design, execution, or analysis of the study, ensuring the impartiality of the findings.







