
3D rendering of the skeleton of Graulia branchiodonta. The auditory organ includes the bony wings (red) on the ossified lung (white) which transmitted sound vibrations to the inner ear (not shown) located in the prootic bone in the skull (pink) Credit: © L. Manuelli–MHNG
In A Nutshell
- Scientists found evidence that ancient coelacanths may have used their bony lungs to detect sound vibrations and transmit them to the inner ear, giving the lung a dual role as both a breathing and hearing organ.
- Two 240-million-year-old fossil species revealed never-before-seen bony projections on their lung chambers, which researchers believe may have helped relay sound toward the brain.
- The auditory system appears to have been a consistent feature of the coelacanth lineage for hundreds of millions of years, eventually disappearing as the fish adapted to deeper ocean environments and the lung shrank.
- If early relatives of coelacanths shared this ability, hearing may have evolved in fish long before the first vertebrates developed eardrums on land.
Long before the first creature hauled itself onto land, an ancient fish was apparently doing something no one expected with its lungs: listening.
New research has found evidence that ancient coelacanths (pronounced SEE-la-kanths) may have picked up underwater sound vibrations through their ossified lungs and transmitted them toward the inner ear, giving the organ a dual role as both a breathing structure and a hearing device. These fish were part of the lineage that also gave rise to land animals, which means the discovery is a much bigger deal than one ancient species.
Published in Communications Biology, the study draws on CT scans of fossils dating back 240 million years alongside specimens of the living coelacanth Latimeria chalumnae, one of two coelacanth species alive today. What the researchers found may push back the origin of hearing in vertebrates further than anyone had previously considered.
How the Ancient Coelacanth Lung May Have Doubled as a Hearing Organ
Sound travels through water as a wave of pressure. Some fish detect that pressure using gas-filled chambers in their bodies, which flex in response to those pressure waves and relay the signal to the inner ear. Species like catfish and goldfish accomplish this through a chain of small bones called the Weberian apparatus, a kind of built-in sound relay. In ancient coelacanths, the researchers believe, the lung itself played that role.
Scanning two fossil species, Graulia branchiodonta and the newly described Loreleia eucingulata, the team found a previously unnoticed pair of bony projections at the front of each animal’s ossified lung chambers. Dubbed chamber wings, these ridges extended upward and anchored to the outer sheath of the notochord, the stiff rod-like structure that ran along the spine of early vertebrates. The researchers propose that sound-pressure vibrations could have traveled from the lung through those projections, along the notochord or surrounding soft tissue, and into the inner ear through a network of fluid-filled canals known as the perilymphatic system.
That canal network already existed in the living Latimeria chalumnae, but for decades no one could explain what it was for. In a fish whose lung has shrunk to a small, mostly nonfunctional remnant, there was no obvious way for sound to have reached it. Now the picture is clearer. In ancient coelacanths, the lung was apparently large enough, rigid enough, and close enough to the inner ear to plausibly function as a sound receiver. As coelacanths adapted to deeper, darker ocean environments over millions of years and no longer needed to surface for air, the lung shrank, the sound pathway filled in with connective tissue, and the hearing function faded away.
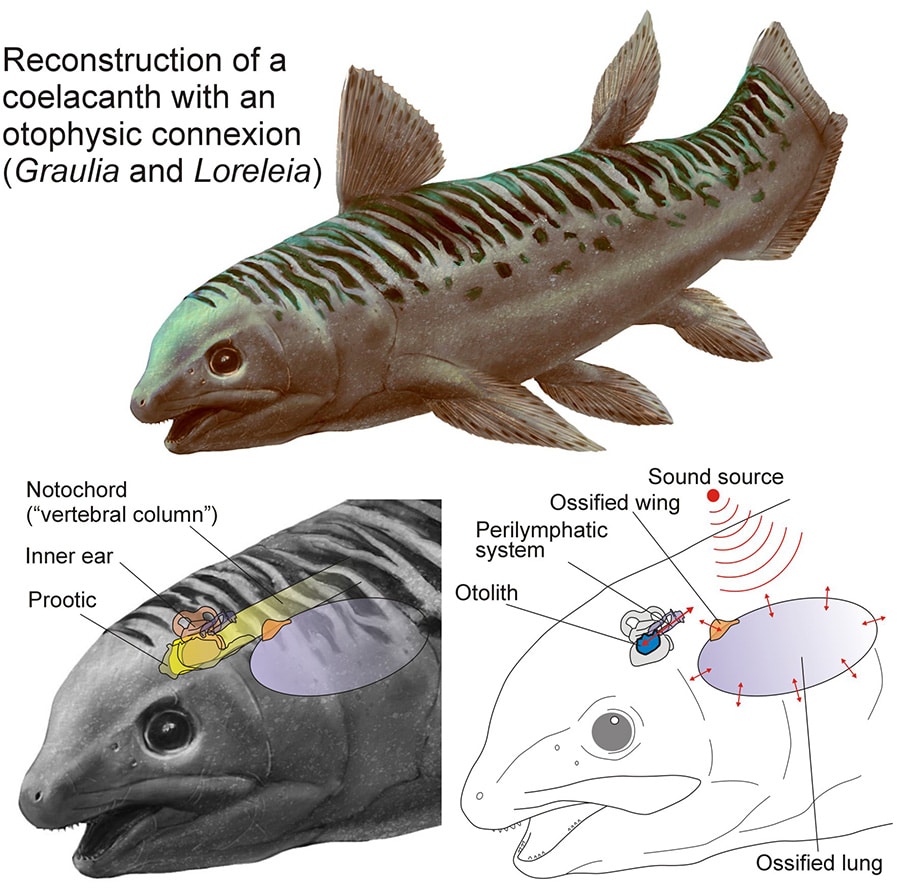
© A. Beneteau (reconstruction), L. Cavin (diagram) – MHNG
What Synchrotron Scanning Revealed Inside 240-Million-Year-Old Fossils
Reconstructing soft-tissue pathways in ancient fossils is no small feat. To do it, the team used synchrotron phase-contrast microCT scanning, which fires very powerful X-rays through a specimen to build a three-dimensional picture of its internal anatomy without disturbing a single bone. Scanning took place at the European Synchrotron and Radiation Facility in Grenoble, France, using fossils pulled from Middle Triassic rock in eastern France.
Graulia had three lung chambers and Loreleia had two, though incomplete preservation in both specimens leaves open the question of whether those chambers were truly separate or connected to one another. Both species carried the chamber wings at the front, a feature never before documented in any coelacanth. Alongside the fossil work, the researchers mapped the perilymphatic canal system in multiple specimens of Latimeria chalumnae, from newborn pups to an adult male, and found the anatomy consistent across all of them. Critically, a closely related arrangement appeared in Diplocercides kayseri, a coelacanth from the Late Devonian period roughly 380 million years ago, suggesting the system was not a one-off. It appears to have been a defining feature of the coelacanth lineage for an incredibly long stretch of time.
Two sensory patches in the inner ear, the basilar papilla and the amphibian papilla, are connected to that same canal network in coelacanths. Both patches also drive sound detection in amphibians, a link that points toward a shared evolutionary origin well before the first vertebrate stepped onto dry ground.
What Ancient Coelacanth Hearing Means for the Evolution of All Land Animals
Coelacanths belong to the sarcopterygians, the group of lobe-finned fishes whose lineage eventually produced amphibians, reptiles, birds, and mammals. That ancestry makes the hearing question a lot bigger than one ancient fish.
If ancient coelacanths were detecting sound pressure, possibly even including airborne sound, through their lungs, other early sarcopterygians may have had a similar capability. As the researchers write, “early sarcopterygians may have possessed inner ear organs capable of detecting airborne sound, without tympanic specialization, prior to the transition to land.” Tympanic specialization means an eardrum, and the takeaway is that the ability to hear may have predated that structure by a wide margin.
In other words, the ear in its most primitive form may not have been a land adaptation at all. It may have originated in fish, built around a lung that had learned, over hundreds of millions of years, to listen.
Paper Notes
Limitations
Because soft tissue almost never survives in ancient rock, the proposed connection between the chamber wings and the inner ear cannot be directly observed in the fossil species. The pathway by which vibrations may have traveled, whether through the notochord, surrounding tissue, or a structure resembling the bronchial columella found in tadpoles, remains uncertain. Incomplete preservation also makes it unclear whether the lung chambers in the fossil species were truly separate or interconnected. Both Triassic species are known only from juvenile specimens, so how these structures may have developed in adults is unknown. Direct examination of the Triassic species’ inner ear anatomy was also not possible due to incomplete bone development in the relevant skull regions.
Funding and Disclosures
Funding was provided by the Swiss National Science Foundation (grant 207903). Synchrotron imaging was conducted at the European Synchrotron and Radiation Facility in Grenoble, France. Specimens were drawn from the American Museum of Natural History, the Muséum National d’Histoire Naturelle, the Zoological Museum of the University of Copenhagen, and other institutions. All authors declare no competing interests.
Publication Details
“A dual respiratory and auditory function for the coelacanth lung” was authored by Luigi Manuelli, Gaël Clément, Marc Herbin, Bernd Fritzsch, Per E. Ahlberg, Kathleen Dollman, and Lionel Cavin, representing institutions including the Natural History Museum of Geneva, the University of Geneva, the Muséum National d’Histoire Naturelle (Paris), the University of Nebraska Medical Center, Uppsala University, and the European Synchrotron and Radiation Facility. Published in Communications Biology, a Nature Portfolio journal, in 2026. DOI: https://doi.org/10.1038/s42003-026-09708-6







