
(Photo by Steven HWG on Unsplash)
In A Nutshell
- Researchers built a compact, battery-powered device that reads brain signals to control a robotic walking brace while simultaneously sending artificial leg sensations back to the brain in real time.
- Placing electrodes deep between the brain’s two halves, directly over the leg areas, produced stronger signals and allowed matched tingling sensations to be delivered to both legs, a first for this type of system.
- A single epilepsy patient tested the device over nine days, achieving about 93 percent accuracy in detecting artificial leg sensations, with no adverse events.
- The system has not yet been tested in people with spinal cord injuries, and significant development remains before any clinical use, though the team reports receiving an FDA Investigational Device Exemption to pursue that next step.
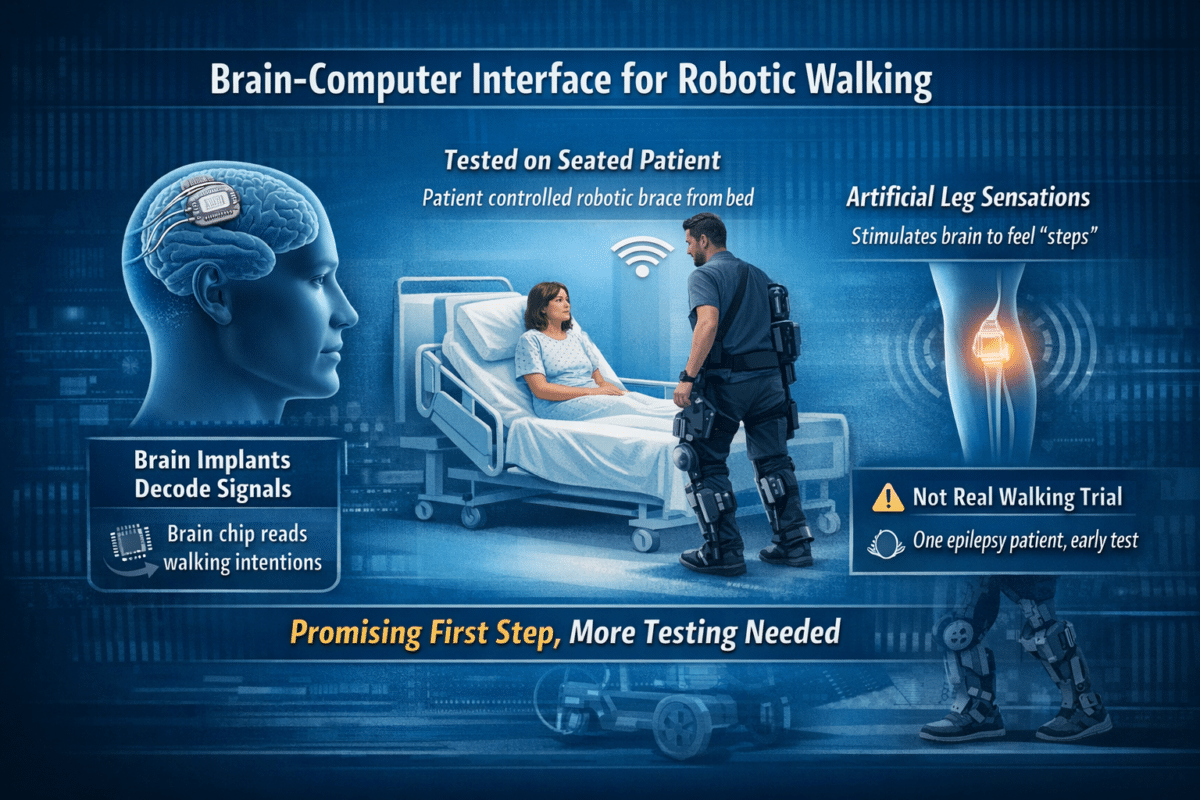
Seated in a hospital bed, a 50-year-old woman made stepping motions with her legs. A robotic brace across the room responded, taking steps in real time. Each time a leg swung forward, she felt a tingling sensation in the matching limb, delivered directly to her brain. No wires ran between her and the brace. No external computer processed her thoughts. A device the size of a small circuit board, powered by a rechargeable battery, achieved all that.
That is the core of a new study published in the journal Brain Stimulation by researchers at the University of California, Irvine, the University of Southern California, and Caltech. For people with severe spinal cord injuries, who typically lose both movement and sensation in their legs, the research points toward something that has not existed before: a single compact device that reads the brain’s walking commands and sends artificial sensory feedback back in return, simultaneously.
Previous brain-controlled walking systems only ran in one direction. The brain sent a signal, the machine moved, and the user felt nothing. That gap matters more than it might seem. Sensation is not just a bonus feature of walking. Without it, gait slows, balance suffers, and the risk of falling goes up. By closing that loop, this device is attempting to restore something closer to how the body actually works.
A Smarter Place to Listen in the Brain
One key design decision separated this system from earlier attempts. Most brain implants for movement sit on the outer surface of the brain, which is easier to access surgically but picks up mostly hand and arm signals. Leg-related activity is tucked deeper, in the groove running between the brain’s two halves. By placing electrode grids there, directly over the leg movement and sensory areas on both sides, the team captured stronger and more reliable signals than prior systems achieved. Earlier work by the same group, using the outer surface, managed only one-sided sensory feedback. This version delivered matched sensations to both legs, a first for this kind of system.
When the participant made stepping motions, onboard chips read her brain activity and wirelessly commanded the robotic brace to step. As the brace swung each leg, ankle sensors detected the motion and triggered a small electrical pulse to the opposite side of her brain’s sensory region, left brain for right leg, right brain for left. She felt a tingling in the corresponding leg with each step.
One Participant, Nine Days, Striking Results
Recruiting for this study was not straightforward. Getting electrode grids into the deep interhemispheric region of the brain is rare, done only in certain epilepsy patients undergoing pre-surgical evaluation. That is why only one participant was enrolled. She was not paralyzed, which means the study cannot yet confirm the system will work the same way in people with spinal cord injuries, whose brain signals may have weakened or reorganized after years without use. The researchers note that prior studies suggest those signals can be retrained over time, but it remains to be tested directly.
Over nine days, the team mapped her brain, identified the 15 electrodes with the most useful leg-movement signals, and dialed in stimulation settings that reliably produced distinct tingling sensations in each leg. Then they ran a blind test: a researcher walked an unknown number of steps out of her sight, each step triggering one artificial sensation. She counted what she felt using a hand counter. Her accuracy came out to 100 percent for right-leg sensations and 86 percent for left, about 93 percent combined. The odds of hitting that by guessing were less than one in a million.
For the full system test, she completed 10 runs over two days, each about two and a half minutes long. Her brain signals controlled the brace, worn by a researcher for safety reasons, while artificial sensations were delivered back to her with each step. Performance was strong across the board and improved on day two. In all 10 runs, she confirmed the sensations matched the correct leg and said the feedback made the task easier. No adverse events occurred.
What Has to Happen Before This Reaches Patients
People with complete spinal cord injuries, the most severe classification, currently have less than a 5 percent chance of regaining independent walking with existing treatments. Whether a device like this one could move that number is still an open question. The team has said, anecdotally, that the system received an FDA Investigational Device Exemption to begin testing in spinal cord injury patients, which is the necessary next step. The device would also need to be miniaturized further before it could be fully implanted, though the researchers point out that a one-directional version has already reached a size comparable to existing implantable neural devices.
For now, what exists is proof that the concept works. A brain can be read, a robotic exoskeleton can be driven, and artificial leg sensations can be delivered back, all by one small device, all at once.
Disclaimer: This article is based on a peer-reviewed proof-of-concept study and is intended for general informational purposes only. The technology described has not been approved for clinical use and has not yet been tested in people with spinal cord injuries. Nothing in this article should be construed as medical advice. Readers with questions about spinal cord injury treatment options should consult a qualified medical professional.
Paper Notes
Limitations
The study enrolled a single participant, a 50-year-old woman with epilepsy who did not have a spinal cord injury. Bilateral interhemispheric electrode placement of the kind used here is extremely rare, which is why no additional participants could be recruited. Because the participant was able-bodied, it is unknown whether the system would perform comparably in paralyzed individuals, whose leg motor cortex activity may be diminished or reorganized following injury or prolonged disuse. The motor task involved seated stepping motions rather than actual standing or walking, and the robotic brace was worn by a researcher, not the participant. Sensory feedback covered only the swing phase of each step, and the study did not formally assess how well participants could integrate that feedback into motor control. The 3.5-second lag between movement intent and brace actuation may need to be reduced for practical use. All findings are preliminary and require replication in larger groups, including people with spinal cord injuries.
Funding and Disclosures
The work was supported by the National Science Foundation, grant number 1646275. Several authors declared potential competing interests. Drs. Do, Nenadic, Liu, Heydari, and Andersen jointly hold patents and government grant funding directly related to the work. Dr. Kellis conducted the reported work prior to joining Blackrock Neurotech, and Dr. Sohn conducted the work prior to joining Abbott Laboratories. The study was approved by the Institutional Review Boards of the University of California, Irvine, and Rancho Los Amigos National Rehabilitation Center.
Publication Details
The study, “Real-time brain-computer interface control of walking exoskeleton with bilateral sensory feedback,” was authored by Jeffrey Lim, Po T. Wang, Won Joon Sohn, Derrick Lin, Shravan Thaploo, Luke Bashford, David A. Bjanes, Angelica Nguyen, Hui Gong, Michelle Armacost, Susan J. Shaw, Spencer Kellis, Brian Lee, Darrin J. Lee, Payam Heydari, Richard A. Andersen, Zoran Nenadic, Charles Y. Liu, and An H. Do. It was published in Brain Stimulation, Volume 19, 2026, article 103065. DOI: 10.1016/j.brs.2026.103065. Available online February 28, 2026.







