
(Image by Andrii Yalanskyi on Shutterstock)
In A Nutshell
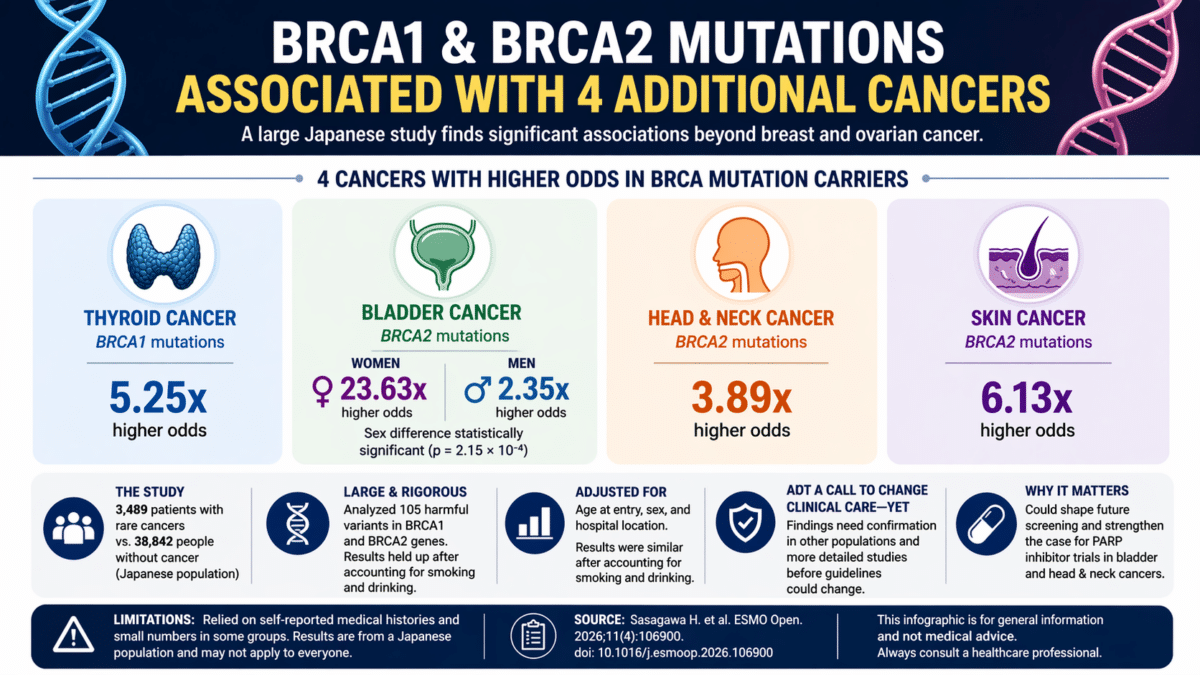
- A new Japanese study found BRCA1 and BRCA2 mutations were linked to four less common cancers: thyroid, bladder, head and neck, and skin cancer.
- The study was large, but it still had limits, including self-reported medical histories and small numbers in some subgroups.
- The strongest sex difference showed up in bladder cancer, where BRCA2-related risk appeared much higher in women than in men.
- The findings could shape future screening and treatment research, but they are not a green light to change medical practice yet.
For decades, mutations in the BRCA1 and BRCA2 genes have been synonymous with breast and ovarian cancer. These are the genes behind Angelina Jolie’s widely publicized preventive surgery and the revolution in genetic testing that followed. Now, a large study out of Japan found significant associations between harmful BRCA mutations and four less common cancers: thyroid, bladder, head and neck, and skin cancers.
The research, published in ESMO Open, drew on a national Japanese biobank of more than 42,000 people. Carriers of harmful BRCA mutations faced anywhere from roughly four to six times the normal odds of developing one of these four cancers, and the risks held up even after researchers accounted for lifestyle factors like smoking and drinking. Because the study was conducted entirely in a Japanese population, the findings will need to be confirmed in other groups before clinicians can act on them broadly.
Bladder and head and neck cancers, in particular, are known for poor prognoses when caught late. Previous research has shown metastatic cases of these cancers can carry five-year survival rates as low as 5% to 20%, and more than half of head and neck cancer patients are already in advanced stages by the time of diagnosis. If inherited BRCA mutations are partly driving these cancers, that raises the possibility, though not yet the certainty, of earlier detection and a stronger case for testing drugs like PARP inhibitors that are already reshaping treatment for breast and ovarian cancer.

How the BRCA Study Worked
Researchers drew on BioBank Japan, a large national registry that collected blood samples and health information from people across the country between 2003 and 2018. The team focused on nine types of cancer considered less common, including bladder, bone, brain, head and neck, soft tissue, skin, testis, thyroid, and ureteral cancer. They compared the genetics of 3,489 patients diagnosed with at least one of these cancers against 38,842 people who had no cancer diagnosis at all. Participants reported their own cancer histories, which introduces the possibility of error, though the authors argue major bias is unlikely because people reported diagnoses without knowing their BRCA status.
The scientists zeroed in on the portions of DNA that contain the BRCA1 and BRCA2 genes, identifying 105 harmful genetic variants among 994 total variants found. Variant classification followed criteria established by an international consortium dedicated to interpreting the clinical meaning of BRCA mutations.
To determine whether carriers of these mutations were more likely to develop certain cancers, the team used a rigorous analytical approach that adjusted for age, sex, and the hospital where participants were enrolled. They set a strict threshold for declaring a result meaningful, applying a well-established correction for the fact that they were testing multiple cancer types at once, a common safeguard to prevent false alarms.
Cancers Linked to BRCA Mutations
After that statistical filtering, four clear associations emerged. Harmful BRCA1 mutations were associated with thyroid cancer, with carriers facing about 5.25 times the odds of developing the disease compared to non-carriers. Harmful BRCA2 mutations were tied to three cancers: bladder cancer (about 4.67 times the odds), head and neck cancer (about 3.89 times the odds), and skin cancer (about 6.13 times the odds).
A suggestive link also appeared between BRCA2 mutations and thyroid cancer, but it didn’t clear the study’s strict threshold, meaning it needs further investigation before being considered reliable.
These associations held up across several additional analyses. When the researchers adjusted for smoking history, drinking history, or both, the results remained essentially unchanged, suggesting these genetic risks aren’t simply reflecting lifestyle habits.
Perhaps the most provocative finding involved bladder cancer and the starkly different risks faced by women versus men carrying BRCA2 mutations. Among female carriers, the odds of bladder cancer were about 23.6 times higher than in non-carriers. Among male carriers, that figure dropped to about 2.35 times. The difference between sexes was statistically meaningful. But the authors caution that only seven female carriers and six male carriers appeared in the bladder cancer group, a small sample that demands careful interpretation before anyone reads too much into that 23.6-fold figure.
None of the female bladder cancer patients with BRCA2 mutations had a prior history of breast or ovarian cancer, meaning their BRCA status hadn’t been flagged through the usual channels. Whether that reflects gaps in testing, pure chance in a small group, or something else entirely is not something this study can answer.
Implications for Patients
The researchers offered a potential biological explanation, still a hypothesis rather than a proven mechanism, for why women might be hit harder by bladder cancer. Cystitis, a type of bladder infection and inflammation, is far more common in women. Scientific literature suggests chronic inflammation can fuel cancer development through DNA damage. People who already have a reduced ability to repair DNA damage because of BRCA2 mutations could face a compounding effect when repeated bladder infections enter the picture. The authors call this “another possibility” and say further studies are needed.
Lifetime risk estimates painted a clearer picture of what carrying these mutations could mean. For BRCA1 carriers, the estimated risk of developing thyroid cancer by age 85 was 10.1%. For BRCA2 carriers, the lifetime risk of skin cancer was estimated at 9.0%, head and neck cancer at 8.3%, and bladder cancer at 12.8% for women and 5.6% for men. To put those numbers in perspective, even single-digit lifetime risks are meaningful for cancers that are often caught late.
It bears emphasizing that these numbers are not yet a basis for changing clinical practice. The study authors themselves write that establishing screening criteria based on BRCA carrier status will require larger analyses with more detailed clinical information. Right now, people who learn they carry BRCA mutations are typically counseled about breast, ovarian, pancreatic, and prostate cancer risks, and that guidance isn’t going to shift overnight.
Beyond screening, the results could strengthen the case for clinical trials testing PARP inhibitors in bladder and head and neck cancers. These drugs work by exploiting the DNA-repair weaknesses in BRCA-mutated cells. Some early research has already shown activity in bladder cancer patients with DNA repair gene mutations, and lab-based work has explored their potential in head and neck cancer. Establishing a firmer genetic link between BRCA mutations and these cancers adds fuel to that effort.
Previous studies looking at BRCA risks and less common cancers were limited by small sample sizes, often including fewer than 200 cases of any given cancer type. This study included more than 300 cases for each of the cancer types that showed meaningful associations, giving the results considerably more statistical weight.
A bigger picture emerges from the data: the medical community may be underestimating how many types of cancer are influenced by BRCA mutations. Each new association discovered adds another reason for carriers to stay watchful and another potential avenue for treatment. For cancers that respond poorly to existing therapies when caught late, genetic insights like these could eventually save lives, pending the confirmatory research the authors themselves say is needed.
Disclaimer: This article summarizes a peer-reviewed study and is intended for general informational purposes. It does not constitute medical advice. The research was conducted in a Japanese population, and the findings may not translate directly to other groups. People concerned about inherited cancer risk, BRCA testing, or personal or family cancer history should consult a qualified healthcare provider or genetic counselor. Clinical guidelines for BRCA-related cancer screening have not changed based on this study.
Paper Notes
Limitations
The study relied on self-reported medical histories from participants, which introduces the possibility of information bias and misclassification. The researchers noted that participants reported their cancer diagnoses without knowledge of their genetic carrier status, making it unlikely that the reporting itself was skewed by that knowledge. Previous studies using BioBank Japan data have also shown that self-reported and physician-based diagnoses produced comparable genetic evidence. Additionally, limited clinical information meant the researchers could not fully evaluate whether BRCA-associated cancers were more aggressive or lethal than non-BRCA cases. To partially address this, they referenced data from the Center for Cancer Genomics and Advanced Therapeutics (C-CAT), which collects information on patients with advanced cancer after standard treatment. Among 122 advanced bladder cancer patients in that dataset, three were BRCA2 carriers. One died approximately two years after diagnosis, while the other two survived three to four years and remained alive. No BRCA1 carriers were found among 158 advanced thyroid cancer patients after standard treatment in C-CAT. The sex-stratified analyses involved small numbers of carriers in some subgroups, which the authors acknowledged limits statistical power and requires cautious interpretation. The study also drew exclusively from a Japanese population, so whether these risk estimates translate to other groups remains an open question.
Funding and Disclosures
This work was supported by the Japan Agency for Medical Research and Development and the Australian National Health and Medical Research Council. The authors declared no conflicts of interest. The funders did not play a role in study design, data collection, analysis, interpretation, manuscript writing, or the decision to submit for publication.
Publication Details
The paper, titled “BRCA1 and BRCA2 pathogenic variants increase the risk of four less common cancer types,” was published in ESMO Open, Volume 11, Issue 4, 2026. Authors include H. Sasagawa, M. Endo, Y. Iwasaki, Y. Usui, Y. N. Koyanagi, G. Innella, J. Hadler, M. T. Parsons, K. Numakura, Y. Kamatani, Y. Murakami, K. Matsuo, K. Matsuda, A. B. Spurdle, T. Habuchi, and Y. Momozawa. The corresponding author is Yukihide Momozawa at the RIKEN Center for Integrative Medical Sciences in Yokohama, Japan. Institutional affiliations span RIKEN, Akita University Graduate School of Medicine, Aichi Cancer Center, QIMR Berghofer Medical Research Institute (Brisbane, Australia), the University of Bologna (Italy), the University of Tokyo, and Nagoya University Graduate School of Medicine. The DOI is 10.1016/j.esmoop.2026.106900.







