
(© Ivelin Radkov - stock.adobe.com)
In A Nutshell
- Mice with advanced Alzheimer’s fully recovered after treatment with P7C3-A20, a compound that restores cellular energy balance, performing as well as healthy mice on memory tests
- Two different disease models showed reversal, one driven by amyloid plaques, the other by tau tangles, suggesting the approach targets fundamental brain repair mechanisms
- The key is NAD+ homeostasis; when cells can’t maintain proper energy levels, Alzheimer’s progresses. Restoring that balance reversed brain damage and memory loss in elderly mice
- Human relevance identified: Researchers found 46 proteins that change the same way in human Alzheimer’s brains and were normalized by treatment in mice, offering potential drug targets
For over a century, doctors and scientists have accepted one unchanging truth: Alzheimer’s disease cannot be reversed. Once the brain deteriorates into dementia, there’s no coming back. That assumption just collapsed.
Researchers at Case Western Reserve University have done what generations of scientists thought impossible. They reversed advanced Alzheimer’s disease in mice. Not slowed it. Not stabilized it. Reversed it. Older mice with memory problems, in two different Alzheimer’s-like mouse models (one focused on amyloid plaques, one on tau tangles), regained normal performance on memory tests after treatment with an experimental compound called P7C3-A20.
The mice were elderly animals with clear brain pathology and memory loss. In one model, amyloid plaques were prominent. In the other, tau-related disease features dominated. Their brains healed anyway.
Treatments have focused on slowing decline or managing symptoms, never on turning back the clock. This new work, published in Cell Reports Medicine, upends that approach.
How Did Scientists Reverse Alzheimer’s Disease in Mice?
Researchers used two different mouse models that mimic human Alzheimer’s disease. The first, called 5xFAD mice, develops amyloid plaques similar to those in human patients. The second, PS19 mice, develops tau tangles, another hallmark of the disease. Both types develop memory problems and brain damage that look a lot like human Alzheimer’s.
Scientists divided mice into groups starting at either 2 months old (mid-stage disease) or 6 months old (advanced disease). The mid-stage group received daily injections of either P7C3-A20 or a placebo until 6 months old. The advanced-disease group received treatment until 12 months old. In a separate tau tangle model, 11-month-old mice received treatment for one month. Each treatment group included both male and female mice.
Mice with advanced Alzheimer’s that received P7C3-A20 performed as well on memory tests as healthy mice. In the Morris water maze, a standard test where mice must remember the location of a hidden platform, treated Alzheimer’s mice found the platform as quickly as normal mice. Untreated Alzheimer’s mice struggled. Similar improvements showed up in object recognition tests and other measures of thinking ability.
But the changes went far beyond behavior. Brain tissue analysis showed broad improvements across many Alzheimer’s-linked measures, including reduced plaque accumulation, tau-related changes, blood-brain barrier damage, and inflammation. Most remarkably, the brains began generating new neurons again, a process that normally shuts down in Alzheimer’s.
What Causes Alzheimer’s Disease? The NAD+ Connection
The key to this reversal lies in NAD+, a molecule that works as universal energy currency in cells. Every cell needs NAD+ to power repair processes, fight oxidative stress, and keep DNA intact. When NAD+ levels fall, cells can’t keep up with constant maintenance needs.
The research team discovered that Alzheimer’s severity tracks with disrupted NAD+ homeostasis in both mice and humans. When they examined human brain tissue, people who had died with Alzheimer’s showed disturbed NAD+ metabolism.
But some elderly people had Alzheimer’s-like changes in the brain at autopsy yet had never developed dementia while alive. In this study’s analysis, these individuals showed gene expression patterns suggesting preserved NAD+ homeostasis despite their brain pathology.
P7C3-A20 doesn’t deliver NAD+ directly. Instead, it helps cells restore NAD+ balance when they are under stress (similar to fixing a leaky bucket rather than just pouring more water in).
The treatment also normalized a blood marker tied to Alzheimer’s in the mice. This marker, p-tau217, is used in humans to help diagnose the disease.
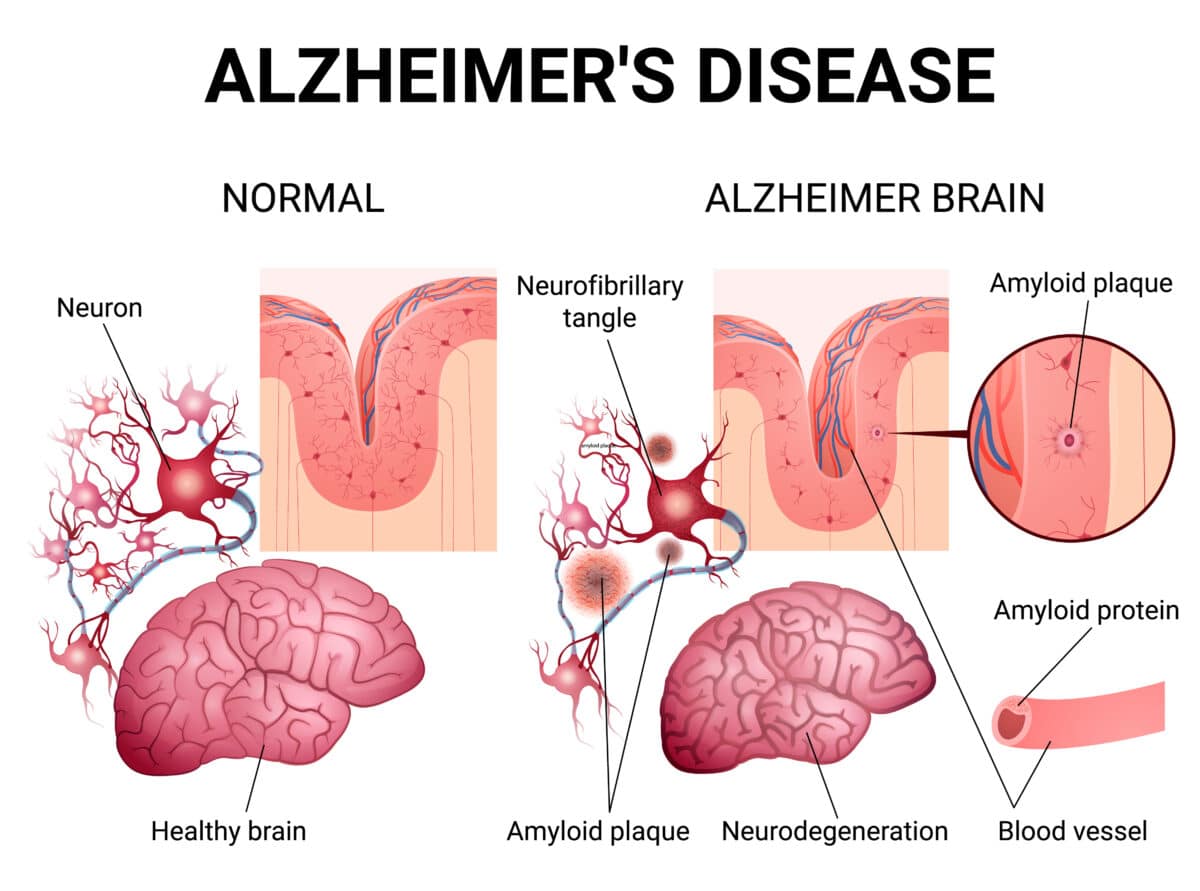
Can Alzheimer’s Disease Be Reversed in Humans?
To identify which aspects of the mouse findings might translate to human patients, researchers compared protein changes in the treated mice with databases of human Alzheimer’s brain tissue. They found 46 proteins that changed the same way in both human and mouse Alzheimer’s brains. All 46 returned to normal with P7C3-A20 treatment.
These proteins affect how cells handle stress, produce energy, and manage inflammation. The finding suggests potential drug targets for treating human Alzheimer’s.
Why Reversing Alzheimer’s Disease Changes Everything
This research overturns what we thought we knew about Alzheimer’s disease and what’s possible once it takes hold. For decades, treatments have aimed only to slow decline, not reverse it.
The authors propose a new way of understanding Alzheimer’s. Rather than being caused by a single toxic trigger like amyloid or tau, the disease may result from the brain’s repair systems breaking down. When cells accumulate too much damage, they can’t keep up with repairs. NAD+ sits at the center of this repair network. Restoring it can reverse the damage.
What was impossible for over a century may not be impossible at all. The question now is how to translate these findings to humans. Recovery from advanced Alzheimer’s is now documented reality in mice and a legitimate target for human medicine.
Disclaimer: This article is for informational purposes only and should not be interpreted as medical advice. Anyone concerned about Alzheimer’s disease should consult qualified healthcare professionals.
Paper Notes
Limitations
Several important caveats apply to these findings. The study relied entirely on genetically engineered mouse models of Alzheimer’s disease. While these models reproduce many features of human Alzheimer’s, the vast majority of human cases are sporadic (arising from interactions between aging, genetics, and environmental factors) rather than from single gene mutations.
The human brain tissue analyzed came from postmortem samples, which show end-stage disease and cannot establish causality. While the matching signals across non-demented individuals with Alzheimer’s pathology, actual Alzheimer’s patients, and P7C3-A20-treated mice suggest that impaired NAD+ homeostasis contributes to disease, the researchers acknowledge they haven’t identified a single specific driver of NAD+ homeostasis disruption.
The study proposes that multiple forms of escalating cellular damage work together to create chronic, unsustainable demand for repair that dysregulates NAD+ homeostasis, but exactly how this cascade starts and whether all Alzheimer’s cases follow the same pattern remains unclear.
Funding and Disclosures
This research was supported by numerous sources including the Valour Foundation, the Wichita Foundation, Department of Veterans Affairs Merit Award I01BX005976, the American Heart Association and Paul Allen Foundation Initiative in Brain Health and Cognitive Impairment (19PABH134580006), and multiple grants from the National Institutes of Health including NIH/NIA R01AG071512, RO1AG066707, 1U01AG073323, and others. Additional funding came from the Alzheimer’s Association, the Rainwater Charitable Foundation, the National Research Foundation of Korea, BrightFocus Foundation, and other organizations. The research also utilized the Louis Stokes VA Medical Center resources and facilities.
The authors note that views expressed are those of the authors and do not reflect the position or policy of the Department of Veterans Affairs of the United States government. Several authors hold related patents, and the senior author is a cofounder of a brain health company.
Publication Details
Title: Pharmacologic reversal of advanced Alzheimer’s disease in mice and identification of potential therapeutic nodes in human brain
Authors: Kalyani Chaubey, Edwin Vázquez-Rosa, Sunil Jamuna Tripathi, Min-Kyoo Shin, Youngmin Yu, Matasha Dhar, Suwarna Chakraborty, Mai Yamakawa, Xinming Wang, Preethy S. Sridharan, Emiko Miller, Zea Bud, Sofia G. Corella, Sarah Barker, Salvatore G. Caradonna, Yeojung Koh, Kathryn Franke, Coral J. Cintrón-Pérez, Sophia Rose, and others (full list available in paper), Jung-A.A. Woo, David E. Kang, Bindu D. Paul, Andrew A. Pieper
Journal: Cell Reports Medicine | Volume and Issue: Volume 7, Article 102535 | Publication Date: January 20, 2026 (published online) | DOI: 10.1016/j.xcrm.2025.102535 | Correspondence: [email protected]
The research was conducted at institutions including Case Western Reserve University, Johns Hopkins University School of Medicine, Seoul National University, University of California Los Angeles, Northwestern University, and other collaborating institutions. Full datasets are available at https://zenodo.org/records/17455622.







