
Getty Images For Unsplash+
Late 60s May Be a Critical Age for Alzheimer’s Warning Signs
In A Nutshell
- A Mayo Clinic study of more than 2,000 people found that multiple biological markers linked to Alzheimer’s disease begin changing more rapidly in the late 60s to early 70s.
- Key signals, including brain inflammation, nerve damage, and tau protein levels, all showed distinct acceleration points clustered around age 68 to 71, while amyloid-related changes appeared as early as age 62.
- A blood test for amyloid gave inconsistent results across testing methods, raising caution about its reliability as a standalone screening tool.
- Researchers say the findings could help refine when Alzheimer’s monitoring and screening might be most informative, especially if ongoing preventive drug trials prove successful.
Late midlife is often spent planning retirement and spoiling grandchildren. A large Mayo Clinic study now suggests that same stretch of life may also be when several biological signals linked to Alzheimer’s disease begin changing more quickly, at least at the population level. Published in Alzheimer’s & Dementia, the study analyzed data from more than 2,000 people. Researchers pinpointed specific ages when key biological markers tied to Alzheimer’s risk accelerate, offering new clues about when screening and monitoring might be most informative.
Researchers drew from the Mayo Clinic Study of Aging, a long-running project that tracks brain health among residents of Olmsted County, Minnesota. Using statistical models designed to detect “breakpoints,” distinct ages at which biological markers shift from a gradual drift to a steeper climb or drop, the team found several markers changing course sharply in the late 60s to early 70s. As blood-based Alzheimer’s biomarkers become more important in research and clinical decision-making, a practical question grows louder: when should those tests actually be ordered?
How Alzheimer’s Blood Tests Could Reshape Early Screening
Drawing from the general population rather than a clinic full of people already suspected of having problems, the Mayo Clinic Study of Aging offers a clearer picture of what happens during ordinary aging. Among the 2,082 participants in this analysis, the median age was 71, and 54% were male. The vast majority, 87%, had normal thinking abilities at the time of evaluation, while 11% had mild memory problems and just over 1% had been diagnosed with dementia.
Participants gave blood samples after an overnight fast, tested for proteins tied to Alzheimer’s-related damage: a fragment linked to amyloid plaques (the Aβ42/40 ratio), a tau protein associated with tangled nerve fibers (p-tau181), a brain inflammation marker called glial fibrillary acidic protein (GFAP), and neurofilament light chain (NfL), a sign of nerve cell injury. A smaller group of 462 participants also had blood analyzed using a second, highly precise lab method to cross-check results. Many participants additionally underwent brain imaging and cognitive testing across memory, language, decision-making, and spatial reasoning.
Rather than fitting a single straight line through the data, researchers used models that detect a shift in direction at a particular age, a point where the slope becomes noticeably steeper.
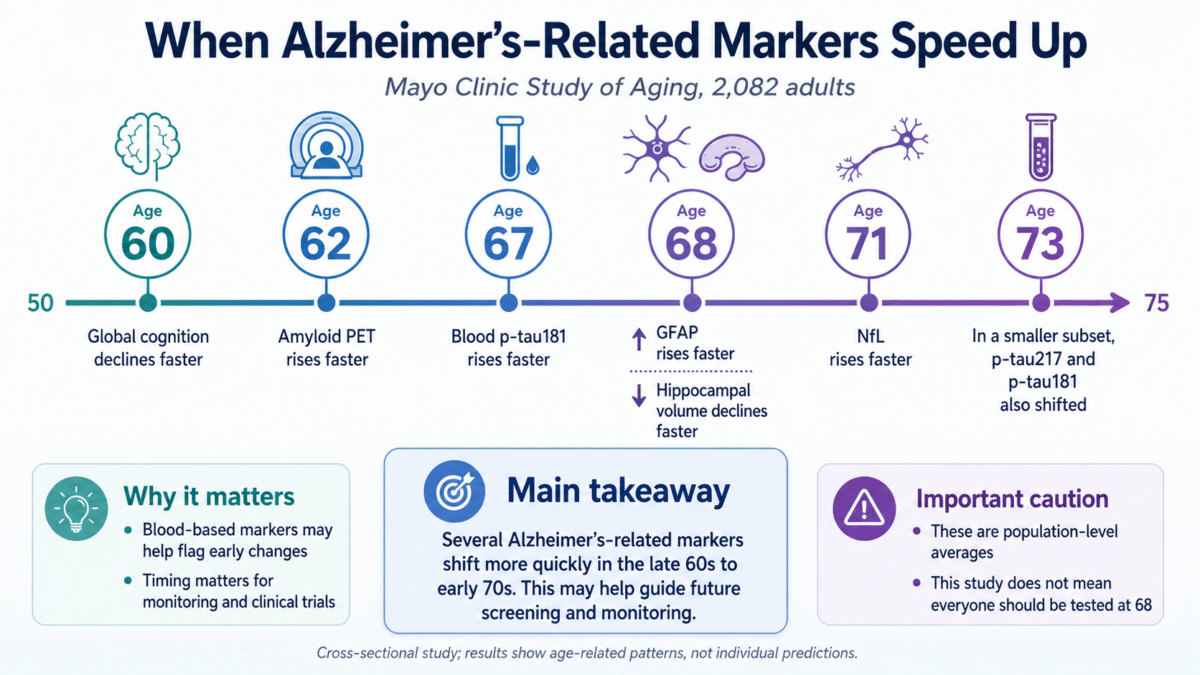
Age 68: When Alzheimer’s Warning Signs Begin Accelerating
A consistent pattern emerged across markers. Brain inflammation (GFAP) broke upward at roughly age 68. Nerve-damage signal (NfL) shifted around age 71. A tau marker (p-tau181) turned a corner near age 67. Brain amyloid measured by PET scans showed a breakpoint at about age 62. Hippocampal volume began declining faster at approximately age 68. Overall thinking ability started dropping more steeply around age 60.
In the smaller group tested with the more precise lab method, two tau-related blood markers, p-tau217 and p-tau181, both showed breakpoints at approximately age 73, supporting the general pattern.
Tau PET scans, which produce direct images of tau deposits in the brain, did not show a clear breakpoint. Only 628 participants had this type of scan, which may have limited the ability to detect a shift.
Taken together, these breakpoints tell a coherent story. Amyloid-related changes and cognitive shifts appeared earliest, roughly in the late 50s to early 60s. Markers of tau buildup, brain inflammation, and nerve damage followed in the late 60s to early 70s. This sequence aligns with what scientists have long theorized about how Alzheimer’s unfolds: amyloid accumulates first, tau follows, and brain-cell injury comes later.
“Breakpoints cluster near late midlife, suggesting windows for screening and monitoring,” the researchers wrote.
Why the Late 60s Are a Key Window for Alzheimer’s Monitoring
If blood-based Alzheimer’s tests are going to inform screening and trial enrollment, knowing when to start testing matters. Prior research has shown that amyloid deposits can reach abnormal levels roughly 17 years before dementia onset, while brain shrinkage and memory problems emerge much closer to diagnosis. These breakpoints are consistent with that timeline.
One wrinkle worth noting: the blood-based amyloid test gave wildly inconsistent results depending on which testing method was used, with the estimated breakpoint age shifting dramatically across platforms. One version of the test produced no detectable breakpoint at all. That inconsistency warrants further study before clinical guidance can be built around it.
Several boundaries apply to what this study can tell us. All breakpoints are population-level averages from a single snapshot in time, not a tracking of the same individuals over years. An earlier breakpoint for cognition should not be read as cognitive decline preceding biological changes within any one person. Participants were also drawn from a single, relatively homogeneous county in Minnesota, limiting how well the findings apply to more diverse populations.
Still, for a field searching for the right moment to intervene against Alzheimer’s, narrowing down an age range when multiple biological signals shift together is meaningful progress. If trials targeting early amyloid or tau buildup prove successful, these breakpoints could help doctors identify who should be tested and when, opening a window for closer monitoring before symptoms become more obvious. Late midlife, it turns out, may be the moment the brain most urgently needs a closer look.
Disclaimer: This study reflects population-level findings and is not a substitute for medical advice. Anyone with concerns about Alzheimer’s risk or cognitive health should speak with a qualified healthcare provider.
Paper Notes
Limitations
Several limitations should be considered when interpreting this research. The study used cross-sectional data, meaning each participant was measured at a single point in time rather than followed over years. Breakpoints therefore reflect population-level patterns and do not map onto disease progression within any one individual. The cohort was overwhelmingly cognitively healthy, with only 1.1% diagnosed with dementia, which may limit the relevance of the findings to later-stage disease. Participants were drawn from Olmsted County, Minnesota, limiting generalizability to more diverse populations. Only 628 participants had tau PET scans, which may have limited statistical power to detect a breakpoint for that marker. Global cognition scores were missing for approximately 6% of participants due to incomplete testing at the time of the visit, related to practical factors such as fatigue or scheduling constraints rather than cognitive status. Breakpoint estimates for the blood-based amyloid marker varied substantially across subsamples and testing platforms, and the researchers called for further replication before those estimates can inform clinical guidance.
Funding and Disclosures
This work was supported by National Institute on Aging grants R01AG34676, RF1AG069052, U01AG006786, P30AG0622677, R01AG041851, R01AG058738, and the GHR Foundation. Avid Radiopharmaceuticals, a subsidiary of Eli Lilly, provided precursor material for the tau PET tracer but did not provide direct funding and was not involved in data analysis or interpretation. Multiple authors reported institutional research support, consulting relationships, or advisory roles with companies including Roche, Eli Lilly, Eisai, Novo Nordisk, Biogen, Siemens Healthineers, and others. Full conflict of interest disclosures are available in the paper’s supporting information.
Publication Details
Title: Breakpoints in Alzheimer’s disease biomarkers and cognition across the aging spectrum: The Mayo Clinic Study of Aging | Authors: Mingzhao Hu, David S. Knopman, Terry Therneau, Angela J. Fought, Ekaterina Hofrenning, Val J. Lowe, Ronald C. Petersen, Alicia Algeciras-Schimnich, Clifford R. Jack Jr., Nikki H. Stricker, Michelle M. Mielke, Prashanthi Vemuri, Jonathan Graff-Radford | Journal: Alzheimer’s & Dementia | DOI: 10.1002/alz.71227 | Corresponding Author: Jonathan Graff-Radford, Mayo Clinic, Rochester, Minnesota







